Advanced Synthetic Route for Dienogest: Enhancing Purity and Commercial Scalability for Global Pharma
The pharmaceutical industry's demand for high-purity hormonal agents continues to drive innovation in synthetic methodology, particularly for complex steroids like dienogest. Patent CN101863947A introduces a transformative approach to synthesizing this potent progestin, addressing long-standing inefficiencies in yield and environmental impact. By utilizing estra-4,9-diene-3,17-dione as a readily available starting material and implementing a robust protection strategy, this method offers a viable pathway for commercial scale-up of complex steroid intermediates. The core innovation lies in the strategic use of 2,2-dimethyl-1,3-propanediol to protect the C3-ketone, a modification that fundamentally alters the reaction landscape by enhancing stability during harsh basic conditions required for subsequent functionalization.
For R&D directors evaluating process viability, the significance of this patent cannot be overstated. Traditional routes often suffer from poor atom economy and difficult purification profiles, leading to bottlenecks in production. This new methodology not only streamlines the synthetic sequence but also ensures that the critical epoxidation step proceeds with exceptional efficiency. As a reliable pharmaceutical intermediates supplier, understanding these mechanistic nuances is essential for guaranteeing batch-to-batch consistency and meeting the stringent purity specifications required by global regulatory bodies.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of dienogest has been plagued by reliance on cumbersome starting materials and inefficient reaction sequences. One prominent legacy route, often cited in earlier literature, utilizes estrone-3-methyl ether as the precursor. This pathway necessitates a Birch reduction followed by epoxidation and cyanide ring opening. However, this approach is fraught with challenges; the Birch reduction requires cryogenic conditions and hazardous reagents like liquid ammonia, posing significant safety risks and operational complexities. Furthermore, the overall yield of this route is notoriously low, primarily because the transformation of the 17-position ketone is performed before the hydrolysis of the 3-position methyl ether, creating a disjointed and inefficient workflow.
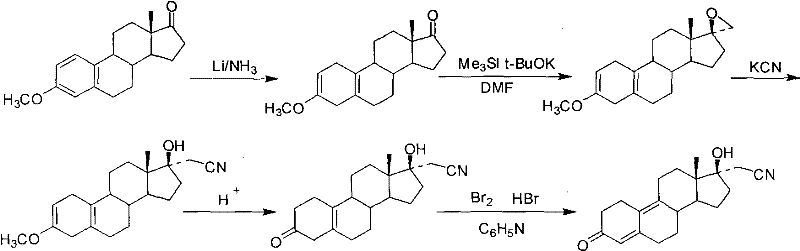
Another conventional strategy involves the use of 5(10)-estrene-3,17-dione, where the C3-ketone is protected prior to epoxidation. While conceptually sound, this method fails in execution due to the instability of the protecting group. When standard protecting groups like propylene glycol are employed, they cannot withstand the strong alkaline conditions necessary for the epoxidation reaction. This incompatibility leads to premature deprotection and side reactions, resulting in dismal yields for the epoxy intermediate. Consequently, manufacturers face high production costs and extensive waste generation, making these legacy routes economically unviable for large-scale cost reduction in API manufacturing.
The Novel Approach
The methodology disclosed in CN101863947A represents a paradigm shift by introducing a chemically robust protecting group that survives the rigors of the synthetic sequence. By employing 2,2-dimethyl-1,3-propanediol, the process creates a ketal at the C3-position that is uniquely resistant to strong bases. This stability allows the subsequent epoxidation at the C17-position to proceed smoothly using trimethylsulfonium iodide and potassium tert-butoxide without compromising the integrity of the molecule. The result is a dramatic improvement in the yield of the epoxy intermediate, which serves as the pivotal junction for introducing the cyanomethyl group.
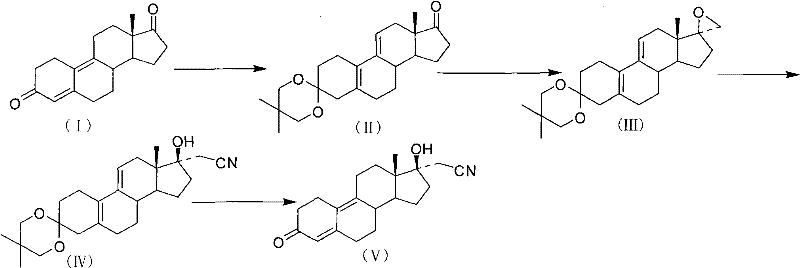
Furthermore, this novel approach addresses the critical issue of waste management. In previous iterations, the workup required diluting the reaction mixture into vast quantities of saturated sodium bicarbonate to neutralize acids, generating excessive wastewater. The patented process innovates here by adjusting the pH to alkaline using a base directly before standard extraction procedures. This simple yet effective modification significantly reduces the volume of effluent, aligning the synthesis with modern environmental compliance standards and reducing the burden on waste treatment facilities.
Mechanistic Insights into Alkali-Stable Ketone Protection
The success of this synthetic route hinges on the electronic and steric properties of the 2,2-dimethyl-1,3-propanediol protecting group. In organic synthesis, ketals are commonly used to protect carbonyl functionalities, but their stability varies widely depending on the substitution pattern. The gem-dimethyl substitution on the propane backbone introduces significant steric hindrance around the acetal carbon. This steric bulk shields the C-O bonds from nucleophilic attack and base-catalyzed hydrolysis. During the epoxidation step, where strong bases like t-BuOK are present to generate the sulfur ylide, a standard ketal might undergo retro-aldol type decomposition or hydrolysis. However, the dimethyl-substituted ketal remains intact, ensuring that the C3-carbonyl is available for the final deprotection step only when intended.
