Revolutionizing C-S Bond Formation: A Scalable Palladium-Indium Catalytic Route for High-Purity Pharmaceutical Intermediates
Revolutionizing C-S Bond Formation: A Scalable Palladium-Indium Catalytic Route for High-Purity Pharmaceutical Intermediates
The landscape of organic synthesis is constantly evolving, driven by the relentless demand for more efficient, sustainable, and cost-effective methodologies to construct critical molecular architectures. In this context, patent CN101808985A introduces a groundbreaking approach to the formation of carbon-sulfur bonds, a structural motif ubiquitous in bioactive molecules, agrochemicals, and advanced materials. This technology leverages the unique reactivity of organosulfur indium complexes in conjunction with a palladium catalyst system to overcome the longstanding limitations of traditional thiolation reactions. By shifting away from the reliance on volatile and malodorous free thiols or unstable thiolate salts, this invention provides a robust platform for generating diverse organosulfur compounds with exceptional precision. For R&D directors and procurement specialists alike, this represents a significant opportunity to streamline synthetic routes, reduce hazardous waste streams, and secure a more reliable supply chain for high-value sulfur-containing intermediates.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the construction of carbon-sulfur bonds has been plagued by significant operational and chemical challenges that hinder efficient commercial manufacturing. Traditional protocols typically rely on the reaction between aromatic halides and thiols in the presence of transition metal catalysts such as copper, nickel, or palladium. However, these legacy methods frequently necessitate the use of strong, atmospherically unstable bases like sodium tert-butoxide to generate the active thiolate nucleophile in situ. This requirement introduces severe safety hazards and complicates process handling, particularly on a large industrial scale where moisture sensitivity can lead to batch failures. Furthermore, conventional processes often demand excessively high reaction temperatures, frequently exceeding 140°C, and prolonged heating times ranging from 24 to 48 hours to drive the reaction to completion. Such energy-intensive conditions not only inflate operational costs but also increase the risk of thermal decomposition of sensitive functional groups, leading to complex impurity profiles that are difficult and expensive to purge during downstream purification.
The Novel Approach
In stark contrast to these cumbersome traditional pathways, the methodology disclosed in patent CN101808985A offers a streamlined and highly efficient alternative that fundamentally redefines the parameters of C-S bond formation. By utilizing pre-formed organosulfur indium complexes as the sulfur source, this novel approach eliminates the need for external strong bases and free thiols, thereby mitigating odor issues and enhancing reaction stability. The core innovation lies in the ability of the indium reagent to act as a potent nucleophile under relatively mild conditions, typically requiring temperatures around 100°C and reaction times as short as 2 to 4 hours. This dramatic reduction in thermal load and processing time translates directly into substantial energy savings and increased reactor throughput. Moreover, the method demonstrates remarkable versatility, accommodating a wide array of substrates including aromatic halides, triflates, and even unsaturated systems, while consistently delivering quantitative yields that minimize raw material waste and maximize overall process economy.
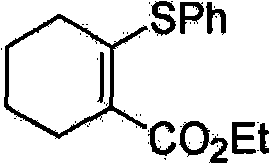
Mechanistic Insights into Palladium-Xantphos Catalyzed Transmetallation
The exceptional performance of this synthetic route can be attributed to the synergistic interaction between the palladium catalyst and the specialized phosphine ligand, specifically 4,5-bisdiphenylphosphine-9,9-dimethylxanthene, commonly known as Xantphos. Mechanistically, the reaction proceeds through a catalytic cycle where the palladium center first undergoes oxidative addition with the organic halide or pseudohalide electrophile. The unique geometric properties of the Xantphos ligand, characterized by its large natural bite angle, play a critical role in stabilizing the palladium intermediate and facilitating the subsequent transmetallation step with the organosulfur indium complex. Unlike simpler monodentate ligands, Xantphos promotes the formation of a coordinatively unsaturated species that is highly receptive to the transfer of the sulfur moiety from the indium atom to the palladium center. This efficient transmetallation is the rate-determining step in many cross-coupling reactions, and the indium reagent's inherent Lewis acidity appears to enhance the nucleophilicity of the sulfur atom, allowing the reaction to proceed rapidly without the need for exogenous activators.
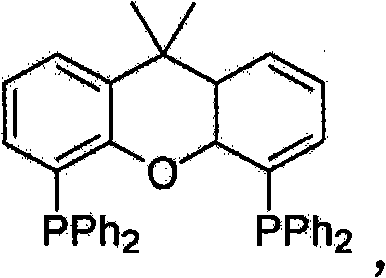
Furthermore, the final reductive elimination step, which releases the desired organosulfur product and regenerates the active palladium catalyst, is significantly accelerated by the electronic environment created by the ligand system. This ensures that the catalytic turnover number remains high throughout the reaction course, allowing for the use of low catalyst loadings (1-10 mol%) without compromising conversion rates. From an impurity control perspective, the mildness of the reaction conditions prevents the formation of common side products such as homocoupled disulfides or dehalogenated arenes, which are frequently observed in harsher copper-mediated protocols. The result is a crude reaction mixture with a significantly cleaner profile, reducing the burden on purification units and ensuring that the final high-purity pharmaceutical intermediates meet stringent regulatory specifications with minimal processing effort.
How to Synthesize Organosulfur Compounds Efficiently
Implementing this advanced synthesis strategy requires careful attention to reagent preparation and atmospheric control to fully realize the benefits of the indium-mediated pathway. The process begins with the generation of the organosulfur indium complex, which serves as the stable sulfur donor, followed by its introduction into the palladium-catalyzed coupling cycle. Operators must ensure that the reaction environment is strictly inert to prevent oxidation of the sensitive intermediates, although the indium reagents themselves exhibit greater stability compared to traditional alkali metal thiolates. The following guide outlines the standardized operational procedure derived from the patent examples, providing a clear roadmap for laboratory validation and subsequent process scale-up. Detailed standardized synthesis steps are provided in the guide below.
