Advanced Synthesis of 3-Trifluoromethyl-1,2,4-Triazoles for Commercial API Manufacturing
The pharmaceutical and agrochemical industries continuously seek robust synthetic pathways for heterocyclic scaffolds that possess high biological activity and metabolic stability. Patent CN114920707B, published in April 2023, introduces a groundbreaking preparation method for 3-trifluoromethyl substituted 1,2,4-triazole compounds, a structural motif prevalent in numerous high-value therapeutic agents. As illustrated in the provided chemical landscape, these triazole cores are integral to the architecture of blockbuster drugs such as Sitagliptin, Factor IXa inhibitors, and GlyT1 inhibitors, where the trifluoromethyl group enhances lipophilicity and bioavailability. This novel methodology distinguishes itself by utilizing the ubiquitous organic solvent N,N-dimethylformamide (DMF) not merely as a medium, but as a critical carbon source, thereby streamlining the synthetic sequence and reducing the reliance on exotic reagents.
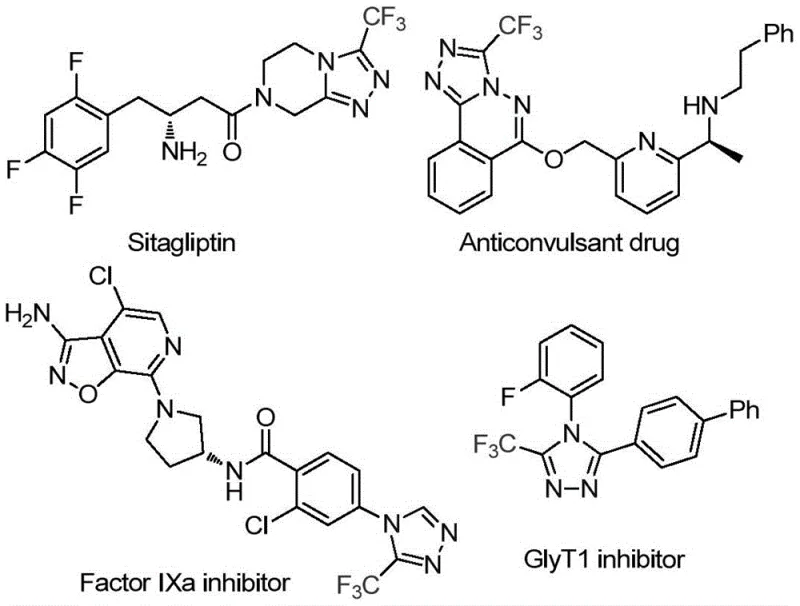
For R&D directors and process chemists, the significance of this patent lies in its ability to access complex fluorinated heterocycles through a simplified operational protocol. The method eliminates the need for specialized catalysts or harsh conditions often associated with traditional triazole synthesis, offering a direct route to high-purity intermediates essential for late-stage functionalization. By leveraging the dual functionality of DMF, the process achieves high reaction efficiency while maintaining a low environmental footprint, addressing the growing demand for green chemistry principles in modern API manufacturing. This report analyzes the technical merits and commercial implications of adopting this iodine-promoted tandem cyclization strategy for large-scale production.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the construction of 3-trifluoromethyl-1,2,4-triazole rings has been fraught with synthetic challenges that hinder efficient commercial scale-up. Traditional routes often necessitate the use of expensive, moisture-sensitive reagents and require stringent anhydrous and anaerobic conditions to prevent side reactions or decomposition of reactive intermediates. Many existing protocols involve multi-step sequences where the introduction of the trifluoromethyl group and the formation of the triazole ring are disjointed processes, leading to cumulative yield losses and increased waste generation. Furthermore, conventional methods frequently rely on stoichiometric amounts of heavy metal catalysts or hazardous oxidants, which complicate downstream purification and raise significant concerns regarding residual metal impurities in the final active pharmaceutical ingredient. These factors collectively drive up the cost of goods sold (COGS) and extend the lead time for process development, creating bottlenecks for supply chain managers aiming for rapid market entry.
The Novel Approach
In stark contrast, the methodology disclosed in CN114920707B represents a paradigm shift towards atom-economical and operationally simple synthesis. The core innovation involves an iodine-promoted tandem cyclization reaction where DMF acts simultaneously as the solvent and the reactant, providing the necessary carbon atoms for ring closure. As depicted in the general reaction scheme below, trifluoroethyliminohydrazide reacts directly with DMF in the presence of molecular iodine under an air atmosphere. This approach obviates the need for inert gas protection or rigorous drying of solvents, significantly lowering the barrier for implementation in standard manufacturing facilities. The reaction proceeds smoothly at temperatures between 110°C and 130°C, demonstrating remarkable tolerance to various functional groups on the aromatic ring, which allows for the synthesis of a diverse library of derivatives without modifying the core protocol.
