Advanced Catalytic Strategy for Manufacturing High-Purity Losartan Intermediates at Scale
Advanced Catalytic Strategy for Manufacturing High-Purity Losartan Intermediates at Scale
The pharmaceutical landscape for antihypertensive agents continues to evolve, with Losartan Potassium remaining a cornerstone therapy as the first non-peptide, orally effective angiotensin II (AT1) receptor antagonist. However, the manufacturing of its free acid form, Losartan, has historically faced challenges regarding purity profiles and physical appearance, which directly impact the quality of the final potassium salt. Patent CN107056756B introduces a transformative approach to this synthesis, leveraging a specific class of cyclic amine catalysts to achieve unprecedented purity levels. This technical breakthrough addresses the critical need for reliable pharmaceutical intermediate suppliers who can deliver materials with minimal impurity burdens. By shifting away from conventional ammonium or zinc salt catalysts, this new methodology not only enhances the chemical integrity of the product but also streamlines the downstream processing required for GMP-grade API production.
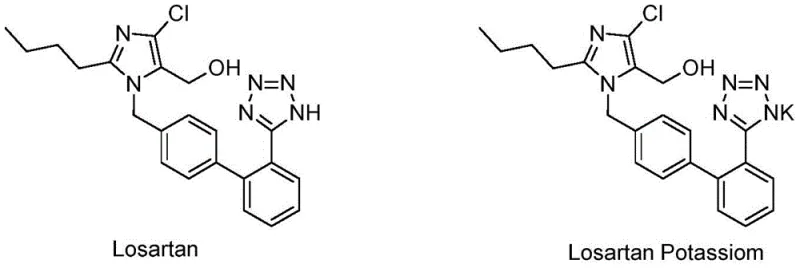
In the context of global supply chains, the ability to produce high-purity Losartan with a single impurity profile reduced to as low as 0.2% represents a significant competitive advantage. The patent details a process where the final product achieves a purity of up to 98.4%, coupled with excellent physical appearance, thereby reducing the necessity for resource-intensive decolorization steps often mandated by older protocols. For R&D directors and procurement managers, this implies a more robust starting material that minimizes the risk of batch failures in subsequent salt formation stages. The strategic implementation of this catalytic system offers a pathway to cost reduction in pharmaceutical intermediates manufacturing by simplifying the overall process flow while simultaneously elevating quality standards to meet stringent regulatory requirements.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of Losartan has relied heavily on catalysts such as ammonium salts, zinc salts, or triethylamine salts to facilitate the critical conversion of the nitrile group to the tetrazole ring. While these traditional reagents are chemically functional, they frequently result in suboptimal outcomes, typically yielding Losartan with purity levels hovering between 96% and 97%. More critically, the physical appearance of the product obtained through these conventional routes is often poor, characterized by discoloration that necessitates multiple rounds of decolorization using activated carbon or similar adsorbents during the subsequent synthesis of Losartan Potassium. These additional purification steps not only increase the consumption of auxiliary materials but also introduce potential yield losses and extend the overall production cycle time. Furthermore, the presence of residual metal ions from zinc-based catalysts can pose significant regulatory hurdles, requiring rigorous testing and validation to ensure compliance with heavy metal limits in the final drug substance.
The Novel Approach
The innovative methodology disclosed in the patent data circumvents these historical bottlenecks by employing a specialized catalyst system based on cyclic amines, specifically those conforming to Formula A where R1 and R2 groups vary among hydrogen, alkyl, or aryl substituents. This novel approach fundamentally alters the reaction environment, promoting a cleaner conversion of the chloronitrile precursor (Compound 1) into the target tetrazole structure. By utilizing catalysts such as pyrrolidine or piperidine derivatives in conjunction with an acid to form an active salt in situ, the process achieves a reaction efficiency that translates directly into higher crude purity. The resulting Losartan exhibits superior appearance and purity metrics right out of the crystallization step, effectively eliminating the need for the cumbersome decolorization procedures associated with older technologies. This shift represents a paradigm change in cost reduction in API manufacturing, as it removes entire unit operations from the production line while enhancing the consistency of the output.
