Advanced Manufacturing of Ceforanide: A Novel Route for High-Purity Antibiotic Intermediates
The pharmaceutical landscape for second-generation cephalosporins has long been dominated by established players, particularly regarding the production of Ceforanide, a broad-spectrum antibiotic known for its efficacy against Gram-negative and Gram-positive bacterial infections. The patent CN101941982A introduces a transformative preparation method that fundamentally alters the synthetic pathway for this critical active pharmaceutical ingredient (API). By shifting away from the hazardous and costly conditions typical of legacy processes, this innovation offers a robust framework for reliable API intermediate supplier networks to enhance their production capabilities. The core breakthrough lies in the strategic modification of reaction parameters, specifically the elevation of condensation temperatures from extreme cryogenic levels to a more manageable range, coupled with a novel purification strategy using N,N-dimethylbenzylamine (DMBA). This technical evolution not only addresses the historical monopoly on Ceforanide production but also aligns with modern green chemistry principles by significantly reducing the environmental footprint associated with solvent usage and waste generation.
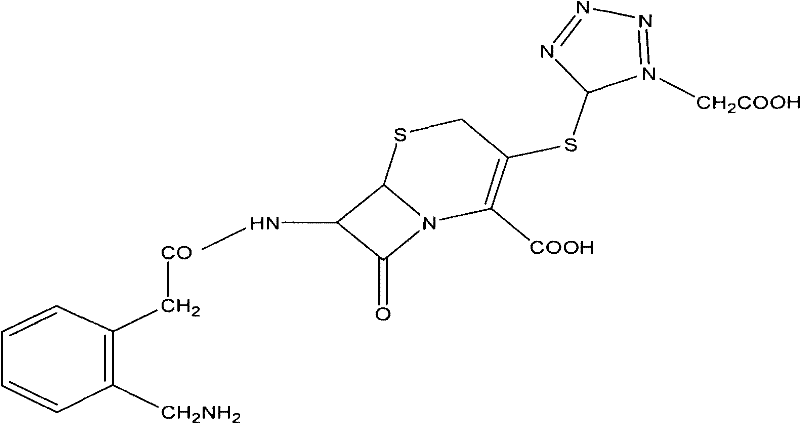
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industrial synthesis of Ceforanide has been plagued by severe operational constraints that hinder scalability and increase production costs. Prior art methods, such as those disclosed in WO2008/010043, necessitate extremely low reaction temperatures ranging from -45°C to -55°C during the critical condensation phase. Maintaining such cryogenic conditions on a commercial scale requires specialized refrigeration equipment and consumes substantial energy, creating a significant barrier to entry for many manufacturers. Furthermore, these conventional routes often rely on complex mixed solvent systems, such as dichloromethane-DMF blends, which complicate downstream processing and solvent recovery. The purification steps in traditional methods are equally problematic, frequently yielding crude products with lower purity profiles that require multiple, inefficient recrystallization cycles. Additionally, the reliance on specific organic bases and acidic workups in older protocols often leads to the generation of excessive saline waste, posing challenges for cost reduction in pharmaceutical intermediates manufacturing and environmental compliance.
The Novel Approach
The methodology outlined in the present patent data represents a paradigm shift by optimizing the thermodynamic and kinetic parameters of the synthesis. A primary advantage is the adjustment of the condensation temperature to a much milder range of -20°C to -25°C, which drastically reduces energy consumption and simplifies reactor engineering requirements. This approach utilizes single solvent systems, such as pure dichloromethane for silylation and DMF for the active ester formation, streamlining the process flow and facilitating easier solvent recycling. Crucially, the introduction of a unique purification step involving the formation of a Ceforanide-N,N-dimethylbenzylamine salt allows for a dramatic improvement in product purity, elevating it from approximately 80% in the crude state to over 95% in the salt form. This selective crystallization technique effectively removes impurities without the need for extensive chromatographic separation, thereby enhancing the overall throughput and economic viability of producing high-purity API intermediates.
