Advanced Metal-Free Synthesis of Tegafur: Scalable Production for Global Pharmaceutical Supply Chains
Introduction to Patent CN111925361B
The pharmaceutical industry is constantly seeking more efficient, safer, and environmentally benign pathways for the synthesis of critical oncology agents. Patent CN111925361B introduces a groundbreaking preparation method for Tegafur, a vital prodrug of 5-fluorouracil used extensively in the treatment of gastrointestinal and breast cancers. This intellectual property represents a significant departure from traditional synthetic routes by employing a metal-free catalytic system based on tetraalkylammonium iodide salts. Unlike conventional processes that rely on harsh thermal conditions or toxic heavy metal catalysts, this innovation utilizes a mild organic oxidation cycle driven by tert-butyl hydroperoxide and a weak base. For global procurement and R&D teams, this patent signals a potential paradigm shift in how high-purity pharmaceutical intermediates are manufactured, offering a route that is not only chemically elegant but also industrially robust and economically viable for large-scale production.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industrial synthesis of Tegafur has been plagued by significant technical and economic hurdles that complicate supply chain management and increase production costs. Traditional methods often necessitate the reaction of 5-fluorouracil with 2,3-dihydrofuran at extremely high temperatures ranging from 150°C to 180°C, requiring specialized high-pressure equipment that increases capital expenditure and operational risk. Alternative strategies involving Lewis acid catalysis frequently suffer from low chemical stability of reagents and the generation of complex by-product profiles that are difficult to separate. Furthermore, recent approaches utilizing transition metal catalysts such as copper, nickel, or palladium acetates introduce a critical liability: the risk of heavy metal contamination. Removing these metal residues to meet stringent International Council for Harmonisation (ICH) guidelines requires additional purification steps, such as chromatography or specialized scavenging resins, which drastically reduce overall yield and extend production lead times.
The Novel Approach
The methodology disclosed in CN111925361B effectively dismantles these barriers by introducing a catalytic system that operates under remarkably mild and safe conditions. By utilizing tetrahydrofuran as both a solvent and a reactant alongside an organic ammonium iodide catalyst, the process eliminates the need for high-pressure reactors and extreme thermal inputs. The reaction proceeds efficiently at temperatures between 50°C and 100°C, significantly lowering energy consumption and enhancing operational safety. Crucially, the absence of transition metals means the final product is free from heavy metal impurities, simplifying the purification workflow and ensuring compliance with global regulatory standards for API intermediates. This approach transforms the synthesis from a hazardous, high-cost operation into a streamlined, green chemistry process that is highly attractive for commercial scale-up.
Mechanistic Insights into Organic Ammonium Iodide Catalysis
The core innovation of this patent lies in the unique mechanistic role played by the tetraalkylammonium iodide salt, which acts as a redox mediator rather than a simple phase transfer catalyst. In the presence of tert-butyl hydroperoxide (TBHP), the iodide anion is oxidized to generate a reactive ammonium hypoiodite intermediate in situ. This transient species is capable of oxidizing the tetrahydrofuran solvent to form a highly reactive oxonium ion intermediate. This electrophilic species then undergoes nucleophilic attack by the 5-fluorouracil substrate, facilitating the formation of the N-glycosidic bond essential for Tegafur structure. This mechanism bypasses the need for aggressive Lewis acids or high-energy thermal activation, allowing the reaction to proceed with high selectivity under weakly basic conditions.
From an impurity control perspective, this mechanism offers distinct advantages for R&D directors focused on product quality. Because the catalytic cycle relies on organic components that are either volatile or easily removed during aqueous workup, the risk of persistent catalyst residues is virtually eliminated. The use of weak bases like potassium carbonate or cesium carbonate further prevents the degradation of the sensitive fluorouracil ring, which can occur under the strong alkaline conditions required by other methods. This results in a cleaner reaction profile with fewer side products, directly translating to higher crude purity and reduced burden on downstream crystallization processes.
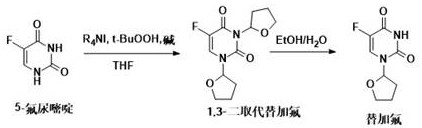
How to Synthesize Tegafur Efficiently
Implementing this novel synthetic route requires precise control over stoichiometry and reaction parameters to maximize yield and purity. The process generally involves a two-stage sequence: first, the oxidative coupling of 5-fluorouracil with tetrahydrofuran to form a protected intermediate, followed by a deprotection step to yield the final active pharmaceutical ingredient. The detailed standardized synthesis steps, including specific molar ratios, temperature gradients, and workup procedures derived from the patent examples, are outlined below to assist technical teams in evaluating process feasibility.
