Advanced Synthesis of Thienopyrrole Derivatives for Commercial Scale-Up
Advanced Synthesis of Thienopyrrole Derivatives for Commercial Scale-Up
The pharmaceutical industry's relentless pursuit of potent glycogen phosphorylase inhibitors has placed significant demand on the supply chain for high-quality heterocyclic intermediates. Patent CN1688587A introduces a transformative methodology for the preparation of thienopyrrole derivatives, specifically addressing the critical bottlenecks of safety and scalability that have historically plagued this chemical class. By delineating a robust pathway that circumvents the explosive hazards associated with traditional acid preparation methods, this intellectual property offers a viable blueprint for the reliable thienopyrrole intermediate supplier seeking to optimize their manufacturing portfolio. The core innovation lies in the strategic use of nitrogen-protecting groups and a mild, base-mediated cyclization protocol that ensures high purity while minimizing operational risks. For R&D directors and procurement specialists alike, understanding the nuances of this process is essential for securing a stable supply of these valuable pharmacophores.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of thienopyrrole cores often relied on direct functionalization strategies that presented severe safety liabilities and yield inconsistencies. As noted in the background of the patent, previous attempts to prepare the necessary acid precursors involved reaction conditions that were difficult to realize practically due to the potential for detonation or uncontrolled exothermic events. These conventional routes frequently necessitated harsh reagents and extreme thermal conditions, which not only compromised the safety of the production facility but also led to complex impurity profiles that were costly to remediate. Furthermore, the lack of effective protecting group strategies in older methodologies often resulted in non-selective reactions, where the sensitive thiophene ring or the nascent pyrrole nitrogen underwent unwanted side reactions. This lack of chemoselectivity forced manufacturers to employ extensive and wasteful purification steps, driving up the cost of goods and extending lead times for high-purity pharmaceutical intermediates.
The Novel Approach
In stark contrast, the novel approach detailed in CN1688587A leverages a sophisticated sequence of protection, formylation, and cyclization to achieve superior process control. The method initiates with the conversion of a thiophene carboxylic acid into a protected amine derivative, effectively masking the nitrogen functionality to prevent interference during subsequent electrophilic substitutions. 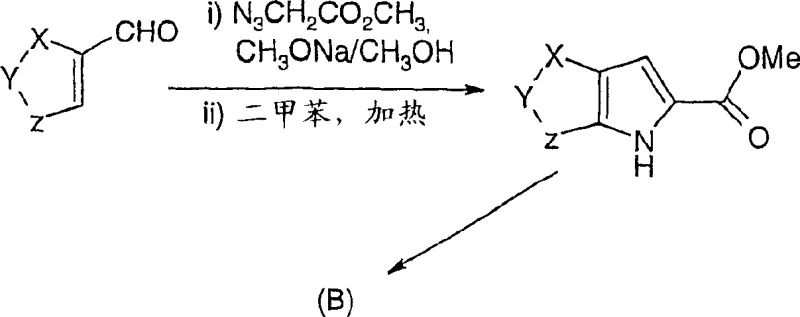 This strategic masking allows for a highly selective Vilsmeier-Haack formylation, introducing the crucial aldehyde moiety required for ring closure without degrading the molecular scaffold. The subsequent cyclization step utilizes mild alkali metal alcoholates, such as sodium methylate in methanol, operating at temperatures ranging from ambient to 70°C. This gentle thermal profile stands in direct opposition to the hazardous conditions of the past, enabling a streamlined workflow that significantly reduces energy consumption and safety infrastructure requirements. By decoupling the ring formation from the initial functionalization, this route offers a modular platform for generating diverse thienopyrrole analogues with consistent quality.
This strategic masking allows for a highly selective Vilsmeier-Haack formylation, introducing the crucial aldehyde moiety required for ring closure without degrading the molecular scaffold. The subsequent cyclization step utilizes mild alkali metal alcoholates, such as sodium methylate in methanol, operating at temperatures ranging from ambient to 70°C. This gentle thermal profile stands in direct opposition to the hazardous conditions of the past, enabling a streamlined workflow that significantly reduces energy consumption and safety infrastructure requirements. By decoupling the ring formation from the initial functionalization, this route offers a modular platform for generating diverse thienopyrrole analogues with consistent quality.
Mechanistic Insights into Base-Mediated Cyclization and Formylation Control
The mechanistic elegance of this synthesis is best observed in the final cyclization step, where the linear precursor undergoes an intramolecular condensation to form the fused bicyclic system. The reaction proceeds through the deprotonation of the active methylene group adjacent to the ester functionality by the alkoxide base, generating a nucleophilic enolate species. This enolate then attacks the electrophilic aldehyde carbon introduced in the previous step, initiating a cascade that closes the pyrrole ring. The use of sodium methylate is particularly advantageous as it serves both as a base to drive the condensation and as a transesterification agent if different ester protecting groups are employed, ensuring the final product is obtained as the methyl ester directly.  This dual functionality simplifies the reaction stoichiometry and reduces the number of unit operations required, which is a critical factor for cost reduction in pharmaceutical manufacturing. The mechanism also accounts for the removal of the nitrogen-protecting group under the same basic conditions or in a subsequent hydrolysis step, revealing the free NH of the pyrrole ring which is essential for the biological activity of the final drug candidate.
This dual functionality simplifies the reaction stoichiometry and reduces the number of unit operations required, which is a critical factor for cost reduction in pharmaceutical manufacturing. The mechanism also accounts for the removal of the nitrogen-protecting group under the same basic conditions or in a subsequent hydrolysis step, revealing the free NH of the pyrrole ring which is essential for the biological activity of the final drug candidate.
