Scalable Production of High-Purity 2-Isopropyl Thioxanthone via Novel Nitrile Route
Introduction to Advanced Photoinitiator Manufacturing
The global demand for high-performance UV curing materials continues to surge, driven by the need for rapid processing in coatings, inks, and adhesive applications. At the heart of these formulations lies the critical photoinitiator, 2-isopropyl thioxanthone, a Type II free radical initiator known for its exceptional efficiency in unsaturated polyester and acrylic systems. A pivotal advancement in the synthesis of this compound is detailed in patent CN103724320A, which introduces a novel preparation method that fundamentally shifts the paradigm from traditional benzoic acid precursors to a more efficient o-chlorobenzonitrile pathway. This technological breakthrough addresses long-standing industry pain points regarding yield, purity, and operational complexity, offering a robust solution for manufacturers seeking to optimize their supply chains. By leveraging a nucleophilic substitution strategy followed by a controlled acid-catalyzed cyclization, this method achieves a total reaction yield of approximately 90% with product purity exceeding 99%, setting a new benchmark for quality in the specialty chemicals sector.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industrial synthesis of 2-isopropyl thioxanthone has been plagued by significant thermodynamic and kinetic barriers inherent to older synthetic routes. Traditional methods, such as those disclosed in patent CN101570534, rely on o-chlorobenzoic acid and 4-isopropylthiophenol as starting materials, reacting under lithium hydroxide catalysis in tetralin solvents. A critical flaw in this approach is the poor solubility of the resulting intermediate, 2-(4-isopropylphenylthio)benzoic acid, which precipitates out of solution and necessitates harsh high-temperature acidification steps to redissolve for subsequent cyclization. Furthermore, these legacy processes often require reaction temperatures approaching 200°C and utilize expensive reagents like lithium hydroxide in large stoichiometric excesses, leading to substantial waste generation and elevated production costs. The reliance on such aggressive conditions not only compromises energy efficiency but also promotes the formation of isomeric impurities, such as 4-isopropyl thioxanthone, which are difficult to separate and detrimental to the photoinitiator's performance.
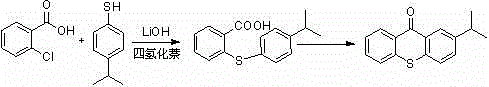
The Novel Approach
In stark contrast, the innovative methodology presented in CN103724320A circumvents these solubility and thermal challenges by employing o-chlorobenzonitrile as the electrophilic partner. This strategic substitution of the carboxylic acid group with a nitrile group dramatically alters the physicochemical properties of the intermediate, 2-(4-isopropylphenylthio)benzonitrile, rendering it highly soluble in common organic solvents like toluene and xylene at room temperature. This enhanced solubility eliminates the need for complex high-temperature separation and acidification procedures, allowing the reaction to proceed smoothly in a homogeneous phase. Consequently, the cyclization step can be conducted at significantly milder temperatures, typically between 60°C and 100°C, which drastically reduces energy consumption and minimizes thermal degradation. The result is a streamlined, two-step process that delivers superior yields and simplifies downstream purification, representing a quantum leap in process engineering for photoinitiator manufacturing.
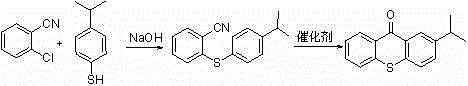
Mechanistic Insights into Nucleophilic Substitution and Cyclization
The core of this advanced synthesis lies in a meticulously orchestrated sequence of nucleophilic aromatic substitution followed by intramolecular Friedel-Crafts-type cyclization. In the first stage, 4-isopropylthiophenol is deprotonated by a mineral base, such as sodium hydroxide, to generate a highly reactive thiolate anion. This nucleophile attacks the electron-deficient aromatic ring of o-chlorobenzonitrile, displacing the chloride ion to form the thioether linkage. The presence of the electron-withdrawing nitrile group ortho to the leaving group activates the ring sufficiently to allow this substitution to occur at moderate temperatures (110-170°C) without the need for expensive transition metal catalysts. Following the formation of the thioether intermediate, the system undergoes a profound transformation upon the addition of concentrated sulfuric acid. The acid acts as both a dehydrating agent and a catalyst, protonating the nitrile nitrogen to increase its electrophilicity, which facilitates an intramolecular attack by the adjacent aromatic ring. This cyclization closes the central thioxanthone ring system, establishing the rigid, planar conjugated structure essential for the molecule's photochemical activity.
From an impurity control perspective, the choice of the nitrile precursor plays a pivotal role in maintaining high product fidelity. Unlike the carboxylic acid route, where the intermediate's tendency to aggregate and precipitate can lead to localized hot spots and incomplete reactions, the soluble nitrile intermediate ensures uniform reaction kinetics throughout the vessel. This homogeneity prevents the formation of oligomeric by-products and limits the isomerization that typically leads to the unwanted 4-isopropyl isomer. Furthermore, the mild cyclization conditions (50-120°C) prevent the sulfonation of the aromatic rings, a common side reaction in harsher sulfuric acid environments. The final purification via solvent recrystallization, using alcohols or hydrocarbons, effectively removes any trace unreacted starting materials or minor sulfonated species, guaranteeing a final product with a purity profile that meets the stringent requirements of high-end optical applications.