- Dissolve 5-fluorouracil in tetrahydrofuran and add a tetraalkylammonium iodide catalyst, a weak base such as potassium carbonate, and an aqueous tert-butyl hydroperoxide solution.
- Heat the reaction mixture to 50-100°C to generate the 1,3-disubstituted intermediate, then quench with water and extract the organic phase.
- Treat the crude intermediate with an ethanol-water solution at 40-70°C to remove the protecting group, followed by recrystallization to obtain high-purity Tegafur.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this metal-free synthesis route offers compelling strategic advantages that extend beyond simple chemistry. The shift away from expensive transition metal catalysts and high-pressure infrastructure directly impacts the cost structure of manufacturing, while the use of commodity chemicals enhances supply security. By simplifying the purification train and reducing energy intensity, manufacturers can achieve significant operational efficiencies that improve margin potential in a competitive generic drug market.
- Cost Reduction in Manufacturing: The elimination of precious metal catalysts such as palladium or copper removes a major cost driver from the bill of materials. Furthermore, avoiding the need for specialized metal scavenging resins and complex filtration steps significantly reduces consumable costs and waste disposal fees. The milder reaction conditions also translate to lower energy consumption, as the process does not require maintaining high temperatures or pressures for extended periods, leading to substantial utility savings over the lifecycle of the product.
- Enhanced Supply Chain Reliability: The raw materials required for this synthesis, including 5-fluorouracil, tetrahydrofuran, and tert-butyl hydroperoxide, are widely available commodity chemicals with stable global supply chains. Unlike specialized organometallic catalysts which may face supply bottlenecks or long lead times, these reagents can be sourced from multiple vendors, reducing the risk of production stoppages. The robustness of the reaction to moisture and oxygen further simplifies logistics, as it does not require the rigorous inert atmosphere handling typical of sensitive metal-catalyzed processes.
- Scalability and Environmental Compliance: The safety profile of this process is markedly improved by operating at atmospheric pressure and moderate temperatures, making it inherently safer for scale-up from pilot plant to commercial tonnage. The absence of toxic heavy metals simplifies wastewater treatment and aligns with increasingly stringent environmental regulations regarding metal discharge. This green chemistry profile not only reduces regulatory compliance costs but also enhances the sustainability credentials of the manufacturing site, a key factor for modern pharmaceutical partnerships.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this patented synthesis method. These answers are derived directly from the experimental data and technical disclosures within the patent documentation to provide clarity on process capabilities and limitations.
Q: Why is the metal-free catalyst system superior for Tegafur production?
A: Traditional methods often utilize transition metal catalysts like copper or palladium, which pose risks of heavy metal residue in the final API. This novel method uses organic ammonium salts, eliminating the need for complex and costly metal scavenging steps, ensuring compliance with strict pharmaceutical impurity guidelines.
Q: What are the reaction conditions required for this synthesis?
A: The process operates under significantly milder conditions compared to prior art, requiring temperatures between 50-100°C rather than the harsh 150-180°C and high pressure previously needed. It also tolerates the presence of water, removing the need for strictly anhydrous environments.
Q: How does this method impact the overall yield and purity?
A: Experimental data indicates that this method achieves high conversion rates with yields reaching up to 77% and HPLC purity exceeding 99%. The mild conditions minimize side reactions and degradation, resulting in a cleaner crude product that simplifies downstream purification.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Tegafur Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of this metal-free catalytic technology for the production of high-value oncology intermediates. As a dedicated CDMO partner, we possess the technical expertise to rapidly adapt and optimize this novel pathway for your specific production needs. Our facilities are equipped to handle complex organic syntheses with extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that we can meet your volume requirements without compromising on quality. We maintain stringent purity specifications across all our operations, supported by rigorous QC labs that utilize advanced analytical techniques to verify the absence of metal residues and ensure batch-to-batch consistency.
We invite you to collaborate with us to leverage this innovative synthesis route for your Tegafur supply chain. Our technical team is prepared to conduct a Customized Cost-Saving Analysis tailored to your current manufacturing setup, demonstrating exactly how this method can improve your bottom line. Please contact our technical procurement team today to request specific COA data from our pilot runs and discuss route feasibility assessments for your upcoming projects.
