Advanced Synthesis of 2-Isopropyl Thioxanthone for High-Efficiency UV Curing Applications
Advanced Synthesis of 2-Isopropyl Thioxanthone for High-Efficiency UV Curing Applications
The global demand for high-performance photoinitiators in the UV curing industry has necessitated a rigorous re-evaluation of synthetic pathways to ensure both economic viability and environmental compliance. Patent CN101817812A introduces a transformative preparation method for 2-isopropyl thioxanthone and its derivatives, addressing critical bottlenecks in traditional manufacturing. This technology leverages a sophisticated condensation reaction between o-chlorobenzoic acid derivatives and 4-isopropyl thiophenol derivatives, followed by a dehydration cyclization catalyzed by concentrated sulfuric acid. Unlike legacy processes that rely on prohibitively expensive reagents or generate substantial hazardous waste, this novel approach utilizes ubiquitous mineral alkalis and common hydrocarbon solvents to achieve product purity levels exceeding 99 percent. For R&D directors and procurement specialists alike, this represents a significant opportunity to optimize the supply chain for reliable photo initiator suppliers while ensuring the consistent quality required for advanced applications in optical fiber manufacturing and automotive coatings.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of thioxanthone derivatives has been plagued by inefficient methodologies that impose severe economic and operational burdens on manufacturers. For instance, earlier patents such as US4902826 described processes utilizing tetralin as a solvent and lithium hydroxide as a base; however, tetralin is not only costly but also presents significant environmental contamination risks, while lithium hydroxide drives up raw material expenses unnecessarily. Other approaches, like those detailed in US4094900, relied on sodium iodide and excessive amounts of thiophenol, creating a stoichiometric imbalance that wastes valuable starting materials and complicates downstream purification. Furthermore, methods involving Friedel-Crafts acylation, as seen in US4661595, often resulted in abysmal yields of approximately 18 percent and utilized N,N-dimethylformamide (DMF), a solvent notorious for its difficulty in recovery and high toxicity profile. These conventional routes frequently generate large volumes of spent acid and produce inseparable byproducts, such as 4-isopropyl thioxanthone, which severely degrade the photo-initiating efficiency of the final product and compromise the integrity of high-purity thioxanthone derivatives required for sensitive electronic applications.
The Novel Approach
In stark contrast, the methodology outlined in CN101817812A revolutionizes the production landscape by implementing a streamlined condensation-cyclization sequence that prioritizes atom economy and operational simplicity. This innovative route replaces expensive lithium salts and iodides with cost-effective sodium hydroxide, fundamentally altering the cost structure of cost reduction in UV curing materials manufacturing. By employing common hydrocarbon solvents such as toluene, xylene, or even industrial diesel and kerosene, the process eliminates the need for specialized, high-boiling polar solvents that are difficult to recycle. The reaction conditions are meticulously optimized to operate under autogenous pressure (1MPa) at temperatures between 150°C and 250°C, which facilitates efficient dehydration and drives the equilibrium toward the desired condensation product without the formation of stubborn impurities. This strategic shift not only simplifies the post-reaction workup—requiring only pH adjustment and phase separation—but also ensures that the final crystallization yields a faint yellow solid with exceptional purity, thereby meeting the stringent specifications demanded by modern commercial scale-up of complex photoinitiators.
Mechanistic Insights into Condensation and Acid-Catalyzed Cyclization
The core of this synthetic breakthrough lies in the precise orchestration of a nucleophilic aromatic substitution followed by an intramolecular electrophilic cyclization. Initially, the reaction between the o-chlorobenzoic acid derivative and the 4-isopropyl thiophenol derivative proceeds via a base-mediated condensation mechanism. In the presence of mineral alkali and water at moderate temperatures (70-100°C), the thiolate anion is generated in situ, which acts as a potent nucleophile attacking the electron-deficient aromatic ring of the chlorobenzoic acid. This step is critical for forming the thioether linkage, and the use of aqueous alkali ensures that the reaction medium remains homogeneous enough to facilitate rapid kinetics while avoiding the side reactions often associated with anhydrous strong bases. Following this initial coupling, the introduction of a hydrocarbon solvent allows for azeotropic reflux dehydration, effectively removing water from the system to prevent the hydrolysis of the newly formed bond and pushing the reaction equilibrium forward according to Le Chatelier's principle.