This mechanistic robustness directly translates to impurity control. In less stable systems, partial deprotection during the epoxidation or cyanation steps can lead to a complex mixture of byproducts, including those where the C3-ketone reacts with the cyanide or the epoxide reagent. These impurities are structurally similar to the target molecule and are notoriously difficult to separate via crystallization or chromatography. By maintaining the protecting group throughout the harsh middle stages of the synthesis, the process ensures a cleaner reaction profile. The final deprotection using acid (such as perchloric acid or hydrochloric acid in acetone) is highly selective, cleaving the ketal to reveal the conjugated enone system of dienogest without affecting the newly installed 17α-cyanomethyl group.
How to Synthesize Dienogest Efficiently
The synthesis of dienogest via this patented route is a streamlined four-step process designed for industrial scalability. It begins with the selective protection of the starting dione, followed by a high-yielding epoxidation and cyanation sequence, and concludes with a clean deprotection. The operational simplicity, combined with the use of common solvents like THF, DMF, and ethanol, makes this route highly attractive for manufacturing. Detailed standardized synthesis steps, including specific molar ratios and temperature controls derived from the patent examples, are provided in the guide below to assist technical teams in replicating this high-efficiency pathway.
- Selective protection of the C3-ketone group in estra-4,9-diene-3,17-dione using 2,2-dimethyl-1,3-propanediol and acetyl chloride at low temperatures (-40°C).
- Epoxidation at the C17 position using trimethylsulfonium iodide and a strong base (e.g., potassium tert-butoxide) in DMF, leveraging the stability of the protecting group.
- Ring-opening cyanation using sodium cyanide in methanol/ethanol, followed by acidic deprotection to yield the final dienogest API.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the transition to this optimized synthetic route offers tangible benefits beyond mere chemical elegance. The primary advantage lies in the accessibility of the starting material, estra-4,9-diene-3,17-dione. Unlike the specialized precursors required for Birch reduction routes, this dione is a commodity steroid intermediate available from multiple global sources. This diversification of supply sources mitigates the risk of raw material shortages and provides leverage in price negotiations, ensuring a more resilient supply chain for high-purity dienogest.
- Cost Reduction in Manufacturing: The most significant economic driver of this process is the drastic improvement in yield during the critical epoxidation step. In legacy processes, yields for this transformation were often suboptimal due to protecting group failure, necessitating the processing of larger batches of expensive starting materials to achieve the same output. By securing yields exceeding 90% in this step, the new method maximizes the throughput of existing reactor capacity. Additionally, the elimination of complex cryogenic Birch reduction equipment reduces capital expenditure and energy consumption, leading to substantial operational cost savings without compromising product quality.
- Enhanced Supply Chain Reliability: The robustness of the chemistry directly correlates to supply reliability. Processes that rely on unstable intermediates or hazardous reagents like liquid ammonia are prone to unplanned downtime and safety shutdowns. This patented route operates under relatively mild conditions (mostly ambient to moderate heating) and uses stable reagents. The simplified workup procedure, which avoids the generation of massive wastewater volumes, also reduces the logistical burden on plant utilities. This operational stability ensures consistent delivery schedules, reducing lead time for high-purity hormonal APIs and allowing partners to maintain leaner inventory levels.
- Scalability and Environmental Compliance: As regulatory scrutiny on pharmaceutical manufacturing intensifies, the environmental footprint of a synthesis route becomes a key selection criterion. The modified workup procedure described in the patent significantly lowers the E-factor (mass of waste per mass of product) by avoiding excessive dilution. This reduction in wastewater volume simplifies effluent treatment and lowers disposal costs. Furthermore, the high selectivity of the reaction minimizes the formation of toxic byproducts, facilitating easier purification and ensuring that the final API meets rigorous international safety standards for residual solvents and impurities.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis route. These insights are derived directly from the experimental data and claims within patent CN101863947A, providing a factual basis for decision-making. Understanding these details is crucial for technical teams assessing the feasibility of technology transfer and for procurement specialists evaluating the long-term viability of the supply source.
Q: Why is the 2,2-dimethyl-1,3-propanediol protecting group superior for dienogest synthesis?
A: Unlike traditional propylene glycol protectors, the 2,2-dimethyl variant offers superior steric bulk and alkali resistance. This stability is critical during the subsequent epoxidation step which requires strong bases, preventing premature deprotection and significantly boosting the yield of the epoxy intermediate.
Q: How does this patent address environmental concerns in steroid manufacturing?
A: The process optimizes the workup procedure by adjusting the reaction pH to alkaline using base prior to standard operations, rather than diluting the reaction mixture into massive volumes of saturated sodium bicarbonate. This modification drastically reduces the volume of wastewater generated, aligning with modern green chemistry standards.
Q: What are the typical yields for the critical epoxidation step in this route?
A: According to the experimental data in patent CN101863947A, the epoxidation step achieves yields between 90.7% and 96.9%. This is a substantial improvement over previous methods where yields were compromised by the instability of the protecting group under basic conditions.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Dienogest Supplier
The synthesis of dienogest described in CN101863947A exemplifies the kind of process innovation that drives value in the modern pharmaceutical supply chain. At NINGBO INNO PHARMCHEM, we specialize in translating such patented methodologies into commercial reality. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the high yields observed in the lab are maintained at an industrial scale. We operate state-of-the-art facilities equipped with rigorous QC labs capable of verifying stringent purity specifications for every batch, guaranteeing that our dienogest intermediates meet the exacting standards of global regulatory agencies.
We invite potential partners to engage with our technical team to explore how this optimized route can benefit your specific supply chain needs. By collaborating with us, you gain access to a Customized Cost-Saving Analysis tailored to your volume requirements. We encourage you to contact our technical procurement team today to request specific COA data and comprehensive route feasibility assessments, ensuring a seamless integration of high-quality dienogest into your manufacturing pipeline.