How to Synthesize 2-Isopropyl Thioxanthone Efficiently
Implementing this novel synthetic route requires precise control over reaction parameters to maximize the benefits of the nitrile chemistry. The process begins with the activation of the thiol component in a water-immiscible solvent, followed by the coupling with the nitrile and the final acid-mediated ring closure. Operators must pay close attention to the dehydration step to ensure complete conversion to the thiolate salt, as residual water can hydrolyze the nitrile or dilute the sulfuric acid catalyst. The subsequent addition of sulfuric acid must be performed under strict temperature control, ideally using an ice-water bath initially to manage the exotherm, before ramping up to the optimal cyclization temperature. For a comprehensive, step-by-step technical guide including specific molar ratios, stirring speeds, and safety protocols, please refer to the standardized operating procedure below.
- React 4-isopropylthiophenol with mineral alkali in a solvent like toluene under reflux dehydration to form the thiolate salt.
- Add o-chlorobenzonitrile to the mixture and heat to 110-170°C to obtain the 2-(4-isopropylphenylthio)benzonitrile intermediate.
- Cyclize the intermediate by slowly adding concentrated sulfuric acid at 50-120°C, followed by workup and recrystallization.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this nitrile-based synthesis route translates into tangible strategic advantages that extend far beyond simple chemical yield. By shifting away from the legacy benzoic acid pathway, manufacturers can unlock significant cost reductions and operational efficiencies that directly impact the bottom line. The elimination of expensive reagents like lithium hydroxide and the reduction in energy-intensive high-temperature steps create a leaner, more cost-effective production model. Moreover, the improved solubility of intermediates simplifies the physical handling of materials, reducing the risk of equipment fouling and minimizing downtime for cleaning and maintenance. These factors combine to create a more resilient supply chain capable of meeting fluctuating market demands with greater agility and reliability.
- Cost Reduction in Manufacturing: The economic benefits of this process are driven primarily by the substitution of costly raw materials and the optimization of energy usage. By replacing lithium hydroxide with inexpensive sodium hydroxide and avoiding the need for high-boiling solvents like tetralin, the direct material costs are substantially lowered. Additionally, the ability to conduct the cyclization at temperatures below 120°C, compared to the nearly 200°C required by older methods, results in drastic savings on steam and cooling utilities. The high yield of the reaction further amplifies these savings by maximizing the output per batch, effectively spreading fixed overhead costs over a larger volume of saleable product.
- Enhanced Supply Chain Reliability: Supply continuity is bolstered by the use of commodity-grade raw materials that are widely available in the global chemical market. O-chlorobenzonitrile and 4-isopropylthiophenol are established industrial chemicals with robust supply networks, reducing the risk of shortages that can plague specialized reagents. The simplified process flow, which avoids complex filtration and high-pressure steps, also reduces the likelihood of mechanical failures or process upsets that could interrupt production schedules. This reliability ensures that downstream customers in the coatings and ink industries receive their shipments on time, every time, fostering stronger long-term partnerships.
- Scalability and Environmental Compliance: From an environmental and scalability standpoint, this method offers a cleaner, greener alternative to traditional synthesis. The reduction in spent acid generation and the avoidance of volatile, corrosive reagents like thionyl chloride minimize the burden on waste treatment facilities and lower disposal costs. The process is inherently safer to scale up due to the moderate operating pressures and temperatures, allowing for seamless transition from pilot plant to multi-ton commercial production. This alignment with modern environmental, health, and safety (EHS) standards not only mitigates regulatory risk but also enhances the brand reputation of the manufacturer as a sustainable supplier.
Frequently Asked Questions (FAQ)
Understanding the technical nuances of this synthesis is crucial for stakeholders evaluating its potential for integration into their supply chains. The following questions address common inquiries regarding the process mechanics, quality control, and scalability, drawing directly from the empirical data and experimental examples provided in the patent literature. These insights are designed to clarify the operational realities of the nitrile route and demonstrate its viability for industrial application.
Q: Why is the nitrile-based route superior to the benzoic acid route for 2-isopropyl thioxanthone?
A: The nitrile-based route utilizes o-chlorobenzonitrile, which generates an intermediate with significantly better solubility compared to the poorly soluble benzoic acid derivatives. This eliminates the need for high-temperature acidification and complex filtration steps, resulting in higher yields (>90%) and simpler operations.
Q: What are the critical reaction conditions for the cyclization step?
A: The cyclization requires concentrated sulfuric acid (93%-98%) added under ice-water bath conditions initially, followed by heating to a moderate temperature range of 60-100°C. This controlled thermal profile minimizes side reactions and ensures high purity.
Q: Is this process suitable for large-scale industrial production?
A: Yes, the process uses cheap and readily available raw materials, operates at relatively low temperatures, and avoids the generation of large amounts of spent acid associated with traditional methods, making it highly suitable for commercial scale-up.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 2-Isopropyl Thioxanthone Supplier
As the global market for UV-curable materials expands, securing a dependable source of high-quality photoinitiators is paramount for maintaining competitive advantage. NINGBO INNO PHARMCHEM stands at the forefront of this industry, leveraging deep technical expertise to deliver 2-isopropyl thioxanthone that meets the most rigorous performance standards. Our commitment to excellence is underpinned by extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that we can support your needs whether you are in the R&D phase or full-scale manufacturing. We operate stringent purity specifications and utilize rigorous QC labs to guarantee that every batch delivers consistent reactivity and color stability, essential for premium coating and ink formulations.
We invite you to explore how our advanced manufacturing capabilities can drive value for your organization. Our team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements, demonstrating exactly how our optimized processes can reduce your total cost of ownership. We encourage you to contact our technical procurement team today to request specific COA data and route feasibility assessments, and let us partner with you to accelerate your product development timelines.
