Optimizing 4-Trifluoromethyl Nicotinic Acid Production: A Safer Aqueous Ammoniation Strategy for Global Agrochemical Supply Chains
Optimizing 4-Trifluoromethyl Nicotinic Acid Production: A Safer Aqueous Ammoniation Strategy for Global Agrochemical Supply Chains
The global demand for high-performance agrochemicals continues to drive innovation in the synthesis of critical intermediates, particularly for next-generation insecticides like Flonicamid. A pivotal development in this sector is detailed in patent CN112079772A, which introduces a revolutionary method for the ammoniation reaction involved in producing 4-trifluoromethyl nicotinic acid. This technical breakthrough addresses long-standing inefficiencies in traditional manufacturing by replacing hazardous gas ammonia protocols with a controlled aqueous ammonia system. For R&D directors and procurement strategists, this shift represents not merely a procedural tweak but a fundamental restructuring of the production landscape that enhances safety profiles while simultaneously boosting throughput. By mitigating the risks associated with exothermic runaway reactions and simplifying thermal management, this patented approach offers a robust pathway for the commercial scale-up of complex fluorinated intermediates. The implications for supply chain stability are profound, as the method reduces dependency on extreme cryogenic conditions that often bottleneck production capacity during peak seasons.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of 4-trifluoromethyl nicotinic acid has relied heavily on gas ammonia reactions, a process fraught with significant operational challenges and safety liabilities. The conventional methodology necessitates stringent temperature control, typically requiring reaction environments to be maintained at approximately minus 10 °C to prevent thermal degradation. Such extreme conditions impose severe demands on refrigeration infrastructure, leading to escalated energy consumption and increased capital expenditure for specialized cooling equipment. Furthermore, the gas-phase reaction is inherently violent, characterized by rapid heat release that is difficult to dissipate uniformly across large reactor volumes. This localized heating often triggers undesirable side reactions, including the self-polymerization of the unstable double bonds present in the intermediate structures. Consequently, the overall yield suffers, with traditional methods often capping at around 85% purity, necessitating costly downstream purification steps to meet the rigorous specifications required for high-purity agrochemical intermediates. The extended reaction times, ranging from 12 to 18 hours, further constrain facility throughput, creating bottlenecks that limit the ability to respond swiftly to market fluctuations.
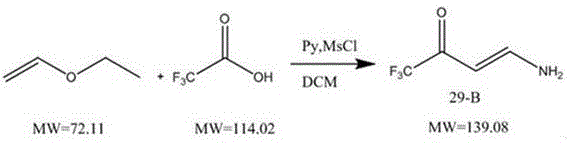
The Novel Approach
In stark contrast to the volatility of gas ammonia, the novel approach delineated in the patent utilizes a diluted aqueous ammonia solution, fundamentally altering the reaction kinetics to favor safety and efficiency. By employing ammonia water with a mass concentration of 15-20%, the process effectively dilutes the reactant concentration, thereby moderating the reaction rate and ensuring a much gentler thermal profile. This modification allows the reaction temperature to be controlled within a significantly more accessible range of minus 5 to 10 °C, drastically reducing the load on cooling systems and minimizing the risk of thermal runaway. The operational window is widened, permitting dropwise addition over a period of 6.5 hours without the fear of sudden exotherms that plague gas-phase alternatives. Moreover, the presence of water acts as a thermal buffer, absorbing excess heat and preventing the localized hot spots that lead to polymerization. As a result, the reaction time is compressed to just 6-8 hours, representing a substantial increase in asset utilization. The final product quality is markedly superior, with assay values consistently reaching 95-96%, demonstrating that this method is a viable solution for cost reduction in pesticide manufacturing through both yield enhancement and energy savings.
Mechanistic Insights into Aqueous Ammoniation Kinetics
The core mechanistic advantage of this process lies in the modulation of nucleophilic attack dynamics through solvent effects and concentration control. In the traditional gas ammonia route, the high local concentration of ammonia molecules leads to an aggressive nucleophilic substitution that releases heat faster than it can be removed by standard jacketed reactors. The introduction of water in the novel method serves a dual purpose: it solvates the ammonia, reducing its effective activity coefficient, and it provides a high-heat-capacity medium to absorb the enthalpy of reaction. This ensures that the conversion of the chlorinated intermediate to the amino-substituted product proceeds smoothly without triggering the decomposition pathways associated with the trifluoromethyl-enone moiety. The careful control of the molar ratio, maintained between 1:1.05 and 1:1.1, ensures that there is sufficient nucleophile to drive the reaction to completion while avoiding a large excess that would complicate downstream waste treatment. This precise stoichiometric balance is critical for maintaining the integrity of the sensitive carbon-carbon double bond adjacent to the carbonyl group, which is prone to Michael-type additions or polymerization under harsh conditions.
Furthermore, the workup procedure leverages phase separation principles to maximize recovery and minimize impurity carryover. Following the reaction, the mixture is allowed to stand, facilitating the separation of the denser dichloromethane layer containing the product from the aqueous phase. A secondary extraction step using fresh dichloromethane ensures that any product remaining in the aqueous layer is recovered, pushing the overall mass balance closer to theoretical limits. The use of anhydrous sodium sulfate for drying removes trace water that could otherwise hydrolyze the product or interfere with the subsequent vacuum distillation. By controlling the distillation pressure at minus 0.09 to minus 0.08 Mpa and keeping the temperature below 60 °C, the process avoids thermal stress on the final molecule. This meticulous attention to downstream processing details ensures that the reliable agrochemical intermediate supplier can deliver material that meets the stringent purity profiles demanded by formulators, free from polymeric tars or hydrolysis byproducts.