- Prepare the catalytic system by stirring palladium acetate and Xantphos ligand in DMF solvent under an inert nitrogen atmosphere to ensure catalyst activation.
- Introduce the nucleophilic substrate, such as an aromatic halide or triflate, to the catalyst mixture and allow for initial coordination at room temperature.
- Add the organosulfur indium complex reagent and a base like DIPEA, then heat the reaction mixture to 100°C for 2-4 hours to achieve quantitative conversion.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain leaders, the adoption of this technology offers compelling economic and logistical advantages that extend far beyond simple yield improvements. The shift towards a stoichiometric, high-efficiency process directly addresses the critical pain points of raw material utilization and waste management that often erode profit margins in fine chemical manufacturing. By eliminating the need for excess reactants and expensive, specialized additives that are common in legacy methods, the overall bill of materials is significantly optimized. Additionally, the reduction in reaction time from days to mere hours allows for a drastic increase in asset utilization, enabling manufacturing facilities to produce more batches within the same timeframe without requiring capital investment in new reactor capacity. This agility is crucial for responding to fluctuating market demands and ensuring continuous supply continuity for downstream pharmaceutical clients.
- Cost Reduction in Manufacturing: The economic impact of this methodology is profound, primarily driven by the elimination of costly purification steps and the reduction in energy consumption associated with prolonged heating. Since the reaction proceeds with quantitative yields and high selectivity, the need for extensive chromatographic purification or recrystallization is minimized, leading to substantial savings in solvent usage and labor costs. Furthermore, the avoidance of strong, hazardous bases reduces the costs associated with specialized containment equipment and waste neutralization protocols. The ability to use readily available aromatic halides and triflates as electrophiles, rather than more expensive or less stable alternatives, further contributes to a lower cost of goods sold, making the final organosulfur intermediates more competitive in the global marketplace.
- Enhanced Supply Chain Reliability: From a sourcing perspective, the reagents required for this process, including indium salts and standard phosphine ligands, are commercially available from multiple established suppliers, mitigating the risk of single-source dependency. The robustness of the reaction conditions means that the process is less susceptible to variations in raw material quality or minor fluctuations in environmental parameters, ensuring consistent batch-to-batch reproducibility. This reliability is paramount for maintaining long-term supply agreements with major pharmaceutical companies, where any disruption can have cascading effects on drug development timelines. The simplified workflow also reduces the technical barrier for contract manufacturing organizations, expanding the pool of qualified vendors capable of executing the synthesis effectively.
- Scalability and Environmental Compliance: As regulatory pressures regarding environmental sustainability intensify, this green chemistry approach positions manufacturers favorably for future compliance. The process generates significantly less hazardous waste due to the absence of heavy metal contaminants often associated with copper catalysts and the reduced volume of solvent required for workup. The mild operating temperatures and lack of noxious thiol odors improve workplace safety and reduce the load on scrubber systems. These factors collectively simplify the environmental permitting process for new production lines and support corporate sustainability goals, making the technology an attractive option for companies aiming to reduce their carbon footprint while maintaining high production volumes.
Frequently Asked Questions (FAQ)
To assist technical teams in evaluating the feasibility of this synthesis route for their specific projects, we have compiled answers to the most common inquiries regarding the practical application of organosulfur indium complexes. These insights are derived directly from the experimental data and technical specifications outlined in the patent documentation, ensuring accuracy and relevance for process development scientists. Understanding these nuances is essential for making informed decisions about integrating this technology into existing manufacturing pipelines.
Q: What are the primary advantages of using organosulfur indium complexes over traditional thiolates?
A: Organosulfur indium complexes offer enhanced nucleophilicity without requiring atmospherically unstable strong bases, allowing for milder reaction conditions and eliminating the malodorous handling issues associated with free thiols.
Q: Can this methodology be applied to sterically hindered substrates?
A: Yes, the use of the Xantphos ligand with a wide bite angle facilitates the oxidative addition and reductive elimination steps even in sterically demanding environments, enabling high yields for complex aromatic systems.
Q: Is this process suitable for large-scale commercial production?
A: Absolutely, the protocol utilizes stoichiometric amounts of reagents without excess, operates at moderate temperatures (100°C), and achieves quantitative yields, making it highly efficient and cost-effective for kilogram-to-ton scale manufacturing.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Organosulfur Compounds Supplier
At NINGBO INNO PHARMCHEM, we recognize that the transition from laboratory innovation to commercial reality requires a partner with deep technical expertise and robust manufacturing capabilities. Our team of experienced chemists has extensively evaluated the palladium-indium catalytic system described in CN101808985A and is fully prepared to adapt this route for the custom synthesis of complex organosulfur intermediates tailored to your specific drug discovery programs. We possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the high yields and purity observed in the lab are faithfully reproduced at an industrial scale. Our state-of-the-art facilities are equipped with rigorous QC labs and advanced analytical instrumentation to guarantee that every batch meets stringent purity specifications, providing you with the confidence needed to advance your candidates through clinical trials.
We invite you to engage with our technical procurement team to discuss how this innovative synthesis method can be leveraged to optimize your supply chain and reduce overall project costs. By requesting a Customized Cost-Saving Analysis, you can gain a clear understanding of the potential economic benefits specific to your target molecule. We encourage you to reach out today to obtain specific COA data and route feasibility assessments that will demonstrate the viability of this approach for your next-generation pharmaceutical intermediates. Let us help you turn this cutting-edge chemistry into a competitive advantage for your business.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
