Revolutionizing Organosulfur Synthesis: A Cost-Effective Nickel-Catalyzed Approach for Pharmaceutical Intermediates
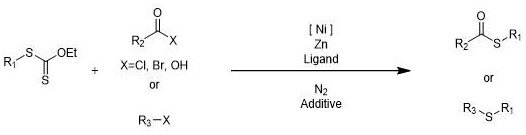
The landscape of organosulfur chemistry is undergoing a significant transformation driven by the urgent need for safer, more efficient, and environmentally benign synthetic methodologies. Patent CN114605306A introduces a groundbreaking approach to synthesizing organic sulfur compounds by leveraging alkyl xanthates as stable, odorless sulfur sources in conjunction with a robust nickel-catalyzed cross-coupling system. This innovation directly addresses the long-standing challenges associated with traditional thiol-based syntheses, such as toxicity, malodorous byproducts, and harsh reaction conditions. By utilizing elemental zinc as a reducing agent and inexpensive bipyridine ligands, this method offers a compelling alternative for the production of high-purity pharmaceutical intermediates and advanced materials. The technology represents a paradigm shift towards sustainable chemical manufacturing, where operational safety and cost-efficiency are paramount without compromising on the structural complexity or purity of the final product.
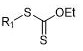
For decades, the construction of carbon-sulfur bonds has been a cornerstone in the synthesis of bioactive molecules, yet the reliance on classical reagents has imposed severe limitations on process scalability and operator safety. Conventional methods typically depend on thiols or disulfides, which are notorious for their pungent, penetrating odors and high toxicity profiles, necessitating specialized containment facilities and extensive waste treatment protocols. Furthermore, these traditional routes often require strong bases to activate the sulfur nucleophile, leading to poor functional group tolerance and potential degradation of sensitive substrates commonly found in drug candidates. The instability and limited commercial availability of many specialized thiol reagents further exacerbate supply chain vulnerabilities, making the consistent production of complex organosulfur architectures a logistical and financial burden for large-scale manufacturers.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
The reliance on mercaptans and inorganic sulfur salts in legacy synthesis pathways creates a bottleneck for modern pharmaceutical and agrochemical development. These reagents not only pose significant health hazards due to their volatility and toxicity but also complicate purification processes, often requiring rigorous scrubbing systems to manage offensive odors that can permeate entire production facilities. The necessity for strong alkaline conditions restricts the scope of compatible electrophiles, frequently resulting in lower overall yields and the formation of difficult-to-remove impurities that compromise the quality of the final active pharmaceutical ingredient. Additionally, the use of precious metal catalysts in some advanced coupling reactions drives up raw material costs, while the sensitivity of these systems to air and moisture demands stringent inert atmosphere controls that increase operational complexity and energy consumption.
The Novel Approach
The methodology disclosed in the patent circumvents these issues by employing alkyl xanthates, which are derived from inexpensive potassium ethyl xanthate, as the primary sulfur source. These intermediates are remarkably stable, easy to handle, and completely free from the irritating odors that plague thiol chemistry, thereby drastically simplifying facility requirements and improving the working environment. The reaction proceeds under mild thermal conditions, typically between 40-80°C, without the need for strong bases, allowing for exceptional compatibility with a wide array of functional groups including esters, nitriles, and halides. This nickel-catalyzed system facilitates the regioselective cleavage of the C-S bond in the xanthate, enabling efficient cross-coupling with diverse electrophiles to generate valuable thioesters and sulfides with high atom economy.
Mechanistic Insights into Nickel-Catalyzed C-S Bond Formation
The core of this technological advancement lies in the unique ability of the nickel catalyst, coordinated with bipyridine derivatives, to activate the alkyl xanthate through a single-electron transfer or oxidative addition mechanism. Unlike palladium systems which can be cost-prohibitive, the nickel species generated in situ forms a highly active catalytic complex that promotes the regioselective fragmentation of the xanthate moiety. This process generates a reactive sulfur-centered radical or anionic species that subsequently engages with the electrophilic partner, whether it be an acyl chloride, aryl halide, or other activated carbon species. The presence of elemental zinc serves as a stoichiometric reducing agent to regenerate the active low-valent nickel species, sustaining the catalytic cycle and ensuring high turnover numbers throughout the reaction duration.
Impurity control is inherently built into this mechanism due to the mildness of the reaction conditions and the specificity of the nickel-ligand interaction. The absence of strong bases prevents base-mediated side reactions such as elimination or hydrolysis, which are common pitfalls in traditional nucleophilic substitutions. Furthermore, the stability of the alkyl xanthate precursor ensures that no volatile sulfur-containing byproducts are released into the headspace, maintaining a clean reaction profile that simplifies downstream processing. The broad substrate scope demonstrated in the patent examples indicates that electronic and steric variations in both the sulfur source and the electrophile are well-tolerated, suggesting a robust mechanistic pathway that is less sensitive to subtle structural changes than conventional methods.
