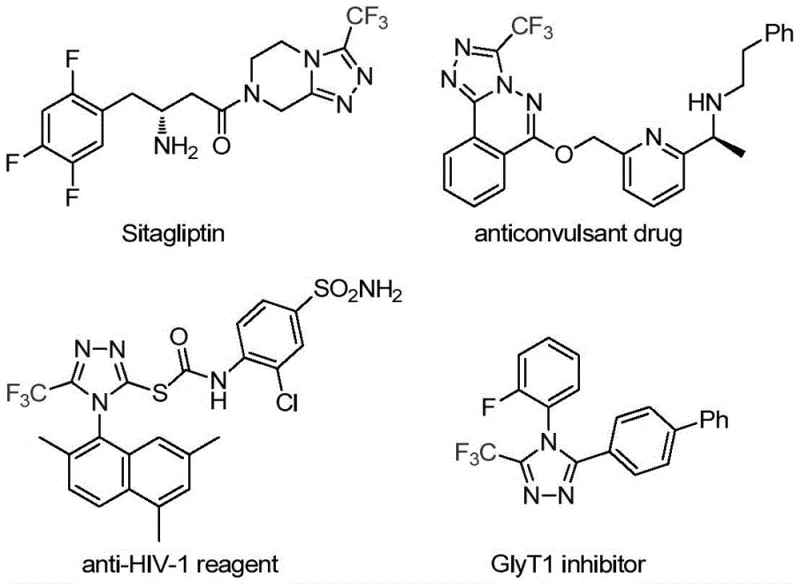
Advanced Catalytic Route for 3-Trifluoromethyl-1,2,4-Triazole Pharmaceutical Intermediates Manufacturing
The pharmaceutical industry continuously seeks robust and efficient synthetic routes for heterocyclic scaffolds that serve as critical building blocks for active pharmaceutical ingredients (APIs). A significant breakthrough in this domain is detailed in patent CN113307778A, which discloses a novel preparation method for 3-trifluoromethyl substituted 1,2,4-triazole compounds. These specific heterocyclic structures are not merely academic curiosities; they are foundational motifs found in numerous high-value drugs, including the widely prescribed antidiabetic agent Sitagliptin and various antiviral reagents. The introduction of a trifluoromethyl group into these molecular frameworks is known to drastically enhance physicochemical properties such as metabolic stability, lipophilicity, and bioavailability, making the efficient construction of these bonds a priority for modern medicinal chemistry.
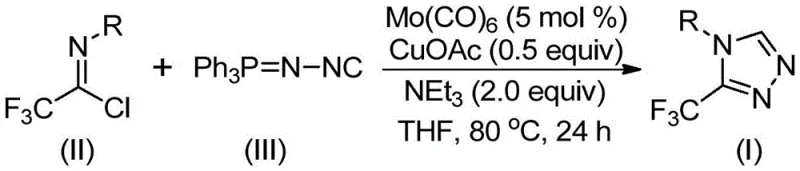
This patented technology represents a paradigm shift from conventional synthesis strategies by employing a dual-metal catalytic system involving molybdenum hexacarbonyl and cuprous acetate. Unlike older methods that often rely on harsh conditions or unstable precursors, this approach operates under relatively mild thermal conditions ranging from 70°C to 90°C. The process utilizes functionalized isonitriles and trifluoroethylimidoyl chloride as key starting materials, reacting them in the presence of a base and molecular sieves within an organic solvent. This strategic combination allows for the direct formation of the 1,2,4-triazole ring with high atom economy and operational simplicity, addressing long-standing challenges in the scalable production of fluorinated heterocycles.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of trifluoromethyl-substituted 1,2,4-triazoles has been fraught with significant technical and safety hurdles that impede efficient commercial manufacturing. Traditional literature reports predominantly describe cyclization reactions involving trifluoroacetyl hydrazine and amidine compounds, or the hydrazinolysis of trifluoromethyl-substituted 1,2,4-oxazolinones. These pathways often necessitate the use of highly reactive and potentially explosive reagents, such as diazonium salts and trifluorodiazoethane, which pose severe safety risks during scale-up operations. Furthermore, multi-component reactions utilizing these dangerous precursors frequently require stringent temperature controls and specialized equipment to manage exothermic events, thereby inflating capital expenditure and operational complexity. Another prevalent issue with legacy methods is the limited scope of substrate compatibility; many established protocols fail to tolerate sensitive functional groups, leading to low yields or complex impurity profiles that require extensive and costly purification steps to resolve.
The Novel Approach
In stark contrast to these hazardous and inefficient legacy processes, the method disclosed in CN113307778A introduces a streamlined and safer catalytic cycle that leverages the synergistic effects of molybdenum and copper catalysis. By utilizing trifluoroethylimidoyl chloride and functionalized isonitrile (specifically Ph3P=N-NC) as the core reactants, this novel route bypasses the need for unstable diazo compounds entirely. The reaction proceeds through a cooperative mechanism where molybdenum hexacarbonyl activates the isonitrile species while cuprous acetate facilitates the cycloaddition, resulting in the rapid assembly of the triazole ring. This approach not only simplifies the reaction setup—requiring only standard heating and stirring in common solvents like THF—but also dramatically expands the chemical space accessible to chemists. The ability to introduce diverse substituents on the aryl ring without compromising yield makes this method a superior choice for generating libraries of drug candidates and optimizing lead compounds.
