Advanced Catalytic Synthesis of 4-Trifluoromethyl Nicotinic Acid for Industrial Scale-Up
Introduction to Patent CN111574440A
The pharmaceutical and agrochemical industries are constantly seeking more efficient pathways to construct complex heterocyclic scaffolds, particularly those containing fluorine motifs which are critical for metabolic stability and bioactivity. A significant breakthrough in this domain is detailed in Chinese Patent CN111574440A, published on August 25, 2020, which discloses a novel preparation method for 4-trifluoromethyl nicotinic acid. This compound serves as a pivotal building block for the synthesis of Flonicamid, a next-generation pyridylamide insecticide known for its novel mode of action against sucking pests and low toxicity to bees. The patented technology represents a paradigm shift from traditional, harsh synthetic routes to a streamlined, catalytic approach that leverages the oxidative functionalization of simple olefins. By utilizing methyl acrylate as a foundational feedstock and employing transition metal catalysis, the inventors have successfully circumvented the severe limitations of cryogenic lithiation and toxic chlorination reagents. This report provides a deep technical analysis of this innovation, evaluating its mechanistic robustness and its profound implications for supply chain optimization and cost reduction in agrochemical intermediate manufacturing.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Prior to this innovation, the synthesis of 4-trifluoromethyl nicotinic acid was plagued by significant operational and economic hurdles that hindered scalable production. The earliest reported methods relied heavily on the direct carboxylation of 4-trifluoromethylpyridine derivatives using strong bases such as Lithium Diisopropylamide (LDA). As illustrated in the reaction schemes of the prior art, these processes necessitate stringent anhydrous conditions and cryogenic temperatures often as low as -75°C to prevent side reactions and ensure regioselectivity.
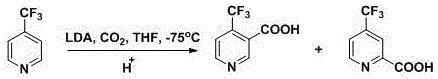
Furthermore, alternative routes involving halogenated pyridines required multiple steps of protection and deprotection, alongside the use of expensive palladium catalysts for dehalogenation. Another prevalent method involved the use of trifluoroacetyl acetic ether and cyanoacetamide, which unfortunately relied on phosphoryl chloride (POCl3) for the crucial chlorination step. This reagent is not only highly toxic and corrosive but also generates substantial amounts of acidic wastewater, posing severe environmental compliance challenges and increasing waste treatment costs. The instability of key intermediates in these older routes, such as 3-methoxy methyl acrylate, further complicated inventory management and reduced overall process reliability.
The Novel Approach
In stark contrast, the methodology disclosed in CN111574440A introduces a remarkably concise and environmentally benign strategy that bypasses these historical bottlenecks. The core innovation lies in the in situ generation of methyl 3-oxopropionate via the catalytic oxidation of methyl acrylate, a commodity chemical with excellent availability and low cost. This intermediate is then condensed with 4-amino-1,1,1-trifluoro-3-buten-2-one to form a stable enamine precursor, which subsequently undergoes a one-pot cyclization and hydrolysis under basic conditions. By shifting the reaction temperature window to a mild range of 25-90°C, the new process eliminates the need for energy-intensive cooling systems and specialized cryogenic reactors. This transition from stoichiometric strong bases to catalytic oxidation not only simplifies the operational protocol but also drastically improves the safety profile of the manufacturing facility, making it an ideal candidate for reliable agrochemical intermediate supplier networks seeking sustainable growth.
Mechanistic Insights into Catalytic Oxidation and Cyclization
The chemical elegance of this new route is rooted in its efficient construction of the pyridine ring through a cascade of condensation and cyclization events driven by thermodynamic stability. The process initiates with the Wacker-type oxidation of methyl acrylate, where a palladium or copper catalyst facilitates the insertion of oxygen to generate the beta-keto ester functionality. This step is critical as it installs the necessary carbonyl group for the subsequent nucleophilic attack. The resulting methyl 3-oxopropionate acts as a versatile electrophile, reacting with the amino group of the trifluoromethylated butenone to form an imine or enamine linkage. This condensation is carefully controlled to maximize the formation of the linear precursor, N-(2-methoxycarbonylvinyl)-4,4,4-trifluoro-3-one-1-butenamine, which serves as the direct scaffold for ring closure.
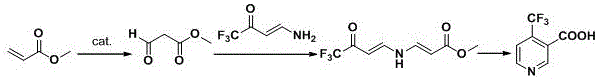
Following the formation of the enamine intermediate, the addition of a base such as sodium methoxide or sodium hydroxide triggers an intramolecular cyclization. The base deprotonates the active methylene group, facilitating a nucleophilic attack on the adjacent carbonyl carbon to close the six-membered pyridine ring. Simultaneously, the ester moiety undergoes hydrolysis to yield the free carboxylic acid, completing the transformation in a highly atom-economical manner. This mechanism avoids the formation of toxic byproducts associated with chlorinating agents and ensures that the trifluoromethyl group remains intact throughout the synthesis. The robustness of this catalytic cycle allows for high purity specifications, as the reaction pathway minimizes the generation of structural isomers or polymeric impurities that are common in radical-based or harsh acidic conditions. For R&D directors, understanding this mechanism highlights the potential for further optimization of catalyst loading and solvent systems to push yields even higher.