Mechanistic Insights into Silylation and Active Ester Condensation
The chemical elegance of this synthesis lies in the precise activation of the beta-lactam nucleus and the subsequent coupling with the side chain. The process begins with the silylation of the key intermediate, 3-MTAA-7-ACA, using silylating agents like BSA (N,O-bis(trimethylsilyl)acetamide) or HMDS (hexamethyldisilazane) in refluxing dichloromethane. This step protects the sensitive functional groups on the cephalosporin ring, rendering the molecule more nucleophilic and stable for the upcoming coupling reaction. Simultaneously, the side chain precursor, derived from 2-aminomethyl-phenylacetic acid (2-AMPA), undergoes a protection sequence where the primary amine is masked with an unsaturated ethylene linkage via reaction with ethyl acetoacetate. This protected amine is then converted into an alkali metal salt and subsequently activated using pivaloyl chloride to form a highly reactive mixed anhydride or active ester species. The convergence of these two streams—the silylated nucleus and the activated side chain—occurs under controlled basic conditions, ensuring high regioselectivity and minimizing the formation of unwanted isomers.
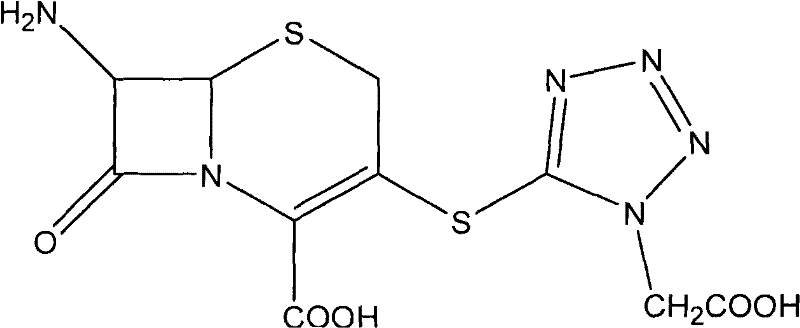
Impurity control is meticulously managed through the manipulation of pH and solvent polarity during the workup phases. Unlike traditional methods that might precipitate the product directly from acidic solutions, leading to co-precipitation of byproducts, this novel route employs a strategic pH swing. The reaction mixture is quenched into an ice-water system, and the product is initially isolated at a specific isoelectric point. The subsequent formation of the DMBA salt acts as a powerful purification filter; the bulky organic base forms a crystalline lattice with the Ceforanide molecule that excludes structurally similar impurities. Upon regeneration of the free acid from this salt using mineral acids in a sterile environment, the resulting Ceforanide exhibits superior crystal habit and purity. This mechanism ensures that the final commercial scale-up of complex antibiotics meets stringent pharmacopeial standards without the need for resource-intensive polishing steps.
How to Synthesize Ceforanide Efficiently
The synthesis of Ceforanide via this novel route involves a sequence of highly controlled chemical transformations designed to maximize yield and safety. The process initiates with the preparation of the 3-MTAA-7-ACA intermediate, followed by the distinct protection and activation of the phenylacetic acid side chain. These two components are then coupled under mild cryogenic conditions, followed by a unique salt-formation purification strategy. The detailed operational parameters, including specific reagent ratios, temperature ramps, and crystallization times, are critical for reproducing the high purity and yield reported in the patent data. For process chemists looking to implement this technology, adhering to the specific solvent choices and pH adjustments described is essential for success.
- Preparation of 3-MTAA-7-ACA intermediate by reacting 7-ACA with MTAA and boron trifluoride in organic solvent.
- Protection of 2-AMPA amine group using ethyl acetoacetate to form an alkali metal salt derivative.
- Condensation of the silylated 3-MTAA-7-ACA with the activated ester derivative at controlled low temperatures (-20°C to -25°C).