Mechanistic Insights into Mo/Cu Co-catalyzed Cycloaddition
The efficacy of this synthesis lies in the intricate interplay between the two metal centers and the specific activation of the reactants. Mechanistically, the reaction is initiated by the coordination of molybdenum hexacarbonyl to the functionalized isonitrile, forming a reactive metal-isocyanide complex that lowers the energy barrier for subsequent bond formation. Simultaneously, the cuprous acetate acts as a Lewis acid promoter, facilitating the nucleophilic attack of the activated isonitrile onto the electrophilic carbon of the trifluoroethylimidoyl chloride. This cooperative activation drives a [3+2] cycloaddition event, constructing the five-membered 1,2,4-triazole ring intermediate with high regioselectivity. Following the ring closure, the system undergoes a crucial elimination step where triphenylphosphine oxide is removed, likely aided by trace water present in the reaction matrix or added during workup, to yield the final stable aromatic triazole product. This mechanistic pathway ensures that the trifluoromethyl group is retained at the 3-position with high fidelity, avoiding the scrambling or loss of fluorine that can occur in more aggressive acidic or basic environments.
From an impurity control perspective, this mechanism offers distinct advantages for maintaining high product purity, a critical parameter for regulatory compliance in pharmaceutical manufacturing. The use of molecular sieves in the reaction mixture plays a pivotal role in scavenging moisture that could otherwise hydrolyze the sensitive imidoyl chloride starting material, thereby minimizing the formation of carboxylic acid byproducts. Furthermore, the mild reaction temperatures (70-90°C) prevent thermal degradation of the product or the formation of polymeric tars that are common in high-temperature cyclizations. The specificity of the Mo/Cu catalytic pair ensures that side reactions, such as homocoupling of the isonitrile or non-selective substitution on the aryl ring, are suppressed. Consequently, the crude reaction mixture typically exhibits a cleaner profile, which simplifies downstream processing and reduces the burden on purification resources, ultimately leading to a more robust and reproducible manufacturing process.
How to Synthesize 3-Trifluoromethyl-1,2,4-Triazole Efficiently
The practical implementation of this synthesis is designed for ease of execution in both laboratory and pilot plant settings. The protocol involves charging a reaction vessel with the requisite molar ratios of molybdenum hexacarbonyl, cuprous acetate, triethylamine, and molecular sieves, followed by the addition of the trifluoroethylimidoyl chloride and the functionalized isonitrile in an aprotic solvent like tetrahydrofuran (THF). The mixture is then heated to the optimal temperature range and stirred for a defined period to ensure full conversion. Detailed standardized operating procedures regarding exact stoichiometry, solvent volumes, and workup specifics are essential for consistent results.
- Combine molybdenum hexacarbonyl, cuprous acetate, triethylamine, molecular sieves, trifluoroethylimidoyl chloride, and functionalized isonitrile in an organic solvent such as THF.
- Heat the reaction mixture to a temperature between 70°C and 90°C and maintain stirring for a duration of 18 to 30 hours to ensure complete conversion.
- Upon completion, filter the mixture, mix the crude sample with silica gel, and perform column chromatography purification to isolate the final high-purity triazole product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this patented methodology translates into tangible strategic benefits that extend beyond mere chemical yield. The shift towards this Mo/Cu co-catalyzed route addresses several critical pain points associated with the sourcing and production of complex heterocyclic intermediates. By eliminating the reliance on hazardous diazonium salts and unstable diazo compounds, the process significantly reduces the safety infrastructure costs and regulatory burdens associated with handling energetic materials. This inherent safety profile allows for more flexible manufacturing scheduling and reduces the risk of production stoppages due to safety audits or incidents, thereby enhancing overall supply chain reliability. Furthermore, the use of commercially available and inexpensive catalysts like cuprous acetate and molybdenum hexacarbonyl, as opposed to precious metals like palladium or rhodium, contributes to substantial cost savings in raw material procurement.
- Cost Reduction in Manufacturing: The economic viability of this process is underpinned by the use of cheap and easily obtainable starting materials, such as aromatic amines and trifluoroacetic acid derivatives, which are commodity chemicals with stable global supply chains. The elimination of expensive transition metal catalysts and the avoidance of cryogenic conditions required by some traditional methods lead to a drastic simplification of the production workflow. Additionally, the high reaction efficiency and selectivity minimize the generation of waste byproducts, reducing the costs associated with waste disposal and solvent recovery. This streamlined approach ensures that the cost of goods sold (COGS) for these valuable intermediates remains competitive, providing a clear margin advantage for downstream API manufacturers.
