Advanced Synthesis of Bis(2,4,6-trimethylbenzoyl)phenylphosphine Oxide for Commercial Scale Production
The global demand for high-efficiency photoinitiators continues to surge, driven by the expanding UV-curing markets in coatings, inks, and advanced composite materials. At the forefront of this sector is bis(2,4,6-trimethylbenzoyl)phenylphosphine oxide, widely known as Initiator 819, a critical component for white formulations and fiber-reinforced systems. A significant technological breakthrough in the manufacturing of this compound is detailed in patent CN112159429B, which introduces a novel preparation method utilizing specific ether compounds as complexing agents. This innovation addresses long-standing challenges in the industry, particularly regarding process safety and environmental compliance. By replacing traditional proton sources with polar aprotic ether complexing agents during the reaction of sodium sand and phenylphosphonic dichloride, the process effectively inhibits the formation of sodium phenylphosphonate polyphosphates. This strategic modification not only promotes the generation of the desired sodium phenylphosphonate intermediate but also eliminates the hazardous phenylphosphine hydrogen process, thereby drastically reducing process costs and enhancing operational safety for large-scale chemical production facilities.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industrial synthesis of acyl phosphine oxides has been plagued by significant safety hazards and environmental inefficiencies. Conventional protocols typically involve reacting metal sodium with phenyl phosphorus dichloride, followed by the addition of a proton source such as alcohols or amines to dissociate the intermediate into phenyl phosphine hydrogen. This phenyl phosphine hydrogen is then reacted with 2,4,6-trimethyl benzoyl chloride under alkaline conditions before final oxidation. The fundamental flaw in this legacy approach lies in the generation of phenyl phosphine, a substance known for its intense, unpleasant odor and high flammability, creating substantial potential safety hazards for plant personnel. Furthermore, the dissociating agent required in these traditional methods often exceeds 2 equivalents and cannot be recycled, leading to the generation of large volumes of wastewater containing high levels of organic matter. Additionally, prior art attempts to use activators like n-butanol have resulted in side reactions where the alcohol reacts with the acyl halide to form esters, consuming valuable raw materials and increasing purification costs, thus rendering the process economically suboptimal for modern polymer additive manufacturing.
The Novel Approach
The methodology disclosed in the referenced patent represents a paradigm shift by introducing specific ether compounds as complexing agents directly into the reaction between sodium sand and phenylphosphonic dichloride. Unlike protic solvents, these polar aprotic ethers, which include cyclic ethers like tetrahydrofuran or specific linear ethers, function by forming a stable complex structure with the generated sodium phenylphosphonate. This complexation effectively inhibits the clustering of intermediates that leads to polyphosphate formation, a major source of yield loss in older methods. By stabilizing the sodium phenylphosphonate, the process accelerates the subsequent reaction rate with mesitylene benzoyl chloride, ensuring high conversion efficiency without the need for the dangerous phenyl phosphine hydrogen step. The use of these complexing agents is highly efficient, requiring low dosages that significantly reduce the organic load in wastewater compared to traditional activating agents. This approach not only mitigates the risk of generating malodorous byproducts but also prevents the formation of mesitylene benzoic anhydride and esters, ensuring a cleaner reaction profile and superior product quality for reliable photoinitiator intermediate suppliers.
Mechanistic Insights into Ether-Complexed Sodium Phenylphosphonate Formation
The core mechanistic advantage of this synthesis lies in the interaction between the ether complexing agent and the sodium phenylphosphonate intermediate. Research indicates that the ether molecules solvate the sodium cations, creating a stable solvated sodium phenylphosphonate species that resists aggregation. In the absence of such complexing agents, the reactive phosphorus centers tend to combine, forming P-P bonds that result in sodium phenylphosphonate polyphosphates, which are essentially dead-end byproducts that lower the overall yield. The presence of the ether maintains the intermediate in a reactive, monomeric, or low-order oligomeric state, allowing it to rapidly attack the carbonyl carbon of the mesitylene benzoyl chloride. This rapid consumption of the intermediate drives the equilibrium forward, promoting continuous generation of sodium phenylphosphonate from the reaction of sodium sand and phenylphosphonic dichloride. However, precise control of the complexing agent concentration is vital; excessive amounts can overly solvate the intermediate, wrapping the negative ion center and sterically hindering the nucleophilic attack on the acyl chloride, while insufficient amounts fail to prevent polyphosphate clustering.
