Advanced Manufacturing of Bromfenac Sodium: A Cost-Effective Non-Friedel-Crafts Route for Global Pharma Supply Chains
Advanced Manufacturing of Bromfenac Sodium: A Cost-Effective Non-Friedel-Crafts Route for Global Pharma Supply Chains
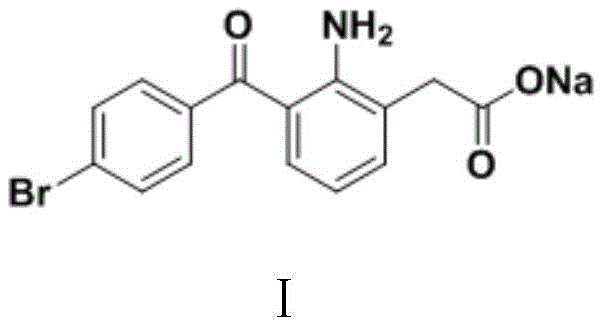
The pharmaceutical industry continuously seeks robust, scalable, and environmentally sustainable pathways for the production of critical non-steroidal anti-inflammatory drugs (NSAIDs). Patent CN110885296B introduces a groundbreaking preparation method for Bromfenac Sodium (I), a potent cyclooxygenase inhibitor widely used in ophthalmic formulations for treating post-operative inflammation and pain. This technology represents a significant paradigm shift from traditional Friedel-Crafts acylation strategies, offering a streamlined synthetic route that begins with readily available p-bromobenzoate derivatives. By leveraging a novel base-catalyzed condensation mechanism followed by a unique halogenation-amination-elimination sequence, this method achieves exceptional purity levels exceeding 99.9% while drastically minimizing the generation of hazardous three-waste streams. For R&D directors and procurement specialists, this patent data signals a viable opportunity to secure a more reliable pharmaceutical intermediates supplier capable of delivering high-quality active ingredients with reduced supply chain volatility.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industrial synthesis of Bromfenac Sodium has been plagued by significant technical and economic bottlenecks inherent to Friedel-Crafts chemistry. Traditional routes, such as those described in earlier US patents and academic literature, typically rely on the acylation of 2-amino-4'-bromobenzophenone or indoline derivatives using stoichiometric amounts of aggressive Lewis acids like aluminum trichloride or boron trichloride. These processes are not only operationally hazardous due to the exothermic nature of the reactions but also necessitate cryogenic conditions, often requiring temperatures as low as -70°C to control regioselectivity, which imposes a massive energy burden on manufacturing facilities. Furthermore, the quenching of these Lewis acid complexes generates voluminous quantities of acidic wastewater containing heavy metal salts, creating severe environmental compliance challenges and inflating waste disposal costs. Additionally, the reliance on specialized starting materials like indoline or 2-methylthioethyl acetate introduces supply chain fragility, as these precursors are often subject to price volatility and limited availability from fine chemical vendors.
The Novel Approach
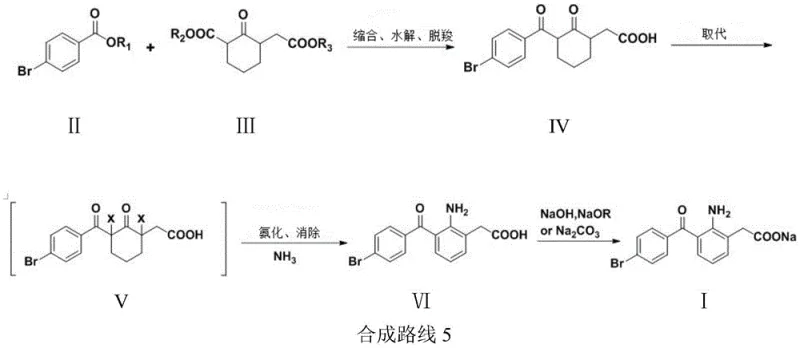
In stark contrast to the legacy methodologies, the innovative pathway disclosed in CN110885296B circumvents the need for harsh Lewis acid catalysts entirely by employing a constructive carbon-carbon bond-forming strategy. This novel approach initiates with the condensation of p-bromobenzoate (II) and a functionalized cyclohexanone derivative (III) under mild basic conditions, followed by hydrolysis and decarboxylation to establish the core ketone framework. The subsequent transformation involves a highly selective dihalogenation at the alpha-positions of the cyclohexanone ring, followed by ammoniation and base-mediated elimination to aromatize the ring system, effectively constructing the aniline moiety in situ. This sequence not only operates at moderate temperatures ranging from 30°C to 90°C, eliminating the need for energy-intensive cryogenic cooling, but also utilizes commodity chemicals that are abundant in the global chemical market. The result is a process that is inherently safer, greener, and economically superior, positioning it as an ideal candidate for cost reduction in pharmaceutical intermediates manufacturing.