Subsequently, the transformation into the tricyclic thioxanthone core is achieved through a rigorous acid-catalyzed cyclization. Upon the addition of 98 percent concentrated sulfuric acid to the organic phase, the carboxylic acid group is activated, likely forming an acylium ion or a mixed anhydride species that serves as a powerful electrophile. This electrophile then attacks the ortho-position of the adjacent phenyl ring (relative to the sulfur atom), closing the central ring to form the thioxanthone skeleton. The patent specifies heating this mixture to 100°C for 6 to 10 hours, a duration sufficient to ensure complete conversion while minimizing the risk of sulfonation or charring of the organic substrate. This mechanistic pathway is superior because it avoids the harsh Lewis acids typically required in Friedel-Crafts reactions, thereby reducing equipment corrosion and simplifying waste treatment. The result is a highly selective formation of the 2-isopropyl thioxanthone structure, as illustrated in the reaction scheme below, which minimizes the generation of regioisomers that plague other synthetic routes.
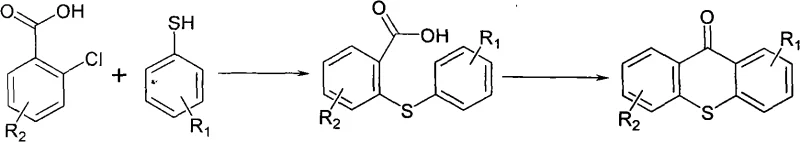
How to Synthesize 2-Isopropyl Thioxanthone Efficiently
Implementing this synthesis protocol requires careful attention to thermal parameters and pressure management to maximize yield and safety. The process begins with the mixing of stoichiometric amounts of o-chlorobenzoic acid, 4-isopropyl thiophenol, and sodium hydroxide in water, followed by a controlled heating phase to initiate the condensation. Once the initial coupling is complete, the system transitions to a high-temperature dehydration phase under nitrogen protection, where the choice of solvent plays a pivotal role in determining the ease of subsequent isolation. The final cyclization step demands precise acid handling and temperature control to ensure the structural integrity of the thioxanthone ring system. For detailed operational parameters, including specific molar ratios and cooling protocols, please refer to the standardized synthesis guide provided below.
- Perform condensation reaction between o-chlorobenzoic acid derivative and 4-isopropyl thiophenol derivative using mineral alkali (NaOH) in water at 70-100°C.
- Add organic hydrocarbon solvent, reflux to dehydrate, then heat under nitrogen protection at 1MPa pressure and 150-250°C for 8-12 hours.
- Adjust pH to 2-3 with hydrochloric acid, separate layers, then add concentrated sulfuric acid to the organic phase for cyclization at 100°C.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this patented methodology offers profound strategic advantages that extend far beyond simple chemical yield improvements. By fundamentally restructuring the input materials, the process decouples production costs from the volatility of rare or specialized reagent markets. The elimination of lithium hydroxide and sodium iodide removes exposure to the price fluctuations of these niche chemicals, replacing them with sodium hydroxide, a commodity chemical with stable global pricing and abundant availability. Furthermore, the ability to utilize broad-cut hydrocarbon solvents like diesel or solvent oil, rather than refined aromatics like tetralin or polar solvents like DMF, significantly lowers the overhead associated with solvent procurement and recycling infrastructure. This flexibility allows manufacturing facilities to source materials locally in various regions, thereby reducing lead time for high-purity photoinitiators and mitigating risks associated with international logistics disruptions.
- Cost Reduction in Manufacturing: The economic impact of switching from precious metal catalysts or expensive organic bases to mineral alkalis cannot be overstated. By utilizing sodium hydroxide instead of lithium hydroxide, the direct material cost per kilogram of product is drastically simplified and reduced. Additionally, the avoidance of sodium iodide eliminates a major cost center, as iodide salts are significantly more expensive than chloride or hydroxide counterparts. The process also negates the need for complex solvent recovery systems required for DMF, leading to substantial capital expenditure savings on plant infrastructure and ongoing energy costs for distillation. These cumulative efficiencies translate into a much more competitive cost basis for the final photo initiator, allowing end-users to benefit from lower pricing without compromising on performance specifications.