How to Synthesize Organosulfur Compounds Efficiently
Implementing this synthesis route requires careful attention to the molar ratios of the catalyst system and the maintenance of an inert atmosphere to prevent oxidation of the low-valent nickel species. The general procedure involves charging a reaction vessel with the nickel precursor, ligand, alkyl xanthate, electrophile, solvent, and zinc powder, followed by heating to the specified temperature range. While the patent provides numerous specific examples optimizing these parameters for different substrates, the fundamental workflow remains consistent, emphasizing the versatility of the platform for generating diverse sulfur-containing scaffolds. For detailed operational parameters and specific substrate optimizations, refer to the standardized synthesis steps outlined below.
- Prepare the reaction vessel under nitrogen atmosphere and add nickel catalyst, bipyridine ligand, alkyl xanthate, electrophile, solvent, additive, and elemental zinc.
- Stir the mixture at room temperature to ensure homogeneity, then heat the reaction to 40-80°C and maintain for 8-24 hours.
- Quench the reaction with water, extract with ethyl acetate, combine organic phases, remove solvent under reduced pressure, and purify via column chromatography.
Commercial Advantages for Procurement and Supply Chain Teams
From a procurement perspective, the transition to this alkyl xanthate-based methodology offers profound economic benefits driven by the replacement of expensive and hazardous reagents with commodity chemicals. The elimination of malodorous thiols removes the need for specialized odor-control infrastructure and reduces the regulatory burden associated with handling toxic volatile organic compounds, leading to substantial operational cost savings. Moreover, the use of nickel instead of precious metals like palladium or rhodium significantly lowers the catalyst cost per kilogram of product, while the high yields reported minimize raw material waste and maximize overall process efficiency. This combination of factors results in a more predictable and cost-effective supply chain for critical organosulfur intermediates.
- Cost Reduction in Manufacturing: The substitution of high-cost precious metal catalysts with abundant nickel salts, coupled with the use of cheap zinc powder as a reductant, drastically reduces the bill of materials for each batch. The avoidance of strong bases and the simplified workup procedures further decrease utility consumption and waste disposal fees, contributing to a leaner manufacturing cost structure without sacrificing product quality or yield.
- Enhanced Supply Chain Reliability: Alkyl xanthates and the required nickel ligands are commercially available in bulk quantities from multiple global suppliers, mitigating the risk of single-source dependency often associated with specialized thiol reagents. The stability of these starting materials allows for longer shelf life and easier logistics, ensuring continuous production capability even during periods of market volatility or transportation disruptions.
- Scalability and Environmental Compliance: The mild reaction conditions and absence of toxic sulfur emissions make this process inherently safer and easier to scale from laboratory to commercial production volumes. The reduced environmental footprint aligns with increasingly stringent global regulations on volatile organic compound emissions and hazardous waste, facilitating smoother regulatory approvals and enhancing the sustainability profile of the manufactured intermediates.
Frequently Asked Questions (FAQ)
The following questions address common technical and operational inquiries regarding the implementation of this nickel-catalyzed synthesis platform. These insights are derived directly from the experimental data and beneficial effects described in the patent documentation, providing clarity on the practical application of this technology for industrial partners seeking to optimize their sulfur chemistry workflows.
Q: What are the primary advantages of using alkyl xanthates over traditional thiols?
A: Alkyl xanthates are stable, commercially available, and crucially, they lack the unpleasant, toxic odor associated with traditional thiol reagents, significantly improving workplace safety and handling.
Q: Does this nickel-catalyzed method require strong bases?
A: No, unlike conventional methods that often rely on harsh basic conditions, this protocol operates under mild neutral conditions, enhancing functional group compatibility and reducing side reactions.
Q: What is the typical yield range for this synthesis method?
A: Under optimized conditions described in the patent, isolated yields for target organosulfur products can reach up to 80%, demonstrating high efficiency for complex molecule synthesis.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Organosulfur Compounds Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of this nickel-catalyzed technology for the production of high-value pharmaceutical and agrochemical intermediates. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory discovery to industrial reality is seamless and efficient. We are committed to delivering products that meet stringent purity specifications through our rigorous QC labs, guaranteeing that every batch of organosulfur compound adheres to the highest industry standards for consistency and performance.
We invite you to collaborate with us to leverage this advanced synthesis method for your specific project needs. Contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your current supply chain. We are ready to provide specific COA data and comprehensive route feasibility assessments to demonstrate how adopting this odorless, high-yield protocol can enhance your competitive advantage in the global market.
