Scalable Synthesis of High-Purity Aryl Sulfones Using Amino Acid-Promoted Copper Catalysis for Global Supply Chains
The landscape of organic synthesis for pharmaceutical intermediates is constantly evolving, driven by the need for more sustainable, cost-effective, and robust methodologies. A pivotal advancement in this domain is documented in Chinese Patent CN1332944C, which discloses a highly efficient method for preparing aryl sulfones through the coupling of aryl halides and hydrocarbyl sulfinates. This technology leverages a synergistic catalytic system comprising copper(I) iodide (CuI) as the catalyst and amino acids, specifically their sodium salts like L-proline sodium salt, as promoting ligands. Unlike traditional methods that often rely on precious metals or harsh oxidative conditions, this invention enables the formation of the carbon-sulfur bond under remarkably mild conditions. The strategic use of amino acid additives not only accelerates the reaction kinetics but also stabilizes the copper species, allowing the transformation to proceed at temperatures significantly lower than previously reported literature methods. For global supply chain managers and R&D directors, this represents a critical opportunity to optimize the production of aryl sulfone scaffolds, which are ubiquitous motifs in modern medicinal chemistry and agrochemical formulations.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of aryl sulfones has been fraught with significant technical and economic challenges that hinder efficient large-scale manufacturing. Conventional routes often involve the oxidation of corresponding aryl sulfides, which can lead to over-oxidation issues and difficult impurity profiles, or the sulfonation of aromatic hydrocarbons under strongly acidic conditions that are incompatible with acid-sensitive functional groups. While transition metal catalysis offered a solution, early palladium-catalyzed methods utilizing aryl boronic acids or sulfonyl chlorides introduced prohibitive costs due to the price of palladium and the necessity for expensive, air-sensitive phosphine ligands. Furthermore, earlier copper-catalyzed variants reported in scientific literature frequently suffered from the requirement of stoichiometric amounts of copper salts, necessitating rigorous and costly purification steps to remove residual heavy metals from the final API intermediate. These legacy processes often demanded high reaction temperatures exceeding 120°C, posing safety risks and limiting the substrate scope to only the most thermally stable compounds, thereby restricting their utility in the synthesis of complex, multifunctional drug candidates.
The Novel Approach
The methodology outlined in patent CN1332944C fundamentally disrupts these limitations by introducing an amino acid-promoted copper catalytic cycle that operates with exceptional efficiency and selectivity. By utilizing catalytic amounts of CuI (typically 5 mol% to 20 mol%) in conjunction with a simple amino acid ligand such as L-proline sodium salt, the reaction achieves high yields under温和 conditions ranging from 80°C to 95°C. This approach eliminates the need for stoichiometric copper, drastically reducing metal waste and downstream purification burdens. The versatility of this system is exemplified by its ability to couple a wide array of aryl iodides and bromides, including those bearing sensitive functionalities like esters, amides, and even protected amino acid side chains. 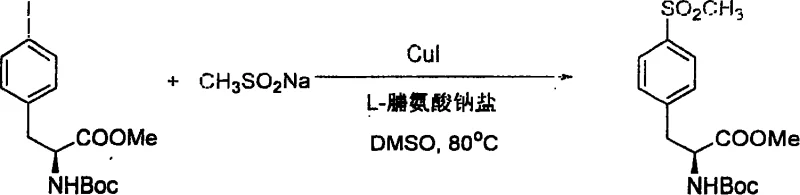 As illustrated in the synthesis of complex chiral derivatives, the mild nature of this protocol preserves stereochemical integrity and prevents the decomposition of labile protecting groups such as Boc, making it uniquely suited for the late-stage functionalization of advanced pharmaceutical intermediates where structural complexity is highest.
As illustrated in the synthesis of complex chiral derivatives, the mild nature of this protocol preserves stereochemical integrity and prevents the decomposition of labile protecting groups such as Boc, making it uniquely suited for the late-stage functionalization of advanced pharmaceutical intermediates where structural complexity is highest.
