Revolutionizing Initiator 819 Production: A Safer, High-Yield Ether-Complexing Strategy for Commercial Scale-Up
The chemical industry's demand for high-efficiency photoinitiators, particularly for UV-curing applications in coatings, inks, and advanced composite materials, has necessitated a rigorous re-evaluation of synthetic pathways for key compounds like bis(2,4,6-trimethylbenzoyl)phenylphosphine oxide, commonly known as Initiator 819. Patent CN112159429A introduces a transformative preparation method that fundamentally alters the reaction landscape by replacing hazardous proton sources with specific ether-based complexing agents. This innovation addresses long-standing issues regarding process safety, environmental impact, and yield consistency that have plagued traditional manufacturing protocols. By integrating a polar aprotic ether into the sodium sand reduction step, the process effectively stabilizes reactive intermediates, preventing the formation of hazardous byproducts and ensuring a more robust supply chain for this critical polymer additive. For R&D directors and procurement specialists, understanding this mechanistic shift is crucial for securing a reliable photoinitiator supplier capable of delivering high-purity materials without the baggage of legacy safety risks.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of acyl phosphine oxides has relied heavily on the use of proton sources such as alcohols or amines to dissociate intermediates, a practice that introduces severe operational liabilities. These conventional routes typically require more than two equivalents of dissociating agents, which are not recyclable and contribute significantly to the organic load in wastewater streams, complicating environmental compliance and waste treatment costs. Furthermore, the presence of proton sources often facilitates the generation of phenylphosphine gas, a substance notorious for its intense, unpleasant odor and high flammability, creating substantial potential safety hazards in large-scale reactor environments. Additionally, when activators like n-butanol are employed, they frequently engage in competitive side reactions with the acyl chloride starting material, leading to the formation of ester byproducts that consume valuable raw materials and necessitate complex, costly purification steps to achieve the required purity standards for electronic or optical applications.
The Novel Approach
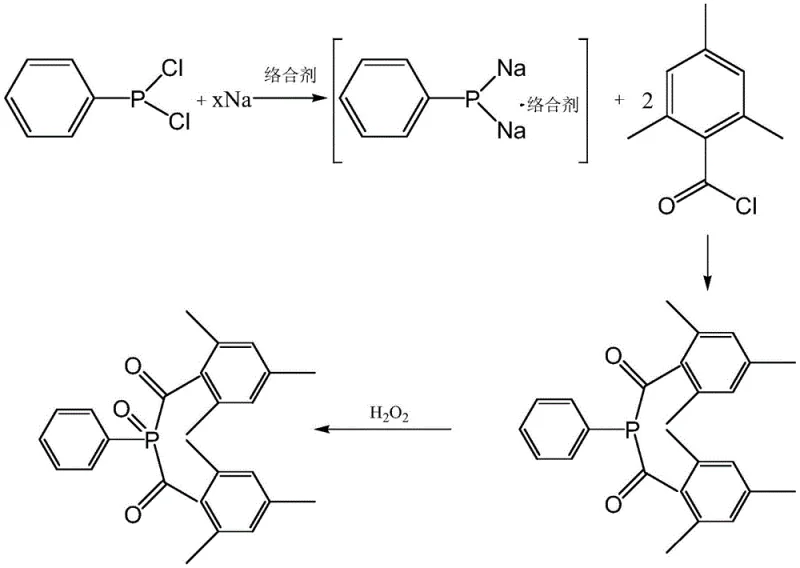
In stark contrast, the methodology disclosed in the patent utilizes a specific class of ether compounds acting as complexing agents to mediate the reaction between sodium sand and phenylphosphonic dichloride. This strategic addition effectively inhibits the formation of sodium phenylphosphonate polyphosphates, which are clustered intermediates containing undesirable P-P bonds that derail the synthesis. By stabilizing the monomeric sodium phenylphosphonate species through coordination, the process promotes the forward reaction towards the desired acyl phosphine intermediate without ever generating free phenylphosphine gas. This elimination of the phenylphosphine hydrogen process not only drastically improves workplace safety by removing toxic and flammable vapors but also simplifies the downstream processing. The use of polar aprotic solvents in low dosages further ensures that the organic content in the resulting wastewater is minimized, offering a greener, more sustainable manufacturing profile that aligns with modern environmental regulations.
