Advanced CuI-Catalyzed Aryl Sulfone Synthesis for Commercial Scale Pharmaceutical Intermediates
The pharmaceutical and agrochemical industries are constantly seeking robust, cost-effective methodologies for constructing carbon-sulfur bonds, specifically for the synthesis of aryl sulfones which serve as critical scaffolds in numerous bioactive molecules. Patent CN1651408A introduces a groundbreaking advancement in this domain by detailing an amino acid-promoted copper(I) iodide catalyzed coupling reaction between aryl halides and hydrocarbyl sulfinates. This innovation represents a significant departure from traditional palladium-catalyzed systems, offering a pathway that operates under remarkably mild conditions while maintaining high efficiency and broad substrate scope. By utilizing L-proline sodium salt as a ligand, the process achieves reaction temperatures approximately 30°C lower than conventional literature methods, thereby enhancing energy efficiency and minimizing thermal degradation of sensitive intermediates. For R&D directors and process chemists, this patent provides a viable alternative that balances high purity requirements with economic feasibility, ensuring that the production of high-purity aryl sulfones can be achieved without the prohibitive costs associated with noble metal catalysts.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of aryl sulfones has relied heavily on methods that present substantial drawbacks for large-scale manufacturing, particularly regarding cost, environmental impact, and functional group tolerance. Traditional approaches often involve the oxidation of aryl sulfides, which can lead to over-oxidation issues and difficult purification processes, or the use of stoichiometric amounts of organolithium or Grignard reagents that require stringent anhydrous conditions and cryogenic temperatures. Furthermore, palladium-catalyzed cross-coupling reactions, while effective, suffer from the high price of palladium precursors and the necessity for complex, air-sensitive phosphine ligands that increase the overall cost of goods sold. Existing copper-catalyzed methods reported in prior art frequently demand stoichiometric quantities of copper salts or excessively high reaction temperatures that exceed 120°C, leading to potential safety hazards and the decomposition of thermally labile substrates. These limitations create significant bottlenecks in cost reduction in pharmaceutical intermediate manufacturing, as the removal of residual heavy metals and the management of harsh reaction conditions add layers of complexity and expense to the supply chain.
The Novel Approach
In stark contrast to these legacy methods, the technology disclosed in CN1651408A utilizes a catalytic system comprising copper(I) iodide and an inexpensive amino acid derivative, specifically L-proline sodium salt, to facilitate the coupling of aryl halides with alkyl sulfinates. This novel approach allows the reaction to proceed efficiently in polar aprotic solvents like DMSO at moderate temperatures ranging from 80°C to 95°C, significantly lowering the energy input required for commercial scale-up of complex pharmaceutical intermediates. The use of a bidentate amino acid ligand stabilizes the copper center, enabling the activation of less reactive aryl bromides and iodides without the need for exotic additives or inert atmosphere gloveboxes beyond standard nitrogen purging. As illustrated in the reaction scheme below, this methodology successfully converts various substituted aryl iodides into their corresponding sulfone derivatives with excellent yields, demonstrating a level of practicality that is essential for reliable aryl sulfone supplier operations.
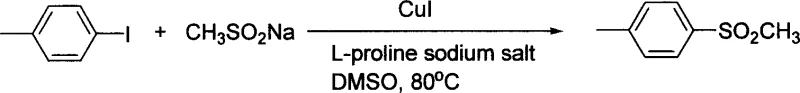
The versatility of this system is further evidenced by its ability to handle diverse electronic environments on the aromatic ring, from electron-donating methoxy groups to electron-withdrawing trifluoromethyl and cyano substituents, ensuring that the synthetic route remains robust across a wide library of potential drug candidates.