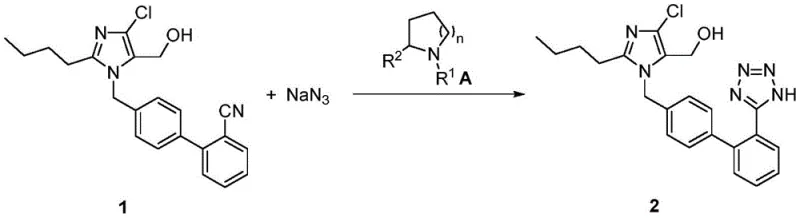
Mechanistic Insights into Amine-Catalyzed Tetrazole Formation
The core of this technological advancement lies in the mechanistic role of the cyclic amine catalyst during the [2+3] cycloaddition reaction between the nitrile group of Compound 1 and sodium azide. In the proposed mechanism, the amine catalyst first reacts with the added acid to form a stable ammonium salt within the organic solvent matrix, typically toluene or xylene. This salt formation is crucial as it likely modifies the solubility profile of the reactants and may act as a phase-transfer facilitator or a specific activator for the azide ion. The presence of tetrabutylammonium bromide further supports this phase-transfer dynamic, ensuring that the inorganic sodium azide is sufficiently available in the organic phase to react with the lipophilic nitrile substrate. The thermal energy applied, raising the system to 95°C, drives the cycloaddition forward, closing the tetrazole ring with high regioselectivity. This precise control over the reaction environment minimizes the formation of side products, such as open-chain amidines or polymerized byproducts, which are common sources of impurities in less optimized systems.
Impurity control is further enhanced by the specific workup protocol designed to complement this catalytic system. Following the reaction, the mixture undergoes a controlled saponification and phase separation process where the basic conditions help to neutralize acidic byproducts and partition impurities into the aqueous layer. The subsequent acidification step is carefully managed, adjusting the pH first to 7.0 and then to 4.0 to induce crystallization. This gradual pH adjustment allows for the selective precipitation of the target Losartan molecule while keeping soluble impurities in the mother liquor. The use of ethyl acetate as a dispersion and washing solvent aids in removing residual organic impurities without dissolving the product crystal lattice. This multi-stage purification strategy, inherent to the new process design, ensures that the final solid possesses the reported 98.4% purity and 0.2% single impurity specification, providing a level of chemical homogeneity that is essential for downstream pharmaceutical formulation.
How to Synthesize Losartan Efficiently
The synthesis of Losartan via this advanced catalytic route offers a streamlined alternative to legacy processes, focusing on operational simplicity and high yield recovery. The procedure begins with the preparation of the active catalytic species in an aromatic solvent, followed by the sequential addition of the chloronitrile precursor and azide source under heated conditions. The robustness of this method allows for extended reaction times without significant degradation, ensuring complete conversion of the starting material. Detailed standardized synthetic steps for implementing this protocol in a pilot or production setting are outlined below, providing a clear roadmap for technical teams aiming to adopt this superior manufacturing technology.
- Prepare the catalytic system by adding a cyclic amine catalyst (Formula A) to an organic solvent like toluene, followed by dropwise addition of acid to form the active salt.
- Introduce the chloronitrile precursor (Compound 1), sodium azide, and tetrabutylammonium bromide to the system, then heat to 95°C to facilitate the tetrazole ring formation.
- Perform post-reaction workup involving saponification with sodium hydroxide, separation of phases, acidification with hydrochloric acid, and crystallization to isolate high-purity Losartan.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this novel catalytic process presents a compelling value proposition centered on operational efficiency and risk mitigation. The primary advantage lies in the substantial simplification of the purification workflow. By eliminating the need for multiple decolorization steps, manufacturers can significantly reduce the consumption of filtering aids and activated carbon, which are recurring cost centers in traditional API production. This reduction in auxiliary material usage translates directly into lower variable costs per kilogram of produced Losartan. Moreover, the higher intrinsic purity of the crude product reduces the likelihood of batch rejections or the need for re-processing, thereby stabilizing production schedules and improving overall equipment effectiveness. The reliability of the supply chain is further bolstered by the use of commercially available and inexpensive catalysts like pyrrolidine and piperidine derivatives, which do not suffer from the supply volatility often seen with specialized transition metal complexes.