Mechanistic Insights into Ether-Complexing Stabilization
The core innovation of this synthesis lies in the coordination chemistry between the ether oxygen atoms and the sodium cations of the intermediate species. When sodium sand reacts with phenylphosphonic dichloride in a nonpolar solvent like toluene, the nascent sodium phenylphosphonate tends to aggregate into insoluble clusters or polyphosphates if left unstabilized. The introduction of ethers such as tetrahydrofuran, dioxane, or anisole provides lone pair electrons that coordinate with the sodium ions, forming a stable solvated complex. This solvation shell prevents the phosphorus centers from approaching each other closely enough to form P-P bonds, thereby maintaining the intermediate in a reactive, monomeric state. This stable solvated species is then primed for rapid nucleophilic attack on the mesitylene benzoyl chloride, accelerating the acylation step and ensuring high conversion rates. The mechanism essentially creates a kinetic pathway that favors the desired product formation while thermodynamically disfavoring the aggregation into inactive polymeric phosphorus species.
Furthermore, the control of impurity profiles is intrinsically linked to the efficiency of this complexation. In traditional methods, the slow reaction of unstable intermediates allows time for side reactions, such as the hydrolysis of acyl chlorides or the formation of anhydrides, to occur. By accelerating the reaction rate of the sodium phenylphosphonate intermediate with the acyl chloride through effective complexing, the window of opportunity for these side reactions is significantly narrowed. The patent data indicates that without the complexing agent, yields plummet to around 40%, whereas the optimized ether-complexing route consistently achieves yields exceeding 90% with purity levels above 99%. This dramatic improvement in selectivity means that the final crystallization steps are far more efficient, requiring less solvent and energy to remove trace impurities, which is a critical factor for maintaining cost-effectiveness in the production of high-value fine chemical intermediates.
How to Synthesize Bis(2,4,6-trimethylbenzoyl)phenylphosphine Oxide Efficiently
The synthesis protocol outlined in the patent provides a robust framework for producing Initiator 819 with industrial viability. The process begins with the careful preparation of a sodium sand suspension in a nonpolar solvent under an inert nitrogen atmosphere, ensuring maximum surface area for the reduction reaction. Following the initial reaction with phenylphosphonic dichloride, the critical addition of the ether complexing agent occurs, which must be timed and dosed precisely to maximize intermediate stability without oversaturating the system. Subsequent acylation with mesitylene benzoyl chloride and final oxidation with hydrogen peroxide complete the transformation. For detailed operational parameters, temperature controls, and specific molar ratios required to replicate this high-yield process, please refer to the standardized synthesis guide below.
- Prepare a sodium sand suspension in a nonpolar solvent like toluene under inert atmosphere at 90-110°C, then react with phenylphosphonic dichloride.
- Introduce a specific ether complexing agent (e.g., THF, dioxane) to stabilize the sodium phenylphosphonate intermediate and prevent P-P bond formation.
- React the stabilized intermediate with mesitylene benzoyl chloride, followed by oxidation with hydrogen peroxide and recrystallization to obtain the final oxide product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the transition to this ether-complexing technology represents a strategic opportunity to de-risk the supply of critical photoinitiators. The elimination of hazardous phenylphosphine gas generation removes a major bottleneck related to safety compliance and reactor downtime, ensuring more consistent production schedules and reliable delivery timelines. Moreover, the reduction in wastewater organic load translates directly into lower environmental treatment costs and reduced regulatory burden, which are increasingly significant factors in the total cost of ownership for chemical manufacturing. The ability to achieve high yields with fewer side products also implies a more efficient use of raw materials, reducing the volatility associated with feedstock pricing and availability.