- Enhanced Supply Chain Reliability: Supply chain resilience is bolstered by the reliance on universally available feedstocks. O-chlorobenzoic acid and thiophenol derivatives are produced at massive scales globally for various industries, ensuring that supply shortages are highly unlikely compared to specialized reagents like 4-isopropyl benzene methyl ether. The robustness of the reaction conditions, which tolerate a range of hydrocarbon solvents, means that production can continue even if specific solvent grades are temporarily unavailable, as the process can adapt to use alternatives like kerosene or xylene. This adaptability ensures continuous operation and reliable delivery schedules, which is critical for downstream customers in the coating and ink industries who operate on just-in-time manufacturing models and cannot afford production stoppages due to raw material scarcity.
- Scalability and Environmental Compliance: From an environmental and scaling perspective, this route is exceptionally well-suited for large-volume production. The absence of heavy metal catalysts or toxic polar solvents simplifies the waste stream, making effluent treatment more straightforward and less costly. The high purity of the crude product (>99 percent) reduces the need for energy-intensive recrystallization cycles or chromatographic purification, which are often bottlenecks in scaling up fine chemical processes. Moreover, the use of closed-system pressurized reactors (1MPa) is standard in modern chemical plants, meaning the technology can be transferred to existing multipurpose reactors without requiring bespoke equipment fabrication. This ease of scale-up ensures that supply can be rapidly expanded to meet surging market demand for UV curing materials in emerging sectors like 3D printing and optical telecommunications.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis technology. These insights are derived directly from the experimental data and comparative analysis presented in the patent documentation, aiming to clarify the operational benefits and chemical rationale behind the process. Understanding these details is crucial for technical teams evaluating the feasibility of integrating this route into their existing manufacturing portfolios.
Q: How does this new synthesis method improve product purity compared to traditional Friedel-Crafts acylation?
A: Traditional Friedel-Crafts methods often suffer from low yields (around 18%) and difficult separation of byproducts like 4-isopropyl thioxanthone. The patented condensation-cyclization route utilizes a specific high-temperature pressurized step that drives the reaction to completion, consistently achieving product purity exceeding 99% without complex chromatographic separation.
Q: What are the primary cost drivers eliminated in this manufacturing process?
A: The process eliminates the need for expensive reagents such as lithium hydroxide, sodium iodide, and tetralin solvents found in prior art. By substituting these with cost-effective mineral alkalis (sodium hydroxide) and common hydrocarbon solvents (like diesel or toluene), the overall raw material expenditure is drastically reduced while maintaining high reaction efficiency.
Q: Is this synthesis route suitable for large-scale commercial production?
A: Yes, the method is explicitly designed for scalability. It employs robust reaction conditions (1MPa pressure, 200-210°C) and readily available industrial solvents, avoiding the environmental hazards and recovery difficulties associated with polar aprotic solvents like DMF, making it ideal for multi-ton annual production.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 2-Isopropyl Thioxanthone Supplier
At NINGBO INNO PHARMCHEM, we recognize that the transition to more efficient synthetic routes is vital for maintaining competitiveness in the global fine chemicals market. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the theoretical benefits of patent CN101817812A are fully realized in practical, industrial settings. We are committed to delivering products with stringent purity specifications, supported by our rigorous QC labs that verify every batch against the highest industry standards for photo initiators. Our capability to manage complex condensation and cyclization reactions under pressure allows us to offer a supply of 2-isopropyl thioxanthone that is both consistent in quality and reliable in volume, catering to the exacting needs of the UV curing and electronic materials sectors.
We invite you to collaborate with us to explore how this advanced manufacturing technology can optimize your supply chain. Contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We are prepared to provide specific COA data and comprehensive route feasibility assessments to demonstrate how our optimized synthesis of 2-isopropyl thioxanthone can drive value and efficiency for your organization.