- Cost Reduction in Manufacturing: The economic benefits of this process are driven by the elimination of expensive and time-consuming purification stages. Traditional methods often require extensive post-reaction treatments to remove colored impurities and metal residues, which incur both material and labor costs. By achieving high purity directly from the crystallization step, this new method drastically reduces the operational expenditure associated with downstream processing. Additionally, the improved yield reported in the patent examples suggests a more efficient utilization of the valuable chloronitrile precursor, minimizing raw material waste. This efficiency gain is critical for maintaining competitive pricing in the generic pharmaceutical market, where margin pressures are constant.
- Enhanced Supply Chain Reliability: The reliance on simple, commodity-grade reagents such as toluene, sodium azide, and cyclic amines ensures a resilient supply chain. Unlike processes dependent on proprietary or scarce catalysts, this method utilizes chemicals that are widely available from multiple global suppliers, reducing the risk of supply disruptions. The robustness of the reaction conditions, which tolerate standard heating and stirring protocols without requiring exotic equipment like high-pressure reactors, further enhances manufacturability. This accessibility allows for flexible sourcing strategies and easier scale-up across different production sites, ensuring consistent availability of high-purity Losartan for downstream customers.
- Scalability and Environmental Compliance: From an environmental and scalability perspective, the process offers distinct advantages. The reduction in purification steps inherently lowers the volume of solid waste generated, particularly spent carbon and filtration cakes contaminated with heavy metals. This aligns with increasingly stringent environmental regulations regarding waste disposal in chemical manufacturing. Furthermore, the use of standard organic solvents that can be recovered and recycled contributes to a greener process footprint. The simplicity of the operation, involving standard agitation and temperature control, makes the technology highly scalable from pilot batches to multi-ton commercial production, facilitating rapid capacity expansion to meet market demand without compromising on quality or safety standards.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this high-purity Losartan synthesis method. These insights are derived directly from the patent specifications and are intended to clarify the operational benefits and chemical principles underlying this advanced manufacturing route. Understanding these details is crucial for technical teams evaluating the feasibility of integrating this process into their existing production capabilities.
Q: How does the novel amine catalyst improve Losartan purity compared to traditional methods?
A: Traditional methods using ammonium or zinc salts often yield Losartan with 96-97% purity and poor appearance, requiring multiple decolorization steps. The novel cyclic amine catalysts described in CN107056756B enable direct synthesis with purity up to 98.4% and single impurities reduced to 0.2%, eliminating the need for extensive purification.
Q: What are the key operational parameters for the tetrazole formation step?
A: The reaction requires heating the mixture of chloronitrile, sodium azide, and catalyst to approximately 95°C for about 42 hours. Critical controls include maintaining a molar ratio of Compound 1 to sodium azide between 1:2.0 and 1:2.5, and using tetrabutylammonium bromide as a phase transfer facilitator.
Q: Is this synthesis method suitable for large-scale industrial production?
A: Yes, the method is explicitly designed for industrial scalability. It utilizes common organic solvents like toluene or xylene and avoids complex transition metal catalysts that require difficult removal steps. The simplified workup procedure involving standard acid-base extraction and crystallization ensures robust supply chain continuity.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Losartan Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of adopting cutting-edge synthetic methodologies to maintain a competitive edge in the global pharmaceutical market. Our team of expert chemists has thoroughly analyzed the potential of the catalytic system described in CN107056756B and is well-positioned to translate this laboratory-scale innovation into commercial reality. We possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from pilot studies to full-scale manufacturing is seamless and efficient. Our state-of-the-art facilities are equipped with rigorous QC labs capable of verifying stringent purity specifications, guaranteeing that every batch of Losartan we produce meets the highest international standards for pharmaceutical intermediates.
We invite you to collaborate with us to leverage this advanced technology for your supply chain needs. By partnering with our technical procurement team, you can access a Customized Cost-Saving Analysis tailored to your specific volume requirements and quality targets. We encourage you to reach out today to request specific COA data and route feasibility assessments, allowing us to demonstrate how our commitment to innovation can drive value and reliability in your Losartan procurement strategy.
