Advanced Cefdinir Manufacturing: Overcoming Traditional Limitations with Novel Activation Chemistry
Advanced Cefdinir Manufacturing: Overcoming Traditional Limitations with Novel Activation Chemistry
The pharmaceutical industry constantly seeks robust synthetic routes for broad-spectrum antibiotics that balance high purity with economic viability. Patent CN101817835A introduces a transformative preparation method for Cefdinir, a critical third-generation oral cephalosporin known for its stability against beta-lactamases. This innovation centers on the strategic use of Pentafluorophenol as an activating group for the side chain condensation, marking a significant departure from traditional, harsher activation methods. By optimizing the coupling of (Z)-2-(2-aminothiazole-4-yl)-2-triphenylmethyl iminoacetic acid with the core nucleus 7-AVCA, this technology addresses long-standing challenges in impurity control and process safety. For R&D directors and procurement specialists, understanding this shift is vital for securing a reliable cefdinir supplier capable of delivering consistent quality.
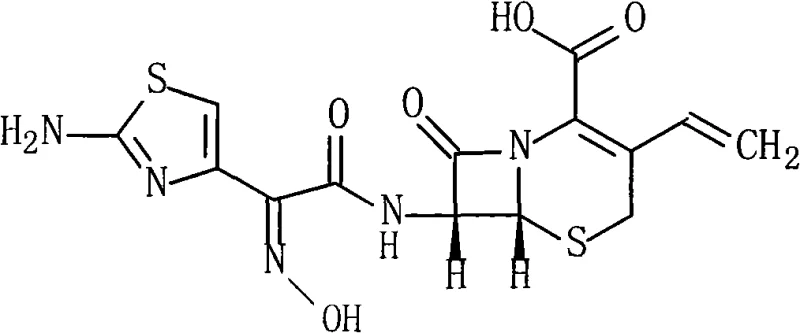
The structural integrity of Cefdinir, characterized by its vinyl group at the 3-position and the specific oximino acetoxyl side chain at the 7-position, dictates its biological activity. The patent details a pathway that preserves this delicate architecture while streamlining the assembly process. Unlike earlier methods that relied on aggressive chlorinating agents or complex heterocyclic activators, this approach utilizes the electron-withdrawing properties of the pentafluorophenyl group to facilitate nucleophilic attack under milder conditions. This not only enhances the stereochemical fidelity of the (6R, 7R) configuration but also simplifies the downstream purification, directly impacting the cost reduction in antibiotic manufacturing.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of Cefdinir has been plagued by operational complexities and environmental concerns associated with traditional activation strategies. Prior art, such as that disclosed in Chinese patent CN101182327A and various US patents, often employed Phosphorus Pentachloride (PCl5) to convert the carboxylic acid side chain into an acyl chloride. This necessitates strictly anhydrous conditions, which are energy-intensive to maintain and prone to failure on a large scale, leading to hydrolysis and reduced yields. Furthermore, other methods utilized 2-mercaptobenzimidazole (Acibenzolar) as an activating group, which, while effective, introduces significant challenges in post-reaction treatment. The removal of Acibenzolar residues requires rigorous purification steps, often resulting in the formation of new, difficult-to-separate impurities that compromise the final product's purity profile.
The Novel Approach
The methodology outlined in CN101817835A circumvents these pitfalls by employing Pentafluorophenol as the activating agent in the presence of triethylamine within a DMF solvent system. This substitution fundamentally alters the reaction landscape, relaxing the stringent requirement for absolute anhydrous conditions that characterize acyl chloride routes. The resulting pentafluorophenyl ester is sufficiently reactive to couple efficiently with the amine group of 7-AVCA at low temperatures (5-10°C), yet stable enough to minimize side reactions. This strategic choice eliminates the generation of corrosive phosphorus byproducts and avoids the introduction of sulfur-containing heterocyclic impurities, thereby streamlining the workflow. For supply chain heads, this translates to a more robust process with fewer failure points and a cleaner crude product profile.
Mechanistic Insights into Pentafluorophenol-Mediated Activation
The core of this synthetic advancement lies in the formation of an active ester intermediate through nucleophilic acyl substitution. In the initial step, the carboxylic acid of the trityl-protected thiazole side chain reacts with Pentafluorophenol, activated by triethylamine, to form a pentafluorophenyl ester. The strong electron-withdrawing nature of the five fluorine atoms on the phenyl ring significantly increases the electrophilicity of the carbonyl carbon, making it highly susceptible to nucleophilic attack by the 7-amino group of the 7-AVCA nucleus. This mechanism proceeds with high regioselectivity, ensuring that the acylation occurs exclusively at the desired position without affecting the sensitive beta-lactam ring or the vinyl group. The reaction is conducted at controlled low temperatures to further suppress potential racemization or degradation of the beta-lactam scaffold.
Following the coupling, the deprotection step utilizes formic acid under reflux conditions to cleave the trityl protecting group and the methoxyimino protecting group simultaneously. This dual deprotection is critical for revealing the free amino group on the thiazole ring and the carboxylic acid on the cephem nucleus, yielding the final zwitterionic Cefdinir molecule. The patent specifies precise pH adjustments during workup—first to neutrality to isolate the intermediate, and finally to acidic conditions (pH 2.4-2.8) to precipitate the pure product. This pH-controlled crystallization is a key mechanism for impurity rejection, as it exploits the solubility differences between the target molecule and unreacted starting materials or side products, ensuring the high-purity cefdinir required for pharmaceutical applications.