Mechanistic Insights into Amino Acid-Promoted CuI Catalysis
The mechanistic elegance of this transformation lies in the role of the amino acid ligand, which acts as a bidentate chelator to stabilize the copper center throughout the catalytic cycle. It is hypothesized that the sodium salt of the amino acid coordinates with the CuI species to form a soluble, active copper-amino acid complex that facilitates the oxidative addition of the aryl halide. This coordination sphere lowers the activation energy barrier for the subsequent transmetallation with the sulfinate anion, a step that is often rate-limiting in uncatalyzed or poorly ligated systems. The presence of the amino acid ligand prevents the aggregation of copper species into inactive clusters, ensuring a sustained concentration of the active catalyst throughout the prolonged reaction times of 24 to 36 hours. This stabilization effect is crucial for maintaining high turnover numbers and achieving the consistent yields observed across diverse substrate classes, from electron-rich anisoles to electron-deficient nitriles and trifluoromethyl-substituted aromatics.
From an impurity control perspective, this catalytic system offers superior chemoselectivity compared to radical-based or harsh oxidative methods. The mild thermal profile (80°C - 95°C) minimizes thermal degradation pathways and side reactions such as homocoupling of the aryl halide or desulfonylation, which are common pitfalls in high-temperature sulfone syntheses. 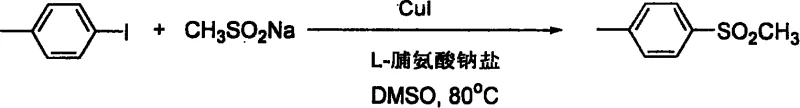 The reaction conditions are sufficiently gentle to tolerate nucleophilic functional groups like free amines and hydroxyls without the need for extensive protection-deprotection sequences, as evidenced by the successful synthesis of p-aminophenyl methyl sulfone and p-hydroxyphenyl derivatives directly from their corresponding halides. This inherent selectivity simplifies the impurity profile, resulting in crude products that are easier to purify via standard crystallization or chromatography, thereby enhancing the overall process mass intensity and reducing solvent consumption in the workup phase.
The reaction conditions are sufficiently gentle to tolerate nucleophilic functional groups like free amines and hydroxyls without the need for extensive protection-deprotection sequences, as evidenced by the successful synthesis of p-aminophenyl methyl sulfone and p-hydroxyphenyl derivatives directly from their corresponding halides. This inherent selectivity simplifies the impurity profile, resulting in crude products that are easier to purify via standard crystallization or chromatography, thereby enhancing the overall process mass intensity and reducing solvent consumption in the workup phase.
How to Synthesize Aryl Sulfones Efficiently
Implementing this amino acid-promoted coupling reaction requires careful attention to reagent quality and atmospheric control to maximize yield and reproducibility. The protocol typically involves charging a reaction vessel with the aryl halide substrate, sodium alkyl sulfinate, CuI catalyst, and the amino acid sodium salt ligand in a polar aprotic solvent such as DMSO or DMF. The detailed standardized synthesis steps see the guide below.
- Combine aryl halide, sodium alkyl sulfinate, CuI catalyst, L-proline sodium salt ligand, and base in DMSO solvent under inert atmosphere.
- Heat the reaction mixture to a moderate temperature range of 80°C to 95°C and maintain stirring for 24 to 36 hours to ensure complete conversion.
- Quench the reaction with water, extract the product using ethyl acetate, dry the organic layer, and purify via silica gel column chromatography.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this CuI-amino acid catalytic technology translates into tangible strategic advantages regarding cost structure and supply reliability. The shift from precious metal catalysts to base metal copper represents a fundamental decoupling from the volatile pricing dynamics of the platinum group metals market. Since copper iodide and common amino acids like proline are commodity chemicals produced on a massive scale globally, their availability is virtually guaranteed, insulating the manufacturing process from supply shocks that frequently affect specialized palladium catalysts. Furthermore, the air stability of both the catalyst and the ligand simplifies storage and handling requirements, eliminating the need for expensive inert atmosphere gloveboxes or specialized cold-chain logistics often mandated for sensitive organometallic reagents, thus streamlining warehouse operations and reducing overhead costs.