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this novel Ceforanide synthesis route offers tangible strategic benefits that extend beyond simple technical metrics. The shift to milder reaction conditions directly translates to reduced operational expenditures (OPEX) by lowering the energy load required for cooling systems. Furthermore, the simplified solvent profile reduces the complexity of waste treatment and solvent recovery units, contributing to a leaner manufacturing footprint. The robustness of the purification method ensures consistent supply quality, mitigating the risk of batch failures that can disrupt downstream formulation schedules. By securing a supply chain based on this improved technology, organizations can achieve reducing lead time for high-purity antibiotics and ensure a more resilient supply of this critical therapeutic agent.
- Cost Reduction in Manufacturing: The elimination of extreme cryogenic requirements (-55°C) in favor of moderate low temperatures (-25°C) significantly lowers energy consumption and equipment maintenance costs. Additionally, the use of single solvent systems reduces the volume of organic waste generated, leading to substantial cost savings in waste disposal and solvent procurement. The high efficiency of the DMBA salt purification step minimizes product loss during refining, thereby improving the overall mass balance and yield of the final API.
- Enhanced Supply Chain Reliability: The reagents utilized in this process, such as boron trifluoride complexes and common silylating agents, are readily available from global chemical suppliers, reducing the risk of raw material shortages. The simplified process flow decreases the likelihood of technical bottlenecks, ensuring a steady and predictable production output. This reliability is crucial for maintaining continuous inventory levels for essential antibiotic medications in the global market.
- Scalability and Environmental Compliance: The mild reaction conditions and reduced hazard profile make this process highly suitable for scaling from pilot plant to multi-ton commercial production. The reduction in three wastes (wastewater,废气, and solid waste) aligns with increasingly strict environmental regulations, facilitating smoother permitting and operation in regulated jurisdictions. This environmental stewardship enhances the corporate sustainability profile of the manufacturing entity.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this novel Ceforanide preparation method. These insights are derived directly from the comparative data and experimental results presented in the patent literature, providing a clear understanding of the process advantages. Understanding these details is vital for stakeholders evaluating the feasibility of adopting this technology for their own manufacturing portfolios.
Q: What are the key advantages of this novel Ceforanide preparation method over prior art?
A: The novel method utilizes milder reaction temperatures (-20°C to -25°C compared to -45°C to -55°C in prior art), employs single solvent systems instead of mixed solvents, and introduces a unique DMBA salt purification step that significantly enhances product purity from roughly 80% to over 95%.
Q: How does the new process improve environmental compliance and safety?
A: By avoiding extreme cryogenic conditions and utilizing a mixing acid-alkaline process for crystallization, the method reduces the generation of three wastes (waste water, gas, and solids) and minimizes the risk of explosion associated with harsh industrial operations, making it safer for large-scale manufacturing.
Q: What is the role of the unsaturated ethylene linkage in the side chain synthesis?
A: The unsaturated ethylene linkage serves as a temporary protecting group for the primary amine in 2-aminomethyl-phenylacetic acid (2-AMPA), allowing for the formation of a stable alkali metal salt that can be effectively converted into an active ester for the final condensation reaction.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Ceforanide Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of accessing advanced synthetic routes for essential antibiotics like Ceforanide. As a premier CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the theoretical benefits of this patent are fully realized in practice. Our state-of-the-art facilities are equipped to handle the specific solvent and temperature requirements of this novel process, while our rigorous QC labs enforce stringent purity specifications to guarantee product quality. We are committed to bridging the gap between innovative patent chemistry and reliable commercial supply.
We invite potential partners to engage with our technical team to explore how this improved synthesis route can optimize your supply chain. By requesting a Customized Cost-Saving Analysis, you can gain a deeper understanding of the economic impact of switching to this method. We encourage you to contact our technical procurement team to obtain specific COA data and route feasibility assessments tailored to your project needs, ensuring a seamless transition to this superior manufacturing standard.