Mechanistic Insights into CuI/L-Proline Catalyzed Coupling
The mechanistic underpinning of this transformation involves the formation of an active copper-ligand complex that facilitates the oxidative addition of the aryl halide, followed by transmetallation with the sulfinate salt and subsequent reductive elimination to forge the C-S bond. The L-proline sodium salt acts not only as a ligand to stabilize the copper species in solution but also potentially assists in the deprotonation or activation of the sulfinate nucleophile, creating a highly reactive species capable of displacing the halide under mild thermal conditions. This catalytic cycle is crucial for maintaining low catalyst loading, typically around 10 mol%, which directly correlates to reduced metal contamination in the final product and simplifies the downstream purification workflow. For quality control teams, understanding this mechanism is vital because it explains the high selectivity observed; the mild conditions prevent side reactions such as homocoupling of the aryl halide or desulfonylation, which are common pitfalls in harsher sulfone synthesis protocols. The stability of the copper-amino acid complex in air also means that the reaction setup is less prone to failure due to minor leaks or oxygen ingress, providing a more forgiving process window for plant operators.
Impurity control is inherently built into this methodology due to the specific coordination environment created by the proline ligand, which disfavors the formation of bis-arylated byproducts or sulfone over-oxidation products. The reaction conditions are sufficiently gentle to preserve sensitive functionalities such as free hydroxyl groups, unprotected amines, and ester moieties, which would otherwise require protection-deprotection sequences in traditional syntheses. This functional group tolerance is exemplified by the successful synthesis of complex molecules like chiral amino acid derivatives, where the stereochemical integrity of the alpha-carbon is maintained throughout the coupling process. By avoiding strong bases or nucleophiles that could epimerize chiral centers, this method ensures the production of high-purity aryl sulfones with defined stereochemistry, a critical parameter for API intermediate manufacturing where enantiomeric excess is strictly regulated. The ability to run these reactions in DMSO, a solvent with high boiling point and excellent solvating power, further enhances the homogeneity of the reaction mixture, promoting consistent heat transfer and reaction kinetics across large batches.
How to Synthesize Aryl Sulfones Efficiently
To implement this technology effectively, process chemists should follow a standardized protocol that optimizes the ratio of catalyst to ligand and ensures proper mixing of the heterogeneous sulfinate salt. The general procedure involves charging a reactor with the aryl halide, sodium alkyl sulfinate, CuI, and L-proline sodium salt in DMSO, followed by heating under nitrogen to the specified temperature range of 80°C to 95°C depending on the substrate reactivity. Detailed standard operating procedures regarding workup, including aqueous quenching and extraction with ethyl acetate, are essential to maximize recovery and minimize solvent waste. The following guide outlines the critical steps for executing this synthesis, ensuring that the benefits of this patent are fully realized in a production setting.
- Charge a reaction vessel with aryl halide (iodide or bromide), sodium alkyl sulfinate, CuI catalyst (10 mol%), and L-proline sodium salt ligand.
- Add anhydrous DMSO as the solvent and maintain the reaction mixture under a nitrogen atmosphere to prevent oxidation.
- Heat the mixture to 80-95°C for 24-36 hours, then cool, extract with ethyl acetate, and purify via silica gel chromatography.
Commercial Advantages for Procurement and Supply Chain Teams
From a procurement perspective, the shift from palladium-based systems to this copper-amino acid catalytic system offers profound economic benefits that extend beyond simple raw material costs. The elimination of expensive noble metals and air-sensitive ligands drastically reduces the bill of materials, while the use of commodity chemicals like L-proline and CuI ensures a stable and reliable supply chain不受 geopolitical fluctuations affecting rare earth or precious metal markets. Furthermore, the mild reaction conditions translate to lower energy consumption for heating and cooling cycles, contributing to a smaller carbon footprint and reduced utility costs for the manufacturing facility. For supply chain heads, the robustness of this method means fewer batch failures and more predictable lead times, as the process is less sensitive to minor variations in raw material quality or environmental conditions. This reliability is paramount for maintaining continuous production schedules for high-purity aryl sulfones, ensuring that downstream customers receive their materials on time without quality deviations.