Mechanistic Insights into Base-Catalyzed Condensation and Aromatization
The core of this technological breakthrough lies in the precise control of the initial condensation reaction between the ester and the cyclic keto-ester. Under the influence of strong bases such as sodium methoxide or potassium tert-butoxide, the enolate formed from the cyclohexanone derivative attacks the carbonyl carbon of the p-bromobenzoate, establishing the critical C-C bond that links the two aromatic precursors. This step is meticulously optimized to prevent self-condensation of the starting materials, a common side reaction that plagues similar Claisen-type condensations. Following the condensation, the intermediate undergoes hydrolysis and decarboxylation, a tandem process that removes the ester protecting groups and extrudes carbon dioxide to yield the stable keto-acid intermediate (IV). The specificity of this reaction sequence ensures that the carbonyl functionality remains intact and correctly positioned for the subsequent halogenation step, thereby maintaining high atom economy and minimizing the formation of structural isomers that are difficult to separate.
The final construction of the aniline ring is achieved through a sophisticated halogenation-amination-elimination cascade that demonstrates remarkable chemoselectivity. The keto-acid intermediate is first subjected to dihalogenation using reagents like bromine or N-bromosuccinimide, which selectively installs halogen atoms at the 1 and 3 positions relative to the ketone. This dihalogenated species (V) is then treated with ammonia, which displaces one of the halogen atoms to form an amino group. Finally, the addition of a base triggers an elimination reaction, removing the second halogen atom and a proton to restore aromaticity, thus converting the cyclohexanone ring into the substituted aniline ring found in Bromfenac (VI). This mechanistic pathway avoids the harsh acidic hydrolysis conditions required in prior art to open indole rings, thereby preserving the integrity of the sensitive bromobenzoyl group and ensuring that the final product exhibits the stringent purity specifications required for ophthalmic applications.
How to Synthesize Bromfenac Sodium Efficiently
The execution of this synthesis requires careful attention to reaction stoichiometry and temperature control to maximize yield and minimize impurity profiles. The process is divided into three distinct operational stages: the formation of the keto-acid backbone, the ring aromatization via halogenation and amination, and the final salt formation. Each stage has been optimized in the patent examples to demonstrate reproducibility on a laboratory scale, providing a clear blueprint for process engineers looking to transfer this technology to pilot or commercial plants. The detailed standardized synthesis steps, including specific solvent ratios, addition rates, and workup procedures, are outlined below to facilitate immediate technical evaluation.
- Condense p-bromobenzoate with 2-alkoxycarbonyl-6-alkoxycarbonylmethylcyclohexanone under basic conditions, followed by hydrolysis and decarboxylation to form the keto-acid intermediate.
- Subject the keto-acid intermediate to dihalogenation, followed by ammoniation and base-catalyzed elimination to construct the aniline ring system.
- Perform a final salification reaction using sodium hydroxide or sodium alkoxide in an alcoholic solvent to yield the final Bromfenac Sodium API.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this novel synthetic route offers compelling strategic advantages that extend beyond simple unit cost metrics. By shifting away from the dependency on scarce and expensive heterocyclic starting materials like indoline or indole, manufacturers can leverage a supply chain built upon bulk commodity esters and cyclohexanones, which are produced globally at massive scales with consistent quality. This diversification of raw material sources significantly mitigates the risk of supply disruptions caused by upstream capacity constraints or geopolitical factors affecting specialty chemical producers. Furthermore, the elimination of corrosive Lewis acids and the associated neutralization steps drastically reduces the volume of hazardous waste generated per kilogram of product, leading to substantial savings in waste treatment fees and environmental compliance overheads.
