Advanced Ligand-Free Palladium Catalysis for Commercial Cyanopyridine Manufacturing
Advanced Ligand-Free Palladium Catalysis for Commercial Cyanopyridine Manufacturing
The pharmaceutical and agrochemical industries are constantly seeking more efficient and sustainable pathways for synthesizing critical heterocyclic building blocks. Patent CN101648909B introduces a groundbreaking method for preparing cyanopyridine derivatives using chloropyridine under the catalysis of ligand-free palladium. This technology represents a significant departure from traditional cyanation protocols, addressing long-standing issues related to toxicity, cost, and operational complexity. By utilizing potassium ferrocyanide as a safe cyanide source and eliminating the need for expensive phosphine ligands, this process offers a robust solution for the commercial scale-up of complex heterocyclic intermediates. The innovation lies not just in the chemical transformation itself, but in the holistic improvement of the manufacturing profile, making it an attractive option for reliable cyanopyridine supplier networks aiming to optimize their production capabilities.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of nitrile-functionalized pyridines has relied heavily on the Rosenmund-von Braun reaction or early palladium-catalyzed methods that suffer from significant drawbacks. The classical approach typically involves the reaction of aryl halides with cuprous cyanide at elevated temperatures, a process notorious for requiring harsh conditions and generating substantial amounts of toxic copper waste that is difficult to remediate. Furthermore, while palladium-catalyzed cyanation improved functional group tolerance, early iterations often necessitated the use of highly toxic cyanide sources such as sodium cyanide or potassium cyanide, posing severe safety risks to personnel and requiring specialized containment infrastructure. Additionally, many modern catalytic systems depend on bulky, electron-rich phosphine ligands to stabilize the palladium center; these ligands are frequently expensive, sensitive to air and moisture, and challenging to remove from the final product, leading to increased purification costs and potential metal contamination issues in the final high-purity cyanopyridine API intermediates.
The Novel Approach
In stark contrast, the methodology disclosed in patent CN101648909B leverages a ligand-free palladium system that fundamentally simplifies the catalytic cycle while enhancing safety and economic viability. By employing potassium ferrocyanide trihydrate as the cyanating agent, the process completely avoids the handling of free cyanide salts, thereby mitigating the acute toxicity hazards associated with traditional reagents. The absence of phosphine ligands is particularly transformative, as it removes a major cost driver and eliminates a complex variable in the reaction optimization landscape. This ligand-free protocol operates effectively in common polar aprotic solvents like N,N-dimethylacetamide or dimethyl sulfoxide, utilizing simple inorganic bases such as potassium carbonate. The result is a streamlined workflow that maintains high conversion rates and yields comparable to ligand-dependent systems, yet with a drastically reduced environmental footprint and lower overall operational expenditure, directly supporting cost reduction in pharmaceutical intermediates manufacturing.
Mechanistic Insights into Ligand-Free Palladium Cyanation
The success of this ligand-free system can be attributed to the unique reactivity of the palladium species in the presence of the ferrocyanide anion. In the absence of stabilizing phosphine ligands, the palladium acetate precursor likely generates highly active, low-coordinate palladium(0) species in situ, which are exceptionally proficient at undergoing oxidative addition into the carbon-chlorine bond of the chloropyridine substrate. This step is often the rate-determining factor in the cyanation of electron-deficient heterocycles, but the high electrophilicity of the ligand-free metal center facilitates this activation even at moderate temperatures ranging from 40 to 200 degrees Celsius. Following oxidative addition, the ferrocyanide ion acts as a transmetallating agent, transferring a cyanide group to the palladium center. The subsequent reductive elimination releases the desired cyanopyridine product and regenerates the active catalyst. This mechanism is remarkably efficient because the ferrocyanide anion itself may play a role in stabilizing the palladium species, preventing the aggregation of palladium black which typically deactivates ligand-free catalysts.
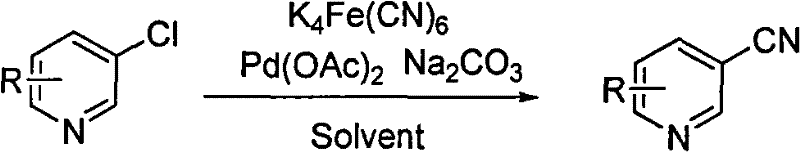
From an impurity control perspective, this mechanistic pathway offers distinct advantages for regulatory compliance and product quality. Traditional methods using alkali cyanides often lead to the formation of hydrolysis byproducts or over-cyanated species due to the high concentration of free cyanide ions in the solution. In the ferrocyanide-mediated process, the cyanide ions are released in a controlled manner through the coordination sphere of the iron complex, effectively buffering the concentration of reactive nucleophile available at any given time. This controlled release minimizes side reactions such as the hydrolysis of the nitrile group to the corresponding amide or acid, which are common impurities in nitrile synthesis. Furthermore, the lack of phosphine ligands means there are no phosphine oxide byproducts or phosphine-palladium complexes to separate, simplifying the downstream purification process and ensuring that the final material meets the stringent purity specifications required for reducing lead time for high-purity cyanopyridine derivatives intended for clinical applications.
