Advanced Chiral Catalytic Hydrogenation for High-Purity Ursodeoxycholic Acid Manufacturing
Advanced Chiral Catalytic Hydrogenation for High-Purity Ursodeoxycholic Acid Manufacturing
The pharmaceutical industry continuously seeks robust synthetic pathways that maximize yield while minimizing environmental impact and production costs. Patent CN102070693A introduces a transformative approach to the synthesis of ursodeoxycholic acid, a critical bile acid used extensively in hepatology and cholesterol management. This technology replaces hazardous and non-selective reduction methods with a sophisticated chiral catalytic hydrogenation process. By leveraging specific transition metal catalysts under controlled alkaline conditions, this method achieves exceptional stereoselectivity, directly addressing the longstanding challenge of epimeric impurity formation. For global procurement leaders and R&D directors, this patent represents a significant opportunity to secure a reliable ursodeoxycholic acid supplier capable of delivering ultra-high purity intermediates without the burden of extensive purification workflows.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
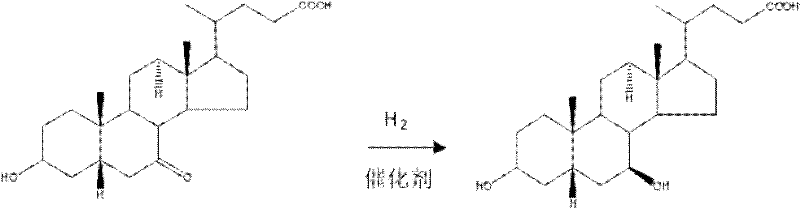
Historically, the industrial production of ursodeoxycholic acid has relied heavily on the reduction of 7-ketodeoxycholic acid using an alcohol and metallic sodium system. While chemically feasible, this legacy approach suffers from inherent thermodynamic and kinetic limitations regarding stereochemical control. The reaction environment created by dissolving metals in alcohol lacks the precise spatial orientation required to distinguish between the alpha and beta faces of the steroid nucleus effectively. Consequently, the reaction typically yields a crude mixture containing only about 80% of the desired ursodeoxycholic acid, with the remaining 20% reverting to the starting material, chenodeoxycholic acid, or forming other isomeric byproducts. This poor selectivity necessitates rigorous and costly downstream purification steps, such as repeated crystallizations or chromatographic separations, which drastically reduce overall process efficiency and increase waste generation.
The Novel Approach
In stark contrast, the methodology disclosed in CN102070693A utilizes a chiral catalytic hydrogenation strategy that fundamentally alters the reaction landscape. By introducing specialized chiral catalysts—such as those based on nickel, palladium, platinum, or ruthenium—into an alkaline reaction medium, the process exerts precise control over the hydrogen addition to the C-7 carbonyl group. This catalytic system ensures that hydrogen atoms are delivered almost exclusively to the beta-face of the molecule, resulting in a product purity exceeding 99%. The elimination of the competing reduction pathway means that the formation of chenodeoxycholic acid is negligible, effectively bypassing the need for difficult separation processes. This technological leap not only simplifies the manufacturing workflow but also significantly enhances the economic viability of producing high-purity pharmaceutical intermediates.
Mechanistic Insights into Chiral Catalytic Hydrogenation
The core innovation lies in the interaction between the chiral catalyst surface and the steroid substrate under alkaline conditions. In this mechanism, the base plays a dual role: it facilitates the formation of an enolate or activated intermediate that coordinates more effectively with the metal center of the catalyst, and it stabilizes the transition state leading to the 7-beta-hydroxy configuration. The chiral ligands or the specific crystal face of the heterogeneous catalyst create a steric environment that energetically favors the approach of molecular hydrogen from one specific direction. This asymmetric induction is critical because the difference in energy between the transition states leading to the alpha and beta alcohols is amplified by the catalyst, driving the equilibrium overwhelmingly toward the desired ursodeoxycholic acid. Understanding this mechanistic nuance is vital for R&D teams aiming to replicate or optimize the process for commercial scale-up of complex pharmaceutical intermediates.
Furthermore, the choice of solvent and reaction parameters profoundly influences the impurity profile. The patent specifies the use of alcohols or esters, which not only dissolve the lipophilic steroid backbone effectively but also participate in the stabilization of the catalytic species. By maintaining the pressure between 0-20MPa and temperatures between 10-80°C, the reaction kinetics are tuned to favor the rapid consumption of the ketone before any epimerization can occur. This tight control over reaction conditions ensures that side reactions, such as over-reduction of other functional groups on the steroid ring or dehydration, are suppressed. The result is a clean reaction mixture where the primary solid obtained after acidification is the target API intermediate, requiring minimal washing to achieve stringent purity specifications required by regulatory bodies.
How to Synthesize Ursodeoxycholic Acid Efficiently
The synthesis protocol outlined in the patent provides a clear roadmap for transitioning from laboratory benchtop experiments to pilot plant operations. The process begins with the preparation of the 7-keto precursor, followed by its dissolution in a selected solvent system like methanol or ethyl acetate. The addition of a base, such as potassium ethoxide or sodium hydroxide, primes the system for the subsequent catalytic step. Once the chiral catalyst is introduced, the reactor is purged and pressurized with hydrogen gas. The reaction proceeds under constant monitoring until conversion is complete, after which the solvent is distilled off, and the product is precipitated by adding water and adjusting the pH.
- Oxidize chenodeoxycholic acid to generate 7-ketodeoxycholic acid using standard oxidation protocols.
- Dissolve the 7-keto intermediate in alcohol or ester solvents, add a chiral catalyst (e.g., Ni, Pd, Pt, Ru) and an alkali base.