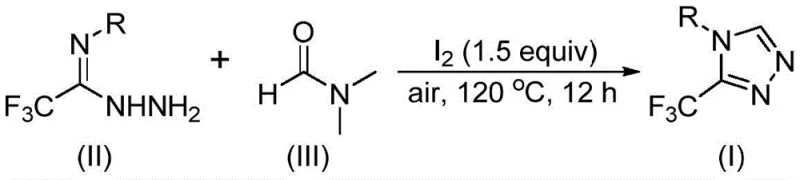
Mechanistic Insights into Iodine-Promoted Tandem Cyclization
Understanding the mechanistic underpinnings of this transformation is crucial for optimizing reaction parameters and ensuring consistent product quality. The reaction mechanism is versatile, with DMF capable of participating via two distinct pathways depending on which functional group acts as the carbon source. In one pathway, the formyl group of DMF undergoes condensation with the hydrazide moiety of the starting material to form a hydrazone intermediate. This is followed by an intramolecular cyclization event that eliminates a molecule of dimethylamine, directly yielding the 3-trifluoromethyl-1,2,4-triazole product. Alternatively, the N-methyl group of DMF can serve as the carbon donor; in this scenario, DMF first reacts with molecular iodine to generate an activated amine salt species. Subsequent nucleophilic addition by the hydrazide, elimination of N-methylformamide, and formation of an azadiene intermediate lead to the final product through oxidative aromatization. This mechanistic flexibility ensures high conversion rates even with varied substrate electronics.
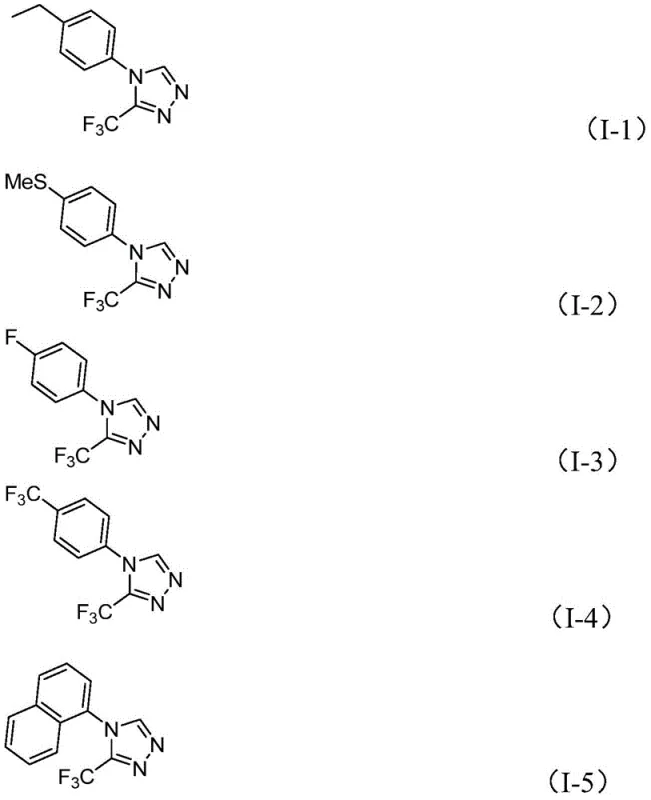
From an impurity control perspective, the use of molecular iodine as a promoter rather than a stoichiometric oxidant helps minimize the formation of over-oxidized byproducts. The reaction conditions are mild enough to preserve sensitive functional groups such as halides and ethers, which are common in medicinal chemistry scaffolds. The patent data indicates that substrates bearing electron-withdrawing groups like trifluoromethyl or halogens, as well as electron-donating groups like methoxy or alkyl chains, are all compatible with the system. This broad substrate scope is evidenced by the successful synthesis of derivatives I-1 through I-5, which feature diverse aromatic substitutions including ethyl, methylthio, fluoro, and naphthyl groups. Such versatility confirms the robustness of the catalytic cycle and its suitability for generating focused libraries for structure-activity relationship (SAR) studies.
How to Synthesize 3-Trifluoromethyl-1,2,4-Triazole Efficiently
The practical execution of this synthesis is designed for ease of handling, making it accessible for both laboratory discovery and pilot plant operations. The procedure involves simply combining the trifluoroethyliminohydrazide precursor, molecular iodine, and DMF in a reaction vessel, followed by heating under ambient air pressure. The simplicity of the setup reduces the requirement for specialized glassware or engineering controls typically associated with hazardous chemistries. For detailed operational parameters, stoichiometry, and workup procedures, please refer to the standardized synthesis guide provided below.
