Advanced Catalytic Synthesis of 4-Trifluoromethyl Nicotinic Acid for Industrial Flonicamid Production
Advanced Catalytic Synthesis of 4-Trifluoromethyl Nicotinic Acid for Industrial Flonicamid Production
The global demand for high-efficiency, low-toxicity insecticides has placed 4-trifluoromethyl nicotinic acid at the center of agrochemical supply chains, serving as the critical precursor for Flonicamid. As detailed in the recent patent CN111574440A, a groundbreaking preparation method has emerged that fundamentally shifts the manufacturing paradigm from hazardous, multi-step halogenation processes to a streamlined catalytic oxidation and cyclization route. This technological leap addresses long-standing bottlenecks in purity and cost, offering a robust solution for the commercial scale-up of complex agrochemical intermediates. By leveraging commodity feedstocks like methyl acrylate and avoiding cryogenic conditions, this innovation not only enhances process safety but also significantly lowers the barrier to entry for high-volume production, positioning it as a vital asset for manufacturers aiming to secure a competitive edge in the pesticide market.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of 4-trifluoromethyl nicotinic acid has been plagued by severe operational hazards and economic inefficiencies, primarily relying on routes that necessitate extreme conditions or toxic reagents. One prevalent legacy method involves the direct carboxylation of 4-trifluoromethylpyridine using Lithium Diisopropylamide (LDA) and carbon dioxide, a process that demands strict anhydrous environments and cryogenic temperatures as low as -75°C, imposing massive energy burdens and requiring specialized reactor infrastructure. Furthermore, alternative pathways often utilize 2,6-dichloro-4-trifluoromethylpyridine as a starting material, which subsequently requires chlorination with phosphorus oxychloride (POCl3) followed by catalytic hydrogenolysis to remove chlorine atoms. 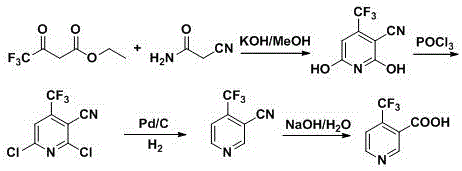 This reliance on POCl3 generates substantial amounts of acidic wastewater and hazardous phosphorous waste, creating significant environmental compliance challenges and disposal costs. Additionally, the hydrogenolysis step carries a inherent risk of over-reducing the sensitive pyridine ring or the trifluoromethyl group, leading to difficult-to-separate impurities that compromise the quality of the final high-purity agrochemical intermediate.
This reliance on POCl3 generates substantial amounts of acidic wastewater and hazardous phosphorous waste, creating significant environmental compliance challenges and disposal costs. Additionally, the hydrogenolysis step carries a inherent risk of over-reducing the sensitive pyridine ring or the trifluoromethyl group, leading to difficult-to-separate impurities that compromise the quality of the final high-purity agrochemical intermediate.
The Novel Approach
In stark contrast, the novel methodology disclosed in patent CN111574440A introduces a constructive strategy that builds the pyridine ring from acyclic precursors under mild, industrially friendly conditions. This approach bypasses the need for pre-functionalized halogenated pyridines entirely, instead initiating the synthesis with the catalytic oxidation of methyl acrylate to generate methyl 3-oxopropionate in situ. This intermediate then undergoes a condensation reaction with 4-amino-1,1,1-trifluoro-3-buten-2-one, followed by a base-mediated cyclization and hydrolysis to yield the target acid.  By operating at temperatures ranging from 25°C to 90°C and utilizing common solvents like toluene or ethyl acetate, this process eliminates the need for expensive cryogenic cooling and exotic reagents. The result is a synthesis pathway that is not only chemically more elegant but also economically superior, offering a viable route for cost reduction in agrochemical intermediate manufacturing by simplifying the unit operations and minimizing waste generation.