How to Synthesize Cyanopyridine Efficiently
The practical implementation of this synthesis route is designed for ease of execution in both laboratory and pilot plant settings. The procedure involves charging a reactor with the chloropyridine substrate, potassium ferrocyanide, potassium carbonate, and a catalytic amount of palladium acetate in a suitable solvent such as N-methyl-2-pyrrolidone or dimethylformamide. The reaction is conducted under an inert atmosphere, typically nitrogen or argon, to prevent oxidation of the active palladium species, and heated to the optimal temperature window identified in the patent examples.
- Prepare the reaction mixture by dissolving chloropyridine, potassium ferrocyanide, potassium carbonate, and palladium acetate in a polar aprotic solvent such as DMF or NMP under inert gas protection.
- Heat the reaction mixture to a temperature range of 40-200°C, preferably between 60-120°C, and maintain stirring for 2 to 20 hours to ensure complete conversion.
- Upon completion, cool the mixture, dilute with ethyl acetate, filter to remove inorganic salts, and wash the organic phase with water and dilute ammonia before drying and concentrating.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this ligand-free technology translates into tangible strategic benefits that extend beyond simple chemistry. The primary advantage lies in the radical simplification of the raw material portfolio; by replacing expensive, air-sensitive ligands and hazardous cyanide salts with stable, commodity-grade chemicals, the supply chain becomes significantly more resilient and less prone to disruption. The use of potassium ferrocyanide, a widely available and non-regulated chemical in many jurisdictions compared to restricted cyanide salts, streamlines logistics and reduces the regulatory burden associated with transportation and storage. This shift not only lowers the direct cost of goods sold but also reduces the indirect costs associated with safety compliance, waste disposal, and insurance, creating a compelling economic case for switching to this novel manufacturing route.
- Cost Reduction in Manufacturing: The elimination of proprietary phosphine ligands represents a direct and substantial saving in raw material costs, as these ligands often constitute a significant portion of the catalyst system expense in traditional cross-coupling reactions. Moreover, the simplified workup procedure, which avoids the need for complex chromatographic removal of ligand residues, reduces solvent consumption and processing time, leading to lower utility and labor costs per kilogram of product. The ability to use lower catalyst loadings while maintaining high turnover numbers further enhances the economic efficiency of the process, making it highly competitive for large-volume production runs where margin compression is a constant challenge.
- Enhanced Supply Chain Reliability: Sourcing high-quality phosphine ligands can sometimes be a bottleneck due to limited global suppliers and long lead times, whereas potassium ferrocyanide and palladium acetate are commoditized chemicals with robust and diversified supply chains. This diversification mitigates the risk of single-source dependency and ensures continuous production capability even during market fluctuations. Additionally, the stability of the reagents allows for longer shelf life and easier inventory management, reducing the risk of material degradation and waste, which is crucial for maintaining a lean and responsive supply chain capable of meeting fluctuating demand from downstream pharmaceutical customers.
- Scalability and Environmental Compliance: The process is inherently scalable due to its reliance on simple thermal activation and standard heterogeneous filtration for catalyst removal, avoiding the complexities of handling pyrophoric or highly toxic reagents on a multi-ton scale. From an environmental standpoint, the avoidance of heavy metal cyanide waste and phosphine oxides significantly reduces the load on wastewater treatment facilities and lowers the cost of hazardous waste disposal. This alignment with green chemistry principles not only satisfies increasingly stringent environmental regulations but also enhances the corporate sustainability profile, which is becoming a key differentiator in supplier selection processes for major multinational corporations.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this ligand-free palladium catalyzed cyanation technology. These insights are derived directly from the experimental data and beneficial effects described in the patent literature, providing a clear understanding of the process capabilities.
Q: Why is ligand-free palladium catalysis preferred for cyanopyridine synthesis?
A: Ligand-free palladium catalysis eliminates the need for expensive and air-sensitive phosphine ligands, significantly reducing raw material costs and simplifying the purification process by avoiding ligand-derived impurities.
Q: What are the safety advantages of using potassium ferrocyanide over traditional cyanating agents?
A: Potassium ferrocyanide is a non-toxic, stable solid that serves as a safe source of cyanide ions, unlike highly toxic sodium cyanide or potassium cyanide, thereby drastically reducing operational hazards and waste treatment burdens.
Q: Can this method be scaled for industrial production of agrochemical intermediates?
A: Yes, the method utilizes accessible raw materials and simple operating conditions without sensitive ligands, making it highly suitable for large-scale commercial production of complex heterocyclic intermediates used in agrochemicals.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Cyanopyridine Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of ligand-free catalytic systems in modernizing the production of critical pharmaceutical intermediates. Our technical team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory discovery to industrial reality is seamless and efficient. We are committed to delivering stringent purity specifications through our rigorous QC labs, guaranteeing that every batch of cyanopyridine derivative meets the exacting standards required for drug substance manufacturing. Our facility is equipped to handle the specific solvent systems and thermal conditions required by this patent, allowing us to offer this advanced technology as a value-added service to our global partners.
We invite you to collaborate with us to leverage this cost-effective and environmentally friendly synthesis route for your next project. Contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We are ready to provide specific COA data from our pilot trials and comprehensive route feasibility assessments to demonstrate how this innovative method can optimize your supply chain and enhance your product competitiveness in the global market.