- Cost Reduction in Manufacturing: The novel process eliminates the need for excessive amounts of proton sources and avoids the generation of difficult-to-remove ester byproducts that plague traditional methods. By preventing the formation of polyphosphate clusters, the reaction efficiency is maximized, meaning less raw material is wasted in side reactions. This qualitative improvement in atom economy and process efficiency leads to substantial cost savings in both material consumption and downstream purification efforts, without the need for expensive catalyst removal steps often associated with transition metal alternatives.
- Enhanced Supply Chain Reliability: Safety is a primary driver of supply continuity; by removing the generation of flammable and malodorous phenylphosphine gas, the manufacturing process becomes inherently safer and less prone to shutdowns due to safety incidents or regulatory inspections. The use of common, commercially available ether solvents and nonpolar hydrocarbons ensures that the supply chain is not dependent on niche or volatile reagents. This robustness allows for scalable production that can respond flexibly to market demand fluctuations, securing a steady flow of high-purity photoinitiator intermediates for downstream customers.
- Scalability and Environmental Compliance: The process is designed with scalability in mind, utilizing standard unit operations such as reflux, liquid-liquid separation, and crystallization that are easily adapted from pilot to commercial scale. The significant reduction in organic wastewater content simplifies effluent treatment, making it easier for manufacturing sites to maintain compliance with stringent environmental discharge standards. This environmental benefit not only mitigates the risk of fines but also enhances the corporate sustainability profile of the supply chain, a key metric for modern procurement strategies in the global chemical market.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this ether-complexing synthesis route. These insights are derived directly from the experimental data and comparative examples provided in the patent documentation, offering clarity on why this method outperforms legacy technologies. Understanding these nuances is essential for technical teams evaluating the feasibility of adopting this new standard for photoinitiator production.
Q: How does the ether-complexing method improve safety compared to traditional proton source methods?
A: Traditional methods often utilize alcohols or amines as proton sources, which can lead to the generation of malodorous and highly flammable phenylphosphine gas. The novel ether-complexing approach stabilizes the sodium phenylphosphonate intermediate directly, completely bypassing the phenylphosphine hydrogen generation step, thereby eliminating this significant safety hazard and odor issue.
Q: What represents the primary advantage of using cyclic ethers over naphthalene or chlorobenzene in this synthesis?
A: Cyclic ethers containing oxygen atoms, such as tetrahydrofuran or dioxane, act as superior ligands that effectively coordinate with sodium ions. This coordination inhibits the formation of unwanted sodium phenylphosphonate polyphosphates (intermediates with P-P bonds). In contrast, non-coordinating solvents like naphthalene fail to prevent these cluster formations, leading to significantly lower yields and difficult purification processes.
Q: Why is the control of the complexing agent dosage critical for reaction efficiency?
A: While the complexing agent is essential for stabilizing the intermediate, excessive amounts can overly solvate the sodium phenylphosphonate, effectively wrapping the negative ion center and hindering its nucleophilic attack on the acyl chloride. Conversely, insufficient amounts fail to prevent polyphosphate clustering. Therefore, maintaining a precise molar ratio is vital to balance stability with reactivity for optimal yield.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Bis(2,4,6-trimethylbenzoyl)phenylphosphine Oxide Supplier
As the global demand for high-performance UV-curing materials continues to surge, securing a partner with deep technical expertise in advanced synthesis methodologies is paramount. NINGBO INNO PHARMCHEM stands at the forefront of this evolution, leveraging cutting-edge process innovations like the ether-complexing strategy to deliver superior quality Initiator 819. Our facility boasts extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that we can meet your volume requirements without compromising on our stringent purity specifications. Supported by our rigorous QC labs, we guarantee that every batch meets the exacting standards required for sensitive applications in electronics, optics, and high-end coatings.
We invite you to collaborate with us to optimize your supply chain and reduce your overall manufacturing costs. Our technical team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume needs, demonstrating how our advanced process efficiencies translate into tangible value for your organization. Please contact our technical procurement team today to request specific COA data and route feasibility assessments, and let us demonstrate why we are the preferred partner for high-purity photoinitiator solutions.