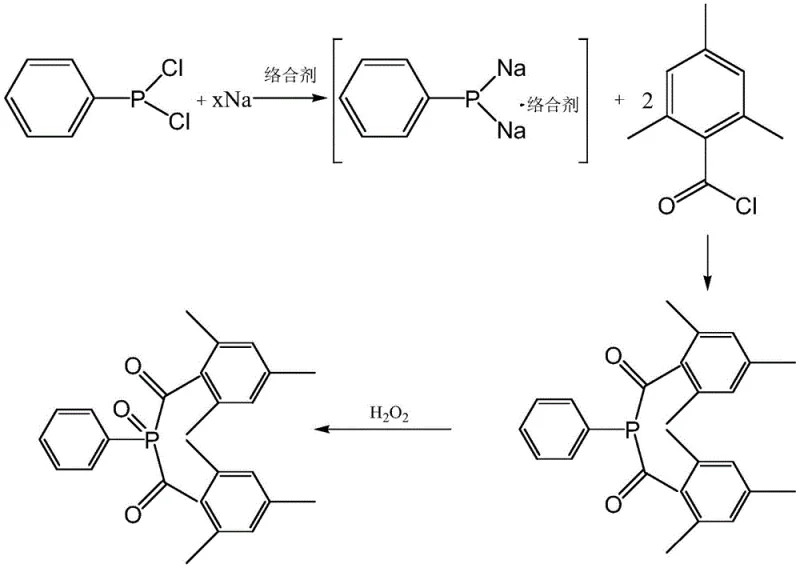
Impurity control is another critical aspect governed by this mechanistic pathway. In traditional processes, unreacted acyl chlorides or slow reaction kinetics can lead to the hydrolysis of acyl chlorides into acids or their condensation into anhydrides during workup. The accelerated reaction kinetics provided by the ether complexing agent ensure that the mesitylene benzoyl chloride is consumed efficiently, minimizing the residence time of this sensitive reagent in the reaction mixture. Consequently, the formation of side products like mesitylene benzoic anhydride is markedly reduced. This high selectivity simplifies the downstream purification process, as fewer acidic or neutral organic impurities need to be removed. The subsequent oxidation step using hydrogen peroxide proceeds smoothly on the purified acylphosphine intermediate, yielding the final bis(2,4,6-trimethylbenzoyl)phenylphosphine oxide with exceptional purity, often exceeding 99.0%, which is essential for applications requiring high-purity OLED material precursors or advanced coating additives.
How to Synthesize Bis(2,4,6-trimethylbenzoyl)phenylphosphine Oxide Efficiently
The synthesis protocol outlined in the patent provides a robust framework for producing Initiator 819 with high consistency and safety. The process begins with the careful preparation of a sodium sand suspension in a non-polar solvent such as toluene, ensuring a high surface area for the initial reaction with phenylphosphonic dichloride. The critical innovation involves the timed addition of the ether complexing agent, either during or after the formation of the sodium phenylphosphonate, to stabilize the intermediate before the introduction of the acyl chloride. Following the acylation, the mixture undergoes a controlled oxidation and a specialized workup involving alkaline washing to remove acidic byproducts, culminating in a recrystallization step that delivers the final product. For detailed operational parameters including temperature gradients, stirring rates, and specific molar ratios validated by experimental data, please refer to the standardized synthesis guide below.
- Prepare sodium sand suspension in a non-polar solvent like toluene under inert atmosphere at 90-110°C.
- React phenylphosphonic dichloride with sodium sand, adding a specific ether complexing agent to stabilize the intermediate.