- Combine molecular iodine, trifluoroethyliminohydrazide, and DMF solvent in a reaction vessel under air.
- Heat the mixture to 110-130°C and maintain reaction for 10-15 hours to ensure complete conversion.
- Perform post-treatment including filtration, washing, and column chromatography to isolate the pure triazole product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this DMF-based synthesis offers tangible strategic advantages that directly impact the bottom line and operational resilience. The primary driver for cost reduction is the dual utility of DMF; by serving as both the bulk solvent and a reactant, the process eliminates the need to purchase and handle separate, often expensive, one-carbon synthons. This consolidation of materials simplifies inventory management and reduces the overall mass intensity of the process. Additionally, the ability to run the reaction under air without stringent moisture control translates to significant savings in utility costs and equipment maintenance, as there is no need for nitrogen blanketing systems or specialized drying trains. The streamlined workflow allows for faster batch turnover times, enhancing the overall throughput of the manufacturing facility.
- Cost Reduction in Manufacturing: The economic benefits of this process are derived from the substitution of costly reagents with commodity chemicals. DMF is a widely available, low-cost industrial solvent, and molecular iodine is used in catalytic or near-stoichiometric amounts, avoiding the expense of precious metal catalysts like palladium or rhodium often found in cross-coupling alternatives. Furthermore, the simplified workup procedure, which typically involves filtration and standard chromatography, reduces the consumption of silica gel and elution solvents. By minimizing the number of unit operations and reagent inputs, the overall variable cost per kilogram of the API intermediate is drastically lowered, improving margin potential for high-volume products.
- Enhanced Supply Chain Reliability: Supply chain continuity is bolstered by the use of readily available starting materials. Trifluoroethyliminohydrazide can be synthesized from common aromatic amines and trifluoroacetic acid, both of which have established global supply chains with multiple qualified vendors. The robustness of the reaction conditions means that production is less susceptible to disruptions caused by environmental fluctuations or minor variations in raw material quality. This reliability ensures consistent delivery schedules for downstream customers, mitigating the risk of stockouts that can halt clinical trials or commercial drug launches. The scalability of the process from gram to ton scale further secures long-term supply agreements.
- Scalability and Environmental Compliance: From an environmental, health, and safety (EHS) standpoint, this method aligns well with modern green chemistry initiatives. The absence of heavy metal catalysts simplifies waste treatment and reduces the burden of hazardous waste disposal. The reaction generates dimethylamine or N-methylformamide as byproducts, which are manageable within standard effluent treatment protocols. The high atom economy and reduced solvent usage contribute to a lower E-factor (environmental factor), making the process more sustainable. This compliance with stricter environmental regulations future-proofs the manufacturing site against evolving regulatory landscapes, avoiding potential fines or shutdowns associated with non-compliant waste streams.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis technology. These answers are derived directly from the experimental data and beneficial effects described in the patent documentation, providing clarity on process capabilities and limitations. Understanding these details is essential for making informed decisions about technology transfer and process validation.
Q: What is the primary advantage of using DMF in this triazole synthesis?
A: DMF serves a dual role as both the reaction solvent and the carbon source (providing either the formyl or methyl group), which significantly simplifies the reagent list and reduces raw material costs compared to traditional methods requiring separate carbon donors.
Q: Does this process require strict anhydrous or anaerobic conditions?
A: No, one of the key operational benefits of this patented method is that it proceeds efficiently under standard air atmosphere without the need for rigorous anhydrous or oxygen-free environments, greatly facilitating industrial scale-up.
Q: What types of substituents are tolerated on the aromatic ring?
A: The method demonstrates broad substrate scope, successfully accommodating various substituents including alkyl, alkoxy, alkylthio, halogens (fluorine, chlorine), and trifluoromethyl groups at ortho, meta, or para positions.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 3-Trifluoromethyl-1,2,4-Triazole Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-quality intermediates play in the success of your drug development programs. Our team of expert process chemists has extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory bench to manufacturing plant is seamless and efficient. We are committed to delivering products that meet stringent purity specifications, supported by our rigorous QC labs equipped with state-of-the-art analytical instrumentation. By leveraging advanced synthetic methodologies like the one described in CN114920707B, we can offer competitive pricing without compromising on quality or supply security.
We invite you to collaborate with us to optimize your supply chain for trifluoromethyl-triazole derivatives. Contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your specific project needs. We are ready to provide specific COA data and comprehensive route feasibility assessments to demonstrate how our manufacturing capabilities can accelerate your timeline to market. Let us be your partner in turning complex chemical challenges into commercial successes.
