Advancing Pharmaceutical Intermediate Synthesis via Efficient C(sp3)-S Bond Construction
The landscape of organosulfur chemistry is undergoing a significant transformation driven by the urgent need for more sustainable and economically viable synthetic methodologies. Patent CN115322128A introduces a groundbreaking approach to constructing C(sp3)-S bonds, a structural motif ubiquitous in high-value pharmaceuticals, agrochemicals, and advanced materials. This technology leverages a novel catalytic system that replaces traditional stoichiometric metal usage with catalytic amounts of inexpensive metal powders, such as manganese or zinc. By utilizing thioformates as a stable and low-toxicity sulfur source, this method addresses long-standing challenges regarding safety, odor, and waste generation. For R&D directors and procurement strategists, this represents a pivotal shift towards greener chemistry that does not compromise on efficiency or yield, offering a robust pathway for the synthesis of complex sulfur-containing intermediates essential for modern drug discovery pipelines.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of C(sp3)-S bonds has been dominated by nucleophilic substitution reactions between alkyl halides and thiols or metal thiosalts. These conventional pathways are fraught with significant operational and safety drawbacks that hinder large-scale adoption. Thiols are notoriously malodorous and highly toxic, posing severe health risks to laboratory personnel and requiring specialized containment infrastructure. Furthermore, these reactions often necessitate harsh conditions, including high temperatures and strong bases, which can lead to poor functional group tolerance and unwanted side reactions like elimination or homodimerization. The reliance on stoichiometric amounts of metals or expensive transition metal catalysts further inflates production costs and complicates downstream purification processes due to heavy metal residue concerns.
The Novel Approach
In stark contrast, the methodology disclosed in CN115322128A utilizes thioformates as a superior sulfur source, which are stable, easy to handle, and devoid of the offensive odors characteristic of thiols. This innovative protocol employs catalytic quantities of cheap metal powders to facilitate the cross-coupling with non-activated alkyl halides under mild conditions, typically ranging from 30°C to 100°C. The reaction exhibits exceptional functional group compatibility, accommodating esters, nitriles, ethers, and even complex drug-like scaffolds without degradation. By shifting from stoichiometric to catalytic regimes and replacing hazardous reagents with benign alternatives, this approach drastically simplifies the workflow while enhancing the overall safety profile and economic feasibility of producing high-purity organosulfur compounds.
Mechanistic Insights into Cheap Metal-Catalyzed C-S Bond Formation
The core of this technological advancement lies in the unique activation mechanism facilitated by the cheap metal catalyst. Unlike traditional palladium-catalyzed cycles that often require intricate ligand systems, this process likely involves the oxidative addition of the low-valent metal species into the carbon-halogen bond of the alkyl halide, generating a highly reactive organometallic intermediate. Simultaneously, the metal catalyst promotes the regioselective cleavage of the C-S bond in the thioformate reagent, effectively generating a sulfur-centered radical or nucleophilic sulfur species in situ. This dual activation strategy ensures efficient coupling to form the desired C(sp3)-S bond while minimizing side reactions.
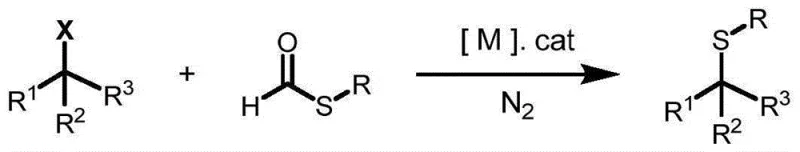
The mechanistic pathway is designed to suppress common pitfalls such as beta-hydride elimination, which frequently plagues alkyl-metal species. The use of thioformates is particularly strategic; their high atom economy ensures that the formyl byproduct is easily managed, and their stability prevents premature decomposition. This controlled radical or ionic pathway allows for the successful coupling of secondary alkyl halides, which are traditionally difficult substrates due to steric hindrance. The result is a highly selective transformation that delivers target molecules with yields reaching up to 90% under optimized conditions, providing R&D teams with a reliable tool for late-stage functionalization of complex molecular architectures.