- Cost Reduction in Manufacturing: The economic benefits of this process are driven primarily by the substitution of high-cost raw materials with inexpensive, widely available esters and the removal of expensive catalyst recovery systems. Traditional Friedel-Crafts processes require significant capital expenditure for corrosion-resistant equipment and extensive wastewater treatment infrastructure to handle aluminum and boron salts; this new method operates with standard stainless steel reactors and generates minimal inorganic waste. Additionally, the higher overall yield and purity reduce the need for resource-intensive recrystallization cycles, further lowering the cost of goods sold (COGS) and improving the margin profile for the final API.
- Enhanced Supply Chain Reliability: Securing a stable supply of critical ophthalmic intermediates is paramount for maintaining uninterrupted drug production schedules. This synthesis route utilizes starting materials that are not subject to the same supply bottlenecks as specialized heterocycles, ensuring a more resilient supply chain capable of withstanding market fluctuations. The robustness of the reaction conditions, which do not require ultra-low temperatures or inert atmosphere protection to the same extent as prior art, also simplifies logistics and storage requirements, allowing for more flexible manufacturing planning and faster response times to sudden increases in market demand.
- Scalability and Environmental Compliance: As regulatory pressures regarding pharmaceutical manufacturing emissions intensify globally, the ability to produce high-volume intermediates with a minimal environmental footprint is a key competitive differentiator. This process is inherently green, avoiding the generation of large volumes of acidic sludge and reducing the consumption of organic solvents through efficient recycling protocols. The mild reaction conditions facilitate safe scale-up from kilogram to multi-tonne batches without the engineering complexities associated with cryogenic reactors, enabling manufacturers to rapidly expand capacity to meet commercial needs while adhering to strict ESG (Environmental, Social, and Governance) standards.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this patented synthesis method. These insights are derived directly from the experimental data and comparative examples provided in the patent documentation, offering a transparent view of the process capabilities and limitations for potential partners and licensees.
Q: How does this new synthesis route improve environmental compliance compared to traditional methods?
A: Unlike conventional Friedel-Crafts routes that generate massive amounts of acidic wastewater from Lewis acid quenching, this novel method utilizes base-catalyzed condensation and mild halogenation, significantly reducing hazardous waste volume and simplifying effluent treatment.
Q: What represents the primary cost-saving driver in this manufacturing process?
A: The primary cost reduction stems from the elimination of expensive and difficult-to-handle raw materials like indoline or indole, replacing them with commodity esters and cyclohexanone derivatives, while also removing the costly downstream purification steps required to remove heavy metal catalysts.
Q: Is this process scalable for commercial tonnage production?
A: Yes, the process operates under mild reaction temperatures (30-90°C) and atmospheric pressure without requiring cryogenic conditions (-70°C), making it highly suitable for safe and efficient scale-up in standard stainless steel reactors.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Bromfenac Sodium Supplier
At NINGBO INNO PHARMCHEM, we recognize that the transition from laboratory innovation to commercial reality requires a partner with deep technical expertise and proven manufacturing capabilities. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the promising metrics observed in patent examples can be reliably replicated in our state-of-the-art facilities. We are committed to delivering high-purity pharmaceutical intermediates that meet stringent purity specifications, supported by our rigorous QC labs equipped with advanced analytical instrumentation to verify identity, assay, and impurity profiles for every batch produced.
We invite global pharmaceutical companies and contract manufacturers to collaborate with us to evaluate the feasibility of integrating this advanced synthesis route into their supply chains. By engaging with our technical procurement team, you can request a Customized Cost-Saving Analysis tailored to your specific volume requirements, along with specific COA data and route feasibility assessments. Let us help you optimize your sourcing strategy for Bromfenac Sodium, ensuring a balance of cost efficiency, quality assurance, and supply security for your critical ophthalmic drug products.