How to Synthesize 4-Trifluoromethyl Nicotinic Acid Efficiently
Implementing this optimized synthesis route requires strict adherence to the thermal and stoichiometric parameters defined in the patent to ensure reproducibility and safety. The process begins with the preparation of the chlorination liquid, which must be cooled effectively before the introduction of the aminating agent to prevent premature reaction. Operators must monitor the dropwise addition rate closely, correlating it with the internal reactor temperature to maintain the critical threshold of 10 °C. Detailed standardized synthetic steps see the guide below for a comprehensive breakdown of the operational protocol.
- Cool the chlorination liquid containing the intermediate in a reaction kettle to a temperature range of -8 to -4 °C.
- Dropwise add 15-20% concentration ammonia water at a molar ratio of 1: 1.05-1.1, maintaining the reaction temperature below 10 °C.
- After addition, verify residual intermediate is <0.5%, separate the lower layer, extract the upper layer with dichloromethane, dry with sodium sulfate, and distill under reduced pressure.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the transition from gas ammonia to aqueous ammoniation translates directly into tangible operational efficiencies and risk mitigation. The elimination of high-pressure gas handling systems reduces the regulatory burden and insurance costs associated with hazardous material storage. Additionally, the shortened reaction cycle time allows for more batches to be produced within the same timeframe, effectively increasing plant capacity without the need for capital-intensive expansion. This flexibility is crucial for maintaining reducing lead time for high-purity nicotinic acid derivatives in a volatile market. The improved yield means less raw material is wasted per kilogram of finished product, directly impacting the cost of goods sold. Furthermore, the milder reaction conditions reduce wear and tear on reactor linings and cooling coils, extending the lifespan of critical assets and lowering maintenance overheads.
- Cost Reduction in Manufacturing: The shift to aqueous ammonia eliminates the need for complex gas scrubbing and containment infrastructure, leading to significant capital and operational expenditure savings. The energy consumption is drastically lowered because the process does not require maintaining cryogenic temperatures of minus 10 °C, allowing for the use of standard chilled water systems instead of specialized brine or glycol loops. Higher yields of 95-96% compared to 85% mean that the effective cost per unit of active ingredient is reduced, as less starting material is lost to side reactions and polymerization. These factors combine to create a leaner manufacturing process that is highly competitive in price-sensitive agrochemical markets.
- Enhanced Supply Chain Reliability: Safety is a primary driver of supply continuity; by removing the intrinsic hazards of gas ammonia leaks and violent exotherms, the risk of unplanned shutdowns due to safety incidents is minimized. The process is more robust against minor fluctuations in cooling capacity, meaning that production schedules are less likely to be disrupted by equipment failures or ambient temperature variations. This reliability ensures that downstream customers receive their shipments on time, fostering stronger long-term partnerships. The ability to operate safely at higher temperatures also expands the geographical locations where production can be sited, diversifying the supply base.
- Scalability and Environmental Compliance: Scaling chemical processes often amplifies heat transfer issues, but this method's reliance on dilution and moderate temperatures makes it inherently scalable from pilot plants to multi-ton reactors. The use of water as a diluent simplifies waste stream management, as aqueous ammonia residues are easier to neutralize and treat than concentrated gas scrubber effluents. Reduced solvent usage during workup, combined with efficient recovery via distillation, aligns with green chemistry principles and helps facilities meet increasingly strict environmental regulations. This sustainability angle is increasingly important for multinational corporations aiming to reduce the carbon footprint of their supply chains.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this ammoniation technology. These insights are derived directly from the experimental data and comparative examples provided in the patent documentation, offering clarity on performance metrics and operational feasibility. Understanding these details is essential for technical teams evaluating the integration of this process into existing manufacturing lines.
Q: How does the aqueous ammoniation method improve safety compared to gas ammonia?
A: The use of ammonia water dilutes the reactant concentration, resulting in a milder reaction with less heat release. This eliminates the strict low-temperature control (-10 °C) required for gas ammonia, significantly reducing intrinsic safety hazards and the risk of runaway reactions.
Q: What yield improvements can be expected with this new process?
A: By preventing local overheating and subsequent polymerization of the unstable double bond structure, the new method achieves product content of 95-96%, compared to approximately 85% with conventional gas ammonia methods.
Q: Is this process suitable for large-scale commercial production?
A: Yes, the process shortens reaction time to 6-8 hours and operates at more manageable temperatures (-5 to 10 °C), making heat exchange and temperature control much easier on a large industrial scale compared to the difficult -10 °C requirement of gas ammonia.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 4-Trifluoromethyl Nicotinic Acid Supplier
At NINGBO INNO PHARMCHEM, we recognize that the transition to safer, more efficient synthetic routes is critical for maintaining competitiveness in the global agrochemical sector. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that innovations like the aqueous ammoniation process are seamlessly translated into industrial reality. We are committed to delivering materials with stringent purity specifications, supported by our rigorous QC labs that utilize advanced analytical techniques to verify every batch. Our infrastructure is designed to handle complex fluorinated chemistries with the highest safety standards, providing our partners with peace of mind regarding both quality and compliance.
We invite you to collaborate with us to optimize your supply chain for Flonicamid and related intermediates. Contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We are prepared to provide specific COA data and route feasibility assessments to demonstrate how our capabilities align with your strategic sourcing goals. Let us help you secure a stable, high-quality supply of critical intermediates for your future projects.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