- Enhanced Supply Chain Reliability: Supply continuity is often threatened by the scarcity of specialized reagents; however, this method relies on robust, shelf-stable reagents that are widely sourced from multiple global suppliers. The tolerance of the reaction to various functional groups means that a single standardized protocol can be used to produce a wide array of derivatives, reducing the need for multiple distinct production lines or specialized equipment setups. This versatility allows manufacturers to respond rapidly to changing market demands for different triazole variants without significant retooling delays. The simplified post-treatment process, involving standard filtration and chromatography, further ensures that batch turnover times are optimized, facilitating just-in-time delivery models for key clients.
- Scalability and Environmental Compliance: The patent explicitly highlights the method's scalability, noting successful expansion to gram-level reactions with maintained efficiency, which serves as a strong indicator for kilogram and tonne-scale feasibility. The mild reaction conditions reduce energy consumption compared to high-temperature or high-pressure alternatives, aligning with modern green chemistry principles and corporate sustainability goals. Moreover, the reduced toxicity of the reagent suite simplifies environmental compliance and effluent treatment, lowering the ecological footprint of the manufacturing process. This alignment with environmental, social, and governance (ESG) criteria is increasingly becoming a prerequisite for partnerships with major multinational pharmaceutical companies.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis technology. These insights are derived directly from the experimental data and beneficial effects reported in the patent documentation, providing a clear understanding of the method's capabilities and limitations for potential adopters.
Q: What are the primary advantages of this Mo/Cu co-catalyzed method over traditional triazole synthesis?
A: This novel method utilizes mild reaction conditions (70-90°C) and avoids hazardous reagents like diazonium salts often found in traditional routes. It employs cheap and readily available starting materials, offering a safer and more cost-effective pathway for large-scale production.
Q: Can this synthesis method accommodate diverse functional groups on the substrate?
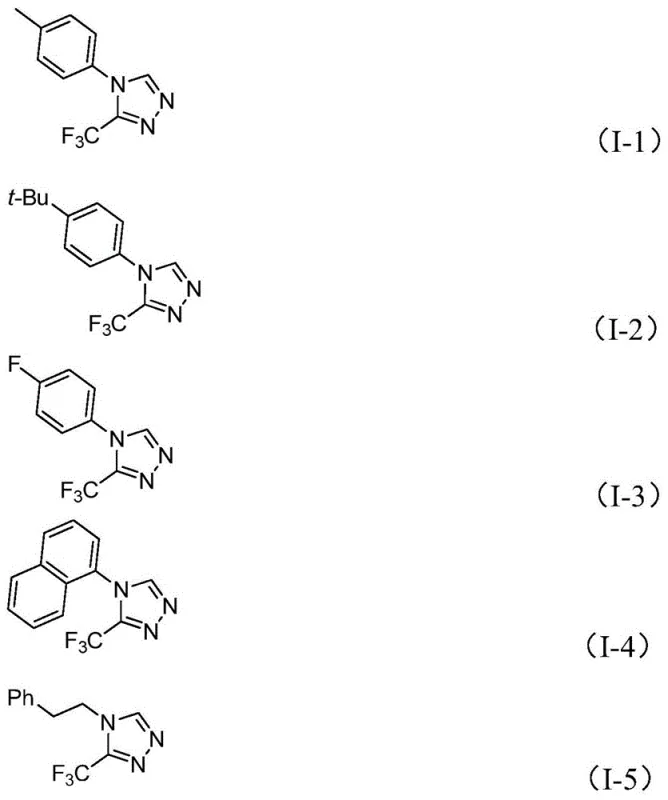
A: Yes, the method demonstrates excellent substrate tolerance. It successfully synthesizes derivatives with various substituents including alkyl, alkoxy, halogen, and nitro groups on the aryl ring, making it highly versatile for designing specific pharmaceutical intermediates.
Q: Is this process suitable for commercial scale-up?
A: The patent explicitly states that the reaction efficiency is high and the operation is simple, allowing for easy expansion to gram-level reactions. The use of standard post-processing techniques like column chromatography further supports its feasibility for industrial scaling.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 3-Trifluoromethyl-1,2,4-Triazole Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-quality heterocyclic intermediates play in the development of next-generation therapeutics. Our team of expert chemists has thoroughly analyzed the technological potential of the Mo/Cu co-catalyzed synthesis route described in CN113307778A and is fully prepared to integrate this advanced methodology into our production portfolio. We possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your supply needs are met with unwavering consistency. Our state-of-the-art facilities are equipped with rigorous QC labs and adhere to stringent purity specifications, guaranteeing that every batch of 3-trifluoromethyl-1,2,4-triazole intermediate meets the highest international standards for pharmaceutical applications.
We invite you to collaborate with us to leverage this cutting-edge synthesis technology for your specific drug development programs. By partnering with our technical procurement team, you can access a Customized Cost-Saving Analysis tailored to your project's unique volume and purity requirements. We encourage you to reach out today to request specific COA data and comprehensive route feasibility assessments, allowing us to demonstrate how our commitment to innovation and quality can accelerate your timeline to market while optimizing your overall production costs.