- Cost Reduction in Manufacturing: The replacement of palladium catalysts with copper iodide and L-proline represents a direct and significant reduction in catalyst costs, as copper is orders of magnitude cheaper than palladium and proline is a bulk commodity chemical. Additionally, the mild conditions eliminate the need for specialized high-pressure or cryogenic equipment, allowing the reaction to be run in standard glass-lined or stainless steel reactors commonly found in multipurpose plants. The simplified workup procedure, which avoids complex scavenging steps for heavy metal removal, further reduces processing time and consumable costs associated with purification resins or activated carbon treatments. These factors combine to lower the overall cost of goods sold, making the final aryl sulfone intermediates more competitive in the global market without compromising on quality standards.
- Enhanced Supply Chain Reliability: Sourcing high-purity palladium catalysts and specialized phosphine ligands can often be a bottleneck due to limited suppliers and long lead times, whereas CuI and L-proline sodium salt are widely available from multiple chemical vendors globally. This diversification of the supply base mitigates the risk of production stoppages caused by raw material shortages, ensuring business continuity even during market disruptions. The stability of the reagents in air also simplifies logistics and storage requirements, as they do not need cold chain shipping or inert atmosphere containment, reducing warehousing costs and handling risks. Consequently, manufacturers can maintain leaner inventory levels while still guaranteeing the availability of critical intermediates for their clients.
- Scalability and Environmental Compliance: The use of DMSO as a solvent and the absence of volatile organic chlorides or highly toxic reagents align well with modern green chemistry principles and environmental regulations. Scaling this reaction from gram to ton scale is straightforward because the exotherm is manageable at 80-95°C, and the viscosity of the reaction mixture remains within operable limits for standard agitation systems. Waste treatment is simplified since the aqueous waste streams contain primarily inorganic salts and copper residues that can be treated with standard effluent protocols, avoiding the generation of persistent organic pollutants. This environmental compatibility facilitates easier permitting for new production lines and supports corporate sustainability goals, making the technology attractive for long-term investment.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this amino acid-promoted coupling technology, derived directly from the experimental data and scope defined in the patent literature. Understanding these nuances helps stakeholders evaluate the fit of this methodology for their specific project pipelines and manufacturing capabilities. The answers provided reflect the proven capabilities of the system as demonstrated in the examples, offering clarity on substrate scope and operational parameters.
Q: What are the advantages of using L-proline sodium salt over traditional phosphine ligands?
A: L-proline sodium salt is significantly cheaper, air-stable, and non-toxic compared to expensive phosphine ligands, reducing overall production costs and safety hazards.
Q: Can this method tolerate sensitive functional groups like hydroxyl or amino groups?
A: Yes, the mild reaction conditions (80-95°C) allow for excellent tolerance of functional groups such as hydroxyl, amino, ester, and cyano groups without protection.
Q: Is this process scalable for industrial manufacturing?
A: Absolutely. The use of inexpensive catalysts, common solvents like DMSO, and moderate temperatures makes this process highly suitable for large-scale commercial production.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Aryl Sulfone Supplier
At NINGBO INNO PHARMCHEM, we recognize the strategic value of adopting innovative synthetic routes like the CuI/L-proline catalyzed coupling described in CN1651408A to enhance our portfolio of pharmaceutical intermediates. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory discovery to industrial reality is seamless and efficient. We are committed to delivering high-purity aryl sulfones that meet stringent purity specifications, leveraging our rigorous QC labs to verify every batch against the highest industry standards. By integrating this cost-effective and environmentally friendly technology into our manufacturing processes, we can offer our partners a superior balance of quality, speed, and value.
We invite you to engage with our technical procurement team to discuss how this advanced synthesis method can be tailored to your specific molecular targets. Request a Customized Cost-Saving Analysis today to understand the potential economic impact of switching to this copper-catalyzed route for your supply chain. We are ready to provide specific COA data and route feasibility assessments to support your R&D and sourcing decisions, ensuring that your projects move forward with the confidence of a reliable partner dedicated to your success.