- Conduct hydrogenation at 10-80°C and 0-20MPa pressure, followed by solvent removal, water addition, acidification, and crystallization to isolate pure ursodeoxycholic acid.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this chiral hydrogenation technology offers profound strategic benefits beyond mere chemical elegance. The primary advantage lies in the drastic simplification of the downstream processing train. By achieving near-quantitative selectivity, the manufacturer eliminates the need for expensive and time-consuming separation units designed to remove the chenodeoxycholic acid epimer. This reduction in unit operations translates directly into lower capital expenditure for new facilities and reduced operating expenses for existing plants. Furthermore, the use of catalytic hydrogenation replaces the hazardous metallic sodium reduction, thereby mitigating significant safety risks associated with handling pyrophoric materials and generating large volumes of reactive waste. This shift aligns perfectly with modern ESG (Environmental, Social, and Governance) goals, making the supply chain more resilient and compliant with increasingly strict environmental regulations.
- Cost Reduction in Manufacturing: The economic impact of this process is driven by the high atom economy and the elimination of yield losses associated with purification. In traditional methods, a significant portion of the valuable starting material is lost or downgraded to a lower-value byproduct, necessitating recycling loops that consume energy and time. With the new chiral catalytic route, the yield of ursodeoxycholic acid is maximized, meaning less raw material is required per kilogram of final product. Additionally, the catalyst loading can be optimized to very low levels (as low as 1/50000 weight ratio in some embodiments), reducing the cost contribution of precious metals. The simplified workup procedure, involving simple distillation and crystallization rather than complex chromatography, further drives down utility costs and labor requirements, resulting in substantial cost savings in API manufacturing.
- Enhanced Supply Chain Reliability: Supply continuity is often threatened by the complexity of synthesis routes that rely on hard-to-source reagents or dangerous conditions that limit production throughput. This hydrogenation method utilizes widely available industrial gases (hydrogen) and common solvents, reducing dependency on niche chemical suppliers. The mild reaction conditions (10-80°C) allow for the use of standard stainless steel reactors without the need for exotic metallurgy required for highly corrosive sodium amalgam processes. This compatibility with standard pharmaceutical manufacturing equipment ensures that production can be scaled up rapidly to meet market demand without lengthy equipment qualification periods. Consequently, partners can expect reducing lead time for high-purity pharmaceutical intermediates, ensuring a steady flow of materials for downstream drug formulation.
- Scalability and Environmental Compliance: Scaling chemical processes often introduces new challenges regarding heat transfer and mixing, but the homogeneous or heterogeneous nature of this catalytic system is well-suited for large-scale batch or continuous flow reactors. The absence of heavy metal waste streams typical of stoichiometric reductions simplifies wastewater treatment and disposal. The process generates primarily water and recovered solvents as waste, which are easier to manage and recycle compared to the saline and organic waste mixtures from sodium reductions. This environmental friendliness not only reduces disposal costs but also future-proofs the manufacturing site against tightening environmental legislation. The ability to run the reaction at moderate pressures (up to 20MPa) is well within the capabilities of modern high-pressure hydrogenation vessels, ensuring safe and efficient commercial scale-up of complex steroid derivatives.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this chiral hydrogenation technology. These insights are derived directly from the experimental data and claims presented in the patent documentation, providing a factual basis for decision-making. Understanding these details helps stakeholders evaluate the feasibility of integrating this route into their existing production portfolios.
Q: How does the chiral catalytic method improve stereoselectivity compared to traditional sodium reduction?
A: Traditional alcohol-sodium systems typically yield a mixture of approximately 80% ursodeoxycholic acid and 20% chenodeoxycholic acid due to poor stereocontrol. The patented chiral catalytic hydrogenation method achieves over 99% selectivity for the desired 7-beta hydroxyl configuration, virtually eliminating the formation of the epimeric impurity and removing the need for complex downstream separation.
Q: What are the operational pressure and temperature ranges for this hydrogenation process?
A: The process operates under relatively mild and controllable conditions, maintaining a hydrogen pressure between 0-20MPa and a reaction temperature ranging from 10-80°C. These moderate parameters facilitate safer scale-up and reduce the energy intensity compared to more extreme reduction conditions often found in older methodologies.
Q: Which types of solvents and catalysts are compatible with this synthesis route?
A: The method demonstrates robust flexibility, utilizing common alcohol solvents like methanol, ethanol, or isopropanol, as well as ester solvents such as ethyl acetate. Compatible chiral catalysts include nickel, palladium, platinum, ruthenium, rhodium, iridium, or copper chromite, allowing manufacturers to optimize based on availability and cost.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Ursodeoxycholic Acid Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of adopting advanced synthetic technologies to maintain competitiveness in the global pharmaceutical market. Our team of expert chemists has extensively analyzed the potential of chiral catalytic hydrogenation for bile acid derivatives and possesses the technical capability to implement this route effectively. We bring extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from patent to practice is seamless. Our facilities are equipped with state-of-the-art high-pressure hydrogenation reactors and rigorous QC labs capable of verifying stringent purity specifications, guaranteeing that every batch of ursodeoxycholic acid meets the highest international standards for safety and efficacy.
We invite pharmaceutical companies and contract manufacturers to collaborate with us to leverage this innovative synthesis method. By partnering with our technical procurement team, you can access a Customized Cost-Saving Analysis tailored to your specific volume requirements and quality needs. We encourage you to contact us today to request specific COA data and route feasibility assessments, allowing you to make informed decisions that will optimize your supply chain and enhance your product portfolio with high-quality, cost-effective active ingredients.