How to Synthesize 4-Trifluoromethyl Nicotinic Acid Efficiently
Implementing this synthesis requires precise control over the oxidation step and the subsequent base-mediated cyclization to ensure maximum conversion and minimal impurity formation. The process begins with the preparation of the beta-keto ester intermediate in a biphasic or mixed solvent system, followed by the careful addition of the amine component. Detailed standard operating procedures regarding catalyst activation, oxygen flow rates, and pH adjustments during the workup are essential for reproducibility. The following guide outlines the critical stages of this patented methodology, providing a framework for laboratory validation and pilot plant trials.
- Oxidize methyl acrylate using a palladium or copper catalyst with an oxidant to form methyl 3-oxopropionate.
- Condense methyl 3-oxopropionate with 4-amino-1,1,1-trifluoro-3-buten-2-one to form the enamine intermediate.
- Perform base-catalyzed cyclization and hydrolysis, followed by acidification to isolate the final 4-trifluoromethyl nicotinic acid.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this novel synthesis route offers transformative benefits that extend far beyond simple yield improvements. The shift away from exotic, temperature-sensitive reagents like LDA and toxic chlorinating agents like POCl3 fundamentally alters the cost structure and risk profile of producing high-purity 4-trifluoromethyl nicotinic acid. By utilizing commodity chemicals such as methyl acrylate and oxygen as the primary oxidant, the raw material costs are significantly reduced compared to the specialized fluorinated pyridines required in legacy processes. This reduction in input costs directly translates to improved margins for downstream API manufacturers and formulators, enhancing the overall competitiveness of the final agrochemical product in the global market.
- Cost Reduction in Manufacturing: The elimination of cryogenic infrastructure and the replacement of expensive stoichiometric bases with catalytic systems lead to substantial capital expenditure savings. Furthermore, the avoidance of hazardous waste streams associated with phosphorus chlorides reduces the financial burden of environmental compliance and waste disposal. The simplified workup procedure, which relies on straightforward filtration and acidification, minimizes solvent consumption and processing time, thereby driving down the overall cost of goods sold without compromising quality.
- Enhanced Supply Chain Reliability: Sourcing strategies are greatly simplified as the key starting materials are bulk petrochemical derivatives with stable global supply chains, unlike the niche intermediates required for the LDA route. This abundance ensures consistent availability and mitigates the risk of supply disruptions caused by the limited production capacity of specialized fluorinated building blocks. The robustness of the reaction conditions also means that production can be maintained across a wider range of geographical locations, diversifying the supplier base and strengthening supply chain resilience against regional instabilities.
- Scalability and Environmental Compliance: The mild reaction temperatures and ambient pressure operations make this process inherently safer and easier to scale from kilogram to multi-ton quantities. The absence of highly exothermic steps or gas-evolving reactions reduces the engineering complexity required for reactor design, facilitating faster technology transfer from lab to plant. Additionally, the greener profile of the synthesis aligns with increasingly stringent global environmental regulations, ensuring long-term operational continuity and reducing the likelihood of regulatory shutdowns due to emissions or effluent violations.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this patented synthesis method. These insights are derived directly from the experimental data and comparative analysis presented in the patent documentation, offering clarity on the practical aspects of adopting this technology. Understanding these details is crucial for stakeholders evaluating the feasibility of integrating this route into their existing manufacturing portfolios.
Q: What are the primary advantages of this new synthesis method over traditional LDA routes?
A: The new method eliminates the need for cryogenic conditions (-75°C) and expensive strong bases like LDA, operating instead at mild temperatures (25-90°C) with cheaper, readily available raw materials.
Q: How does this process address environmental concerns associated with prior art?
A: Unlike previous methods relying on toxic phosphoryl chloride (POCl3) for chlorination, this route utilizes catalytic oxidation and base-mediated cyclization, significantly reducing hazardous waste generation.
Q: Is this synthesis route suitable for large-scale industrial production?
A: Yes, the process features simple operation, mild reaction conditions, and low equipment requirements, making it highly feasible for commercial mass production of agrochemical intermediates.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 4-Trifluoromethyl Nicotinic Acid Supplier
As the global demand for advanced agrochemicals like Flonicamid continues to rise, securing a stable and high-quality supply of key intermediates is paramount for maintaining market leadership. NINGBO INNO PHARMCHEM stands at the forefront of this industry, leveraging our extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production to deliver exceptional value to our partners. Our state-of-the-art facilities are equipped to handle the specific catalytic requirements of this novel synthesis, ensuring that every batch meets stringent purity specifications through our rigorous QC labs and advanced analytical capabilities. We are committed to bridging the gap between innovative academic research and industrial reality, providing a seamless transition from process development to full-scale manufacturing.
We invite forward-thinking organizations to collaborate with us to unlock the full potential of this efficient synthesis route. By partnering with our technical procurement team, you can access a Customized Cost-Saving Analysis tailored to your specific volume requirements and logistical needs. We encourage you to reach out today to request specific COA data and route feasibility assessments, allowing us to demonstrate how our expertise in complex organic synthesis can drive efficiency and profitability in your supply chain.