How to Synthesize Cefdinir Efficiently
The synthesis protocol described in the patent offers a streamlined two-step sequence that is amenable to standard reactor configurations found in modern fine chemical facilities. The process begins with the activation of the side chain acid, followed immediately by the addition of the cephem nucleus, minimizing the handling of unstable intermediates. The subsequent deprotection and isolation steps utilize common solvents like ethyl acetate and tetrahydrofuran, avoiding exotic or highly toxic reagents. This operational simplicity is a major factor in the commercial scale-up of complex cephalosporins, allowing manufacturers to transition from laboratory benchtop to multi-ton production with minimal re-engineering. Detailed standardized synthesis steps see the guide below.
- Activate (Z)-2-(2-aminothiazole-4-yl)-2-triphenylmethyl iminoacetic acid using Pentafluorophenol and triethylamine in DMF at 5-10°C.
- Couple the activated intermediate with 7-AVCA under vigorous stirring, adjust pH to precipitate the protected Cefdinir intermediate.
- Deprotect the intermediate using formic acid reflux, followed by pH adjustment and crystallization to obtain high-purity Cefdinir.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain strategists, the adoption of this Pentafluorophenol-based route offers compelling economic and logistical benefits over legacy technologies. The primary advantage stems from the elimination of hazardous and corrosive reagents like Phosphorus Pentachloride, which not only pose safety risks but also generate substantial waste streams requiring costly disposal. By replacing these with Pentafluorophenol, the process inherently reduces the environmental burden and the associated compliance costs, leading to substantial cost savings in waste management and regulatory reporting. Furthermore, the relaxation of anhydrous conditions lowers the energy consumption required for solvent drying and moisture control, contributing to a leaner manufacturing overhead.
- Cost Reduction in Manufacturing: The high yields reported in the patent embodiments, consistently exceeding 90% for the coupling step and 92% for the final deprotection, directly correlate to improved raw material efficiency. By maximizing the conversion of expensive starting materials like 7-AVCA and the thiazole acid derivative, the cost per kilogram of the active pharmaceutical ingredient is significantly optimized. Additionally, the avoidance of complex purification steps needed to remove Acibenzolar residues reduces solvent consumption and processing time, further driving down the variable costs associated with production.
- Enhanced Supply Chain Reliability: The robustness of this chemical route enhances supply continuity by reducing the risk of batch failures due to moisture ingress or reagent instability. Traditional acyl chloride methods are notoriously sensitive to trace water, which can lead to hydrolysis of the activated species and unpredictable yields. In contrast, the pentafluorophenyl ester approach is more forgiving, ensuring consistent output even in varying plant conditions. This reliability is crucial for maintaining steady inventory levels of high-purity cefdinir, preventing stockouts that could disrupt downstream formulation schedules for global pharmaceutical partners.
- Scalability and Environmental Compliance: From a sustainability perspective, this method aligns with green chemistry principles by minimizing the use of heavy metal catalysts and corrosive halogenating agents. The waste streams generated are less hazardous and easier to treat, facilitating compliance with increasingly stringent environmental regulations in major manufacturing hubs. The process is designed for scalability, with the patent demonstrating successful execution on gram to hundred-gram scales that translate linearly to industrial reactors. This ensures that suppliers can rapidly ramp up production capacity to meet surging market demand for broad-spectrum antibiotics without compromising on quality or safety standards.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding this specific Cefdinir synthesis technology. These insights are derived directly from the experimental data and comparative analysis presented in the patent documentation, providing clarity on the operational benefits and chemical rationale. Understanding these nuances helps stakeholders evaluate the feasibility of integrating this route into their existing supply chains.
Q: What are the advantages of using Pentafluorophenol over Phosphorus Pentachloride in Cefdinir synthesis?
A: Using Pentafluorophenol avoids the strict anhydrous conditions required by Phosphorus Pentachloride, significantly reducing environmental pollution and simplifying post-reaction treatment while maintaining high yields.
Q: How does this novel method improve the purity of the final Cefdinir product?
A: The method avoids the use of 2-mercaptobenzimidazole (Acibenzolar), which can introduce new impurities difficult to remove, thereby achieving purity levels exceeding 99.9%.
Q: Is this synthesis route suitable for large-scale commercial production?
A: Yes, the relaxed reaction conditions, high yields (over 90% in key steps), and simplified purification process make it highly scalable for industrial manufacturing of complex cephalosporins.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Cefdinir Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of adopting advanced synthetic methodologies to meet the evolving demands of the global pharmaceutical market. Our technical team has extensively analyzed the Pentafluorophenol activation route described in CN101817835A and possesses the expertise to implement this high-efficiency process at an industrial scale. We boast extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that our clients receive a consistent supply of material regardless of volume requirements. Our facilities are equipped with rigorous QC labs and adhere to stringent purity specifications, guaranteeing that every batch of Cefdinir meets the highest pharmacopeial standards for impurity profiles and stereochemical integrity.
We invite procurement leaders and R&D directors to collaborate with us to leverage this superior manufacturing technology for your antibiotic portfolios. By partnering with us, you gain access to a Customized Cost-Saving Analysis that quantifies the economic benefits of switching to this greener, more efficient synthesis route. We encourage you to contact our technical procurement team today to request specific COA data from our pilot batches and comprehensive route feasibility assessments tailored to your project timelines. Let us drive your supply chain efficiency forward with chemistry that works.