By operating at temperatures ranging from 25°C to 90°C and utilizing common solvents like toluene or ethyl acetate, this process eliminates the need for expensive cryogenic cooling and exotic reagents. The result is a synthesis pathway that is not only chemically more elegant but also economically superior, offering a viable route for cost reduction in agrochemical intermediate manufacturing by simplifying the unit operations and minimizing waste generation.
Mechanistic Insights into Catalytic Oxidation and Cyclization
The core of this technological advancement lies in the efficient transition metal-catalyzed oxidation of methyl acrylate, which serves as the carbon backbone for the nicotinic acid structure. In the first stage, methyl acrylate reacts in the presence of a catalyst system—preferably comprising palladium chloride and copper chloride or similar transition metal combinations—along with an oxidant such as oxygen. This catalytic cycle facilitates the insertion of oxygen into the allylic position, converting the alkene into the crucial beta-keto ester, methyl 3-oxopropionate, without isolating this unstable intermediate. The reaction kinetics are carefully controlled by maintaining specific molar ratios of methyl acrylate to catalyst and oxidant, typically around 1:0.01-0.05:1-1.5, ensuring high conversion rates while minimizing side reactions such as polymerization or over-oxidation. This in-situ generation is pivotal as it prevents the degradation of the reactive keto-ester, thereby maximizing the atom economy of the initial step and setting a clean foundation for the subsequent condensation.
Following the formation of the keto-ester, the mechanism proceeds through a condensation with 4-amino-1,1,1-trifluoro-3-buten-2-one to form the linear precursor N-(2-methoxycarbonylvinyl)-4,4,4-trifluoro-3-one-1-butenamine. The final transformation involves a base-promoted intramolecular cyclization where a base, such as sodium methoxide or sodium hydroxide, abstracts a proton to initiate ring closure, forming the pyridine nucleus. Simultaneously, the ester moiety undergoes hydrolysis to yield the free carboxylic acid. This tandem cyclization-hydrolysis is conducted under mild thermal conditions (25-90°C), which is critical for preserving the integrity of the trifluoromethyl group. Unlike harsh acidic or reductive conditions found in older methods, this basic environment effectively suppresses the formation of defluorinated byproducts and ensures a clean impurity profile, which is essential for meeting the stringent specifications required by reliable agrochemical intermediate suppliers serving the global pesticide industry.
How to Synthesize 4-Trifluoromethyl Nicotinic Acid Efficiently
Implementing this novel synthesis route requires precise control over reaction parameters to maximize yield and purity, particularly during the catalytic oxidation and the final cyclization steps. The process is designed to be operationally simple, utilizing standard laboratory or plant equipment without the need for specialized high-pressure or cryogenic setups. Operators must carefully monitor the oxidation of methyl acrylate to ensure complete conversion before introducing the amine component, as residual starting material can complicate downstream purification. The subsequent cyclization step benefits from a one-pot procedure where the base is added directly to the condensation mixture, streamlining the workflow and reducing solvent usage. For a comprehensive understanding of the specific stoichiometric ratios, temperature profiles, and workup procedures validated in the patent examples, please refer to the standardized technical guide below.
- Oxidize methyl acrylate using a palladium or copper catalyst with an oxidant to generate methyl 3-oxopropionate in situ.
- Condense the intermediate with 4-amino-1,1,1-trifluoro-3-buten-2-one to form N-(2-methoxycarbonylvinyl)-4,4,4-trifluoro-3-one-1-butenamine.
- Perform ring-closing hydrolysis using a base such as sodium methoxide or sodium hydroxide at mild temperatures to yield the final acid.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this catalytic synthesis route represents a strategic opportunity to optimize the cost structure and reliability of the Flonicamid supply chain. By shifting away from dependency on specialized, halogenated pyridine starting materials—which are often subject to volatile pricing and limited supplier availability—manufacturers can leverage widely available commodity chemicals like methyl acrylate. This fundamental change in raw material sourcing drastically reduces exposure to supply chain disruptions and provides a more stable cost baseline for long-term contracts. Furthermore, the elimination of hazardous reagents like POCl3 and the avoidance of cryogenic processing significantly lower the operational expenditure related to waste treatment, safety compliance, and energy consumption, translating into substantial overall cost savings for the production of high-purity 4-trifluoromethyl nicotinic acid.