- Cost Reduction in Manufacturing: The economic impact of this technology is driven by the drastic reduction in catalyst costs and the elimination of expensive ligand systems. By replacing palladium with copper and proprietary phosphines with bulk amino acids, the direct material cost of the catalytic system is lowered by orders of magnitude. Additionally, the moderate reaction temperatures (80°C - 95°C) significantly reduce energy consumption compared to high-temperature protocols, leading to lower utility costs per kilogram of product. The ability to use catalytic rather than stoichiometric amounts of copper further reduces the burden on waste treatment facilities, lowering the environmental compliance costs associated with heavy metal disposal and wastewater treatment.
- Enhanced Supply Chain Reliability: Supply chain resilience is bolstered by the use of universally available raw materials. Aryl iodides and bromides, along with sodium sulfinates, are standard building blocks available from multiple global suppliers, reducing single-source dependency risks. The robustness of the reaction conditions means that the process is less sensitive to minor variations in raw material quality or environmental factors, ensuring consistent batch-to-batch performance. This reliability allows for more accurate production planning and shorter lead times, as there is less risk of batch failure or the need for re-processing due to catalyst deactivation, which is a common issue with less stable catalytic systems.
- Scalability and Environmental Compliance: The scalability of this process is exceptional due to the absence of hazardous reagents and the use of standard solvents like DMSO. The mild operating conditions reduce the risk of thermal runaway, making the transition from laboratory scale to multi-ton commercial production safer and more predictable. From an environmental standpoint, the use of biodegradable amino acid ligands aligns with green chemistry principles, improving the overall sustainability profile of the manufacturing process. This facilitates easier regulatory approval in markets with strict environmental standards and supports corporate sustainability goals by minimizing the ecological footprint of chemical production.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this amino acid-promoted copper catalysis technology. These answers are derived directly from the experimental data and technical specifications provided in the patent documentation, ensuring accuracy and relevance for process development teams evaluating this route for potential adoption.
Q: What are the primary advantages of this CuI-amino acid system over traditional Palladium catalysis?
A: The CuI-amino acid system offers significant cost reductions by replacing expensive palladium catalysts with abundant copper salts. Furthermore, amino acid ligands like L-proline are non-toxic, air-stable, and environmentally benign compared to specialized phosphine ligands often required for Pd chemistry.
Q: Does this methodology tolerate sensitive functional groups commonly found in drug molecules?
A: Yes, the patent demonstrates excellent chemoselectivity and functional group tolerance. The mild reaction conditions (80°C - 95°C) allow for the successful coupling of substrates containing esters, amides, free amines, hydroxyl groups, and even protected amino acid derivatives without degradation.
Q: Is this process suitable for large-scale commercial manufacturing?
A: Absolutely. The use of inexpensive, air-stable reagents (CuI, L-proline sodium salt) and common solvents (DMSO) simplifies procurement and handling. The moderate temperature requirements reduce energy consumption and safety risks associated with high-pressure or high-temperature reactors, facilitating easy scale-up.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Aryl Sulfone Supplier
The technological breakthroughs detailed in patent CN1332944C underscore the immense potential of amino acid-promoted copper catalysis for the efficient production of high-value aryl sulfones. At NINGBO INNO PHARMCHEM, we have integrated this advanced methodology into our process development portfolio, leveraging our extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production. Our commitment to quality is backed by stringent purity specifications and rigorous QC labs, ensuring that every batch of aryl sulfone intermediate meets the exacting standards required by the global pharmaceutical industry. We understand that consistency and purity are paramount, and our state-of-the-art facilities are designed to deliver products with minimal impurity profiles and reliable physicochemical properties.
We invite you to collaborate with us to optimize your supply chain for aryl sulfone intermediates. Our technical team is ready to provide a Customized Cost-Saving Analysis tailored to your specific project needs, demonstrating how this catalytic route can reduce your overall cost of goods. Please contact our technical procurement team today to request specific COA data for our catalog compounds or to discuss route feasibility assessments for your custom synthesis projects. Let us partner with you to bring your next generation of therapeutics to market faster and more economically.