How to Synthesize Organosulfur Compounds Efficiently
Implementing this synthesis route requires careful attention to reaction parameters to maximize yield and purity. The process is operationally simple, involving the mixing of the alkyl halide substrate, thioformate sulfur source, and metal catalyst in a polar aprotic solvent under an inert nitrogen atmosphere. The reaction temperature and time are adjustable based on the specific substrate reactivity, generally falling within the 8 to 24-hour window. Following the reaction, standard aqueous workup procedures effectively remove inorganic salts and metal residues, followed by chromatographic purification to isolate the final product. For detailed procedural specifics and optimization guidelines tailored to your specific substrate, please refer to the standardized synthesis steps outlined below.
- Under nitrogen protection, add cheap metal powder catalyst (e.g., Mn, Zn), non-activated alkyl halide, and thioformate sulfur source into a reaction flask with organic solvent.
- Heat the reaction mixture to temperatures between 30-100°C and stir for 8-24 hours to allow the coupling reaction to proceed.
- Quench the reaction with water, extract with ethyl acetate, wash with brine, dry over anhydrous sodium sulfate, and purify via silica gel chromatography.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, this technology offers compelling advantages that directly impact the bottom line and supply chain resilience. The substitution of expensive noble metal catalysts with commodity metal powders like manganese or zinc results in a drastic reduction in raw material costs. Furthermore, the elimination of hazardous thiols reduces the regulatory burden and safety compliance costs associated with handling toxic substances. The mild reaction conditions also translate to lower energy consumption, as there is no need for extreme heating or cryogenic cooling, making the process more environmentally sustainable and aligned with green chemistry principles.
- Cost Reduction in Manufacturing: The utilization of cheap metal catalysts and readily available thioformates significantly lowers the bill of materials compared to traditional methods relying on precious metals or specialized ligands. The high atom economy and reduced waste generation further contribute to substantial cost savings in waste disposal and raw material procurement. By streamlining the purification process through the avoidance of complex metal-ligand complexes, manufacturers can achieve higher throughput with lower operational expenditures.
- Enhanced Supply Chain Reliability: The starting materials for this process, including alkyl halides and thioformates, are commercially abundant and can be sourced from multiple suppliers, mitigating the risk of single-source dependency. The robustness of the reaction conditions ensures consistent batch-to-batch reproducibility, which is critical for maintaining steady supply lines to downstream pharmaceutical customers. Additionally, the stability of the reagents allows for easier storage and transportation, reducing logistics complexities and potential delays.
- Scalability and Environmental Compliance: The protocol has been demonstrated to be scalable, with successful gram-scale reactions showing maintained efficiency, indicating readiness for kilogram to ton-scale production. The use of less toxic reagents and the generation of benign byproducts simplify environmental compliance and wastewater treatment processes. This aligns with increasingly stringent global environmental regulations, ensuring long-term operational continuity without the risk of regulatory shutdowns due to safety or pollution violations.
Frequently Asked Questions (FAQ)
The following questions address common technical inquiries regarding the implementation and scope of this catalytic method. These insights are derived directly from the experimental data and beneficial effects reported in the patent documentation, providing clarity on substrate compatibility, catalyst selection, and process safety. Understanding these nuances is essential for process chemists evaluating this technology for integration into existing manufacturing workflows.
Q: What are the advantages of using thioformates over thiols for C-S bond construction?
A: Thioformates offer significantly higher stability, lower toxicity, and lack the pungent odor associated with traditional thiols. They also provide better atom economy and functional group compatibility in catalytic systems.
Q: Can this method be scaled up for commercial production?
A: Yes, the patent demonstrates successful gram-scale synthesis (e.g., 4.5 mmol scale yielding over 1g product with 92% yield), indicating strong potential for commercial scale-up using standard equipment.
Q: What types of metal catalysts are suitable for this transformation?
A: The method utilizes cheap and readily available metal powders such as manganese, zinc, iron, copper, or bis-(1,5-cyclooctadiene)nickel, avoiding the need for expensive noble metals.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Organosulfur Compounds Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of this catalytic C(sp3)-S bond formation technology for the pharmaceutical industry. As a dedicated CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that innovative laboratory methods are seamlessly translated into robust manufacturing processes. Our rigorous QC labs and commitment to stringent purity specifications guarantee that every batch of organosulfur intermediate meets the highest quality standards required for API synthesis, providing our partners with confidence in their supply chain.
We invite you to collaborate with us to leverage this cost-effective and sustainable synthetic route for your next project. Contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your specific molecule. We are prepared to provide specific COA data and comprehensive route feasibility assessments to demonstrate how this technology can optimize your production costs and accelerate your time to market.