- Add mesitylene benzoyl chloride for acylation, followed by oxidation with hydrogen peroxide and crystallization.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this novel synthesis route offers compelling economic and logistical benefits that extend beyond simple yield improvements. The elimination of the phenylphosphine hydrogen generation step fundamentally alters the safety profile of the manufacturing facility, reducing the need for specialized scrubbing systems and extensive safety protocols associated with handling toxic and flammable gases. This simplification of the safety infrastructure translates directly into lower capital expenditure and reduced operational overheads. Furthermore, the significant reduction in wastewater organic content lowers the cost and complexity of effluent treatment, aligning with increasingly stringent environmental regulations globally. By minimizing the formation of difficult-to-remove byproducts like polyphosphates and anhydrides, the process reduces the consumption of solvents and energy required for purification, offering substantial cost savings in the overall cost reduction in polymer additive manufacturing landscape.
- Cost Reduction in Manufacturing: The process achieves cost efficiency primarily through the optimization of raw material utilization and the simplification of the workflow. By avoiding the use of excess proton sources that cannot be recycled, the method reduces the mass of reagents required per kilogram of product. The high selectivity of the reaction minimizes the loss of expensive mesitylene benzoyl chloride to side reactions, ensuring that a greater proportion of input materials are converted into saleable product. Additionally, the reduced burden on wastewater treatment facilities and the lower energy demand for purification steps contribute to a leaner cost structure, allowing manufacturers to maintain competitive pricing even amidst fluctuating raw material markets.
- Enhanced Supply Chain Reliability: Safety is a cornerstone of supply chain continuity in the fine chemical sector. The removal of hazardous phenylphosphine gas from the process significantly decreases the risk of unplanned shutdowns due to safety incidents or regulatory inspections. The use of stable, commercially available ether complexing agents ensures a consistent supply of critical reagents, unlike specialized activators that may face sourcing bottlenecks. This stability allows for more predictable production scheduling and shorter lead times, enabling suppliers to respond more agilely to market demands for commercial scale-up of complex polymer additives without compromising on delivery commitments.
- Scalability and Environmental Compliance: The scalability of this method is supported by its reliance on standard unit operations such as suspension preparation, dropwise addition, and crystallization, which are easily transferable from pilot to production scale. The use of non-polar solvents like toluene, combined with low dosages of polar ethers, facilitates efficient solvent recovery and recycling, further enhancing the green chemistry profile of the process. The reduction in hazardous waste generation and the avoidance of malodorous emissions make this technology highly attractive for manufacturing sites located in regions with strict environmental zoning, ensuring long-term operational viability and reducing lead time for high-purity photoinitiator intermediates.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this advanced synthesis technology. These insights are derived directly from the experimental data and comparative analysis presented in the patent documentation, providing clarity on the operational advantages and chemical principles underlying the process. Understanding these details is crucial for technical teams evaluating the feasibility of adopting this method for their own production lines or for procurement specialists assessing the quality credentials of potential suppliers.
Q: Why is the ether complexing agent critical in this synthesis?
A: The ether complexing agent stabilizes the sodium phenylphosphide intermediate, preventing the formation of unwanted P-P bonded polyphosphates and eliminating the need for hazardous phenylphosphine gas generation.
Q: How does this method improve safety compared to traditional routes?
A: Traditional methods often generate malodorous and flammable phenylphosphine gas; this novel route bypasses that step entirely, significantly reducing explosion risks and odor issues.
Q: What represents the optimal molar ratio for the complexing agent?
A: Patent data suggests a molar ratio of phenylphosphonic dichloride to complexing agent between 1:0.2 and 1:1.0 provides the best balance of yield and reaction rate.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Bis(2,4,6-trimethylbenzoyl)phenylphosphine Oxide Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-performance photoinitiators play in the next generation of UV-curable materials. Our technical team has extensively analyzed the advancements described in patent CN112159429B and integrated similar process intensification strategies into our own manufacturing capabilities. We possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that our clients receive a consistent supply of materials that meet stringent purity specifications. Our rigorous QC labs employ state-of-the-art analytical techniques to verify the absence of critical impurities such as polyphosphates and anhydrides, guaranteeing that every batch of Initiator 819 performs optimally in your final formulation.
We invite you to collaborate with us to optimize your supply chain for photoinitiator intermediates. Our experts are ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements, demonstrating how our advanced synthesis routes can lower your total cost of ownership. Please contact our technical procurement team today to request specific COA data and route feasibility assessments, and let us support your journey towards more efficient and sustainable chemical manufacturing.