- Cost Reduction in Manufacturing: The new process achieves significant economic advantages by removing the need for expensive strong bases like LDA and costly halogenated intermediates, replacing them with affordable commodity feedstocks and reusable transition metal catalysts. The mild reaction conditions (25-90°C) eliminate the high energy costs associated with maintaining cryogenic temperatures (-75°C) or high-pressure hydrogenation, leading to a drastically simplified utility profile. Additionally, the avoidance of toxic chlorinating agents reduces the financial burden of hazardous waste disposal and environmental compliance, further enhancing the overall profit margin for large-scale production facilities.
- Enhanced Supply Chain Reliability: Sourcing strategies are greatly improved as the primary raw materials, such as methyl acrylate and simple amines, are produced globally in massive volumes, ensuring consistent availability and reducing lead times. Unlike legacy routes that depend on niche suppliers for specific chlorinated pyridines or anhydrous LDA, this method allows manufacturers to diversify their supplier base and mitigate the risk of single-source bottlenecks. The robustness of the chemistry also means that production schedules are less likely to be interrupted by equipment failures associated with extreme conditions, ensuring a steady flow of material to downstream formulation plants.
- Scalability and Environmental Compliance: The process is inherently designed for industrial scale-up, utilizing standard stainless steel reactors and atmospheric pressure operations that are easily replicated from pilot plant to commercial tonnage. The absence of heavy metal waste from hydrogenolysis and phosphorous waste from chlorination aligns perfectly with modern green chemistry principles and increasingly strict environmental regulations. This eco-friendly profile not only simplifies the permitting process for new production lines but also enhances the sustainability credentials of the final agrochemical product, appealing to environmentally conscious end-users and regulatory bodies.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this patented synthesis method for 4-trifluoromethyl nicotinic acid. These insights are derived directly from the experimental data and comparative analysis provided in the patent documentation, focusing on the practical implications for industrial adoption. Understanding these nuances is critical for R&D teams evaluating process transfer and for procurement specialists assessing the long-term viability of this supply route.
Q: Why is the new catalytic method superior to traditional LDA carboxylation?
A: Traditional methods require cryogenic conditions (-75°C) and expensive strong bases like LDA, whereas the new patent utilizes mild temperatures (25-90°C) and commodity catalysts, drastically reducing energy and equipment costs.
Q: Does this process avoid the use of toxic chlorinating agents?
A: Yes, unlike previous routes that relied on POCl3 for chlorination and subsequent hydrogenolysis, this method constructs the pyridine ring directly from acyclic precursors, eliminating hazardous waste streams.
Q: Is this synthesis suitable for large-scale manufacturing of Flonicamid intermediates?
A: Absolutely. The process uses readily available raw materials like methyl acrylate and operates under atmospheric pressure with standard stainless steel reactors, making it highly scalable for industrial production.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 4-Trifluoromethyl Nicotinic Acid Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of this catalytic synthesis technology in securing the future supply of critical agrochemical intermediates. As a leading CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the theoretical benefits of this patent are fully realized in a GMP-compliant manufacturing environment. Our state-of-the-art facilities are equipped to handle the specific catalytic requirements of this process, and our rigorous QC labs enforce stringent purity specifications to guarantee that every batch meets the exacting standards required for Flonicamid synthesis. We are committed to delivering consistent quality and supply continuity, acting as a true extension of your production capabilities.
We invite you to collaborate with us to evaluate the feasibility of integrating this advanced route into your supply chain. Our technical team is prepared to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements, demonstrating exactly how this method can improve your bottom line. Please contact our technical procurement team today to request specific COA data, route feasibility assessments, and sample availability, and let us help you secure a competitive advantage in the global agrochemical market.
