Advanced Manufacturing of Tegafur: A Mild, High-Yield Phase Transfer Catalysis Route for Global Supply Chains
The pharmaceutical industry continuously seeks robust synthetic routes for critical antineoplastic agents that balance high purity with operational safety. Patent CN110655507A discloses a groundbreaking preparation method for Tegafur, a widely used prodrug of 5-fluorouracil, which addresses the longstanding challenges of toxicity and harsh reaction conditions associated with legacy manufacturing processes. This innovative approach utilizes a biphasic system comprising 5-fluorouracil and 2-benzoyloxy tetrahydrofuran, mediated by an inorganic base and a phase transfer catalyst under remarkably mild thermal conditions. Unlike traditional methods that rely on hazardous heavy metals or extreme temperatures, this technology achieves exceptional chemical purity exceeding 99.7% with single impurities controlled below 0.1%, representing a significant leap forward in process chemistry for oncology intermediates. The structural integrity of the final product is paramount for therapeutic efficacy, as illustrated by the precise molecular architecture of the target compound.
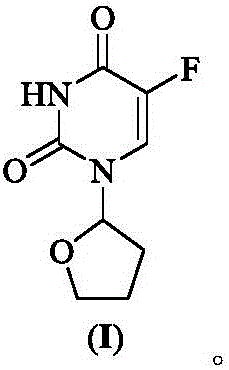
Furthermore, the strategic selection of 2-benzoyloxy tetrahydrofuran as the glycosyl donor provides inherent steric control that drives the reaction towards the desired N1-substitution, effectively minimizing the formation of regioisomeric byproducts that are difficult to remove. This patent not only offers a greener alternative by eliminating environmental pollutants but also simplifies the downstream purification workflow, thereby enhancing the overall economic viability of producing this essential cancer therapy agent on a global scale.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historical manufacturing routes for Tegafur have been plagued by severe operational hazards and inefficiencies that hinder scalable production. Early industrial methods, such as those disclosed in CN103159746A, required reacting 5-fluorouracil with 2,3-dihydrofuran under high temperature and high pressure conditions ranging from 150°C to 180°C, necessitating expensive specialized reactor equipment and posing significant safety risks. Alternative strategies involving pre-derivatization of 5-fluorouracil often relied on toxic mercury or silver salts, which introduced heavy metal contamination risks requiring complex and costly removal protocols to meet regulatory standards. Other approaches utilizing 2-chlorotetrahydrofuran suffered from the extreme instability of the starting material, forcing reactions to be conducted at cryogenic temperatures between -60°C and 10°C, which drastically increases energy consumption and operational complexity. Additionally, methods employing thiophosgene generated corrosive hydrogen chloride gas and exhibited low conversion rates, rendering them unsuitable for modern, environmentally conscious manufacturing facilities.
The Novel Approach
The methodology presented in CN110655507A fundamentally transforms the synthesis landscape by enabling the reaction to proceed at ambient pressure and mild temperatures between 30°C and 40°C. By employing 2-benzoyloxy tetrahydrofuran, the process leverages the steric bulk of the benzoyl group to naturally direct the nucleophilic attack to the N1 nitrogen atom of the uracil ring, thereby achieving high regioselectivity without the need for complex protecting group strategies. The use of inexpensive inorganic bases like sodium acetate or potassium carbonate, combined with efficient phase transfer catalysts such as tetrabutylammonium iodide, creates a highly active yet controlled reaction environment in a water-organic biphasic system. This eliminates the need for toxic heavy metal catalysts entirely, resulting in a cleaner reaction profile that simplifies workup procedures and significantly reduces the environmental footprint of the manufacturing process while maintaining yields consistently above 75%.
Mechanistic Insights into Phase Transfer Catalyzed N-Glycosylation
The core of this synthetic breakthrough lies in the sophisticated interplay between the phase transfer catalyst and the steric properties of the glycosyl donor within a biphasic medium. In this system, the inorganic base deprotonates the 5-fluorouracil in the aqueous phase to generate a nucleophilic anion, which is then transported into the organic phase by the quaternary ammonium salt catalyst. Once in the organic phase, the 5-fluorouracil anion encounters the 2-benzoyloxy tetrahydrofuran, where the bulky benzoyl moiety at the 2-position exerts a profound steric influence. This steric hindrance effectively blocks access to the N3 position of the pyrimidine ring, forcing the nucleophilic substitution to occur exclusively at the N1 position, which is the therapeutically active configuration. This spatial selectivity is critical because N3-substituted isomers are not only inactive but also constitute difficult-to-remove impurities that can compromise the safety profile of the final API.
Impurity control is further enhanced by the stability of the 2-benzoyloxy tetrahydrofuran reagent compared to its chloro- or alkoxy- counterparts, which are prone to decomposition or side reactions under varying conditions. The mild thermal window of 30°C to 40°C prevents thermal degradation of the sensitive fluorouracil ring, a common issue in high-temperature processes, ensuring that the maximum single impurity remains strictly below 0.1%. The biphasic nature of the reaction also facilitates the continuous removal of the product into the organic layer or keeps byproducts in the aqueous phase, depending on solubility profiles, which streamlines the isolation process. Consequently, the resulting crude product exhibits a purity profile exceeding 99.7%, drastically reducing the burden on crystallization and chromatographic purification steps typically required to meet pharmacopeial standards for oncology drugs.
How to Synthesize Tegafur Efficiently
The synthesis of Tegafur via this patented route involves a straightforward sequence of mixing aqueous and organic phases followed by mild heating and standard workup procedures. The process begins with the dissolution of 5-fluorouracil, an inorganic base such as sodium acetate, and a phase transfer catalyst like tetrabutylammonium iodide in purified water to create a homogeneous aqueous solution. To this mixture, a solution of 2-benzoyloxy tetrahydrofuran in a water-immiscible organic solvent such as dichloromethane is added, establishing the biphasic reaction system. The mixture is then heated to a controlled temperature range of 30°C to 40°C and stirred for a duration of 2 to 6 hours until conversion is complete. Following the reaction, the organic layer is separated, washed with water and brine to remove inorganic salts, dried over anhydrous sodium sulfate, and concentrated to yield the high-purity product. For detailed standardized operating procedures and specific parameter optimization, refer to the guide below.
- Dissolve 5-fluorouracil, inorganic base (e.g., sodium acetate), and phase transfer catalyst in purified water to form the aqueous phase.
- Add the water-insoluble organic solution of 2-benzoyloxy tetrahydrofuran to the reaction system and heat mildly to 30-40°C.
- Upon completion, separate the organic layer, wash sequentially with water and brine, dry, and concentrate to obtain crude Tegafur.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this novel synthesis route offers transformative benefits regarding cost structure and operational reliability. The elimination of expensive and toxic heavy metal catalysts such as mercury and silver salts removes a significant cost center associated with both raw material acquisition and hazardous waste disposal. Furthermore, the avoidance of extreme reaction conditions, such as high pressure or cryogenic temperatures, allows for the utilization of standard glass-lined or stainless steel reactors that are commonly available in most multipurpose chemical plants, thereby reducing capital expenditure requirements for new production lines. The stability of the key starting material, 2-benzoyloxy tetrahydrofuran, ensures consistent supply chain continuity, unlike unstable chloro-derivatives that require specialized cold-chain logistics and have short shelf lives.
- Cost Reduction in Manufacturing: The process achieves substantial cost savings by replacing precious metal catalysts with abundant inorganic bases and simple quaternary ammonium salts, which are available at a fraction of the cost. The simplified downstream processing, driven by the high selectivity of the reaction, reduces the consumption of solvents and adsorbents typically needed for extensive purification, leading to a leaner cost of goods sold. Additionally, the mild reaction conditions result in lower energy consumption for heating and cooling compared to legacy high-temperature or cryogenic processes, contributing to a more sustainable and economically efficient production model.
- Enhanced Supply Chain Reliability: By utilizing chemically stable intermediates that do not require specialized storage conditions, the risk of raw material degradation during transit is minimized, ensuring a steady flow of inputs for continuous manufacturing. The robustness of the reaction against minor fluctuations in temperature or mixing rates provides a wider operating window, reducing the likelihood of batch failures and ensuring consistent on-time delivery of the final API intermediate to downstream formulation partners. This reliability is crucial for maintaining the inventory levels required for Just-In-Time manufacturing strategies in the pharmaceutical sector.
- Scalability and Environmental Compliance: The absence of toxic heavy metals and corrosive gases simplifies the environmental permitting process and reduces the regulatory burden associated with effluent treatment. The process generates significantly less hazardous waste, aligning with increasingly stringent global environmental regulations and corporate sustainability goals. The scalability is proven by the use of standard unit operations such as liquid-liquid extraction and vacuum distillation, allowing for seamless transition from pilot plant batches to multi-ton commercial production without the need for complex engineering redesigns.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this advanced Tegafur synthesis technology. These insights are derived directly from the experimental data and technical specifications outlined in the patent documentation, providing clarity on process capabilities and quality outcomes. Understanding these details is essential for technical teams evaluating the feasibility of integrating this route into existing manufacturing portfolios.
Q: How does this method improve regioselectivity compared to traditional alkylation?
A: The use of 2-benzoyloxy tetrahydrofuran introduces significant steric hindrance that selectively favors nucleophilic attack at the N1 position of 5-fluorouracil, effectively suppressing the formation of N3-substituted byproducts and ensuring high regioselectivity.
Q: What are the environmental advantages of avoiding mercury or silver salts?
A: By eliminating toxic heavy metal catalysts like mercury or silver salts, the process removes the need for complex and costly heavy metal removal steps, significantly reducing hazardous waste generation and operator exposure risks.
Q: Is this process suitable for large-scale commercial production?
A: Yes, the reaction operates at atmospheric pressure and mild temperatures (30-40°C) using stable, commercially available raw materials, making it highly adaptable for multi-ton scale manufacturing without requiring specialized high-pressure equipment.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Tegafur Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of delivering high-quality oncology intermediates that meet the rigorous demands of modern drug development. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory innovation to industrial reality is seamless and efficient. We are committed to maintaining stringent purity specifications through our rigorous QC labs, guaranteeing that every batch of Tegafur we produce adheres to the highest international standards for safety and efficacy. Our facility is equipped to handle the specific biphasic reaction conditions described in this patent, leveraging our expertise in phase transfer catalysis to maximize yield and minimize impurities.
We invite global partners to engage with our technical procurement team to discuss how this optimized synthesis route can enhance your supply chain resilience. By requesting a Customized Cost-Saving Analysis, you can gain a clear understanding of the economic benefits specific to your volume requirements. We encourage you to contact us today to obtain specific COA data and route feasibility assessments, allowing you to make informed decisions that drive value and efficiency in your pharmaceutical manufacturing operations.