Impurity control is another pillar of this technological advancement, particularly regarding the formation of amidine byproducts during the formylation stage. The patent elucidates that under certain Vilsmeier-Haack conditions, the reaction between the protected thiophene and the formylating reagent can generate significant proportions of amidine derivatives (Formula VII), which are difficult to separate and reduce overall yield. To mitigate this, the process employs an optimized protocol where the substrate is added to a pre-formed complex of phosphorus oxychloride and DMF, rather than mixing all reagents simultaneously.  This kinetic control, combined with precise temperature management (maintaining the reaction at room temperature for extended periods before quenching), suppresses the over-reaction that leads to amidine generation. Furthermore, the patent describes a remediation strategy where any formed amidine can be hydrolyzed back to the desired aldehyde using acetic acid and water under reflux, providing a safety net that ensures high mass balance and purity. This level of mechanistic understanding allows process chemists to fine-tune reaction parameters to meet stringent regulatory specifications for impurity limits.
This kinetic control, combined with precise temperature management (maintaining the reaction at room temperature for extended periods before quenching), suppresses the over-reaction that leads to amidine generation. Furthermore, the patent describes a remediation strategy where any formed amidine can be hydrolyzed back to the desired aldehyde using acetic acid and water under reflux, providing a safety net that ensures high mass balance and purity. This level of mechanistic understanding allows process chemists to fine-tune reaction parameters to meet stringent regulatory specifications for impurity limits.
How to Synthesize 2-Chloro-6H-thieno[2,3-b]pyrrole-5-carboxylate Efficiently
The practical execution of this synthesis involves a sequence of well-defined unit operations that balance reaction efficiency with ease of isolation. Starting from commercially available 5-chlorothiophene-2-carboxylic acid, the process first installs a tert-butoxycarbonyl (Boc) or similar protecting group via a Curtius rearrangement, creating a stable intermediate that can be stored and handled safely. Following protection, the molecule undergoes formylation to install the aldehyde handle, followed by N-alkylation with a haloacetate to introduce the cyclization precursor. The final ring closure is achieved under basic conditions, yielding the target thienopyrrole ester in high purity. For a comprehensive breakdown of the specific reagents, stoichiometry, and workup procedures required to replicate this high-yielding pathway, please refer to the standardized guide below.
- Protect the amino group of the thiophene starting material using a Curtius rearrangement with diphenyl phosphoryl azide to form a carbamate.
- Perform Vilsmeier-Haack formylation using POCl3 and DMF, optimizing conditions to minimize amidine byproduct formation.
- Alkylate the formylated intermediate with methyl bromoacetate in the presence of a base like potassium carbonate.
- Execute the final cyclization using sodium methylate in methanol under reflux to close the pyrrole ring and yield the target ester.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of the methodology described in CN1688587A translates into tangible operational benefits that extend beyond mere chemical yield. The primary advantage lies in the substantial mitigation of safety risks, which directly correlates to lower insurance premiums and reduced capital expenditure on specialized blast-proof containment equipment. By eliminating the need for hazardous high-energy intermediates, the process facilitates a smoother regulatory approval path for the manufacturing site, ensuring uninterrupted production schedules. Additionally, the use of commodity chemicals such as sodium methylate, potassium carbonate, and common organic solvents ensures that the supply chain remains resilient against raw material shortages, a critical consideration for maintaining continuity in the global pharmaceutical market.
- Cost Reduction in Manufacturing: The streamlined nature of this synthetic route eliminates several costly purification steps that were previously necessary to remove explosive byproducts and complex impurity mixtures. By achieving high selectivity through the use of protecting groups and optimized formylation conditions, the process minimizes the loss of valuable starting materials, thereby improving the overall atom economy. The ability to perform the cyclization and deprotection in a single pot or under mild conditions further reduces energy consumption and solvent usage, leading to significant cost savings in utility and waste disposal. Moreover, the robustness of the reaction conditions allows for the use of less expensive reactor materials, avoiding the need for exotic alloys required for highly corrosive or high-pressure environments.
- Enhanced Supply Chain Reliability: The reliance on widely available starting materials, such as substituted thiophene carboxylic acids and standard alkylating agents, ensures that the production of these intermediates is not bottlenecked by scarce reagents. The process tolerance to minor variations in temperature and reagent quality means that batch-to-batch consistency can be maintained even when sourcing raw materials from multiple vendors. This flexibility is paramount for building a resilient supply chain capable of withstanding geopolitical disruptions or logistics delays. Furthermore, the stability of the protected intermediates allows for the strategic stocking of key precursors, enabling rapid response to sudden increases in demand from downstream API manufacturers without compromising on quality.
- Scalability and Environmental Compliance: The transition from laboratory to commercial scale is facilitated by the moderate operating conditions of this process, which avoid the heat transfer limitations often encountered in highly exothermic reactions. The use of aqueous workups and standard filtration techniques for product isolation simplifies the engineering requirements for large-scale reactors and centrifuges. From an environmental perspective, the reduction in hazardous waste generation and the avoidance of toxic heavy metal catalysts align with modern green chemistry principles, helping manufacturers meet increasingly stringent environmental regulations. The process design inherently supports the implementation of solvent recovery systems, further reducing the environmental footprint and operational costs associated with solvent procurement and disposal.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this thienopyrrole synthesis technology. These insights are derived directly from the experimental data and process descriptions found in the patent literature, providing a factual basis for decision-making. Understanding these details is crucial for evaluating the feasibility of integrating this route into existing manufacturing workflows.
Q: How does this process improve safety compared to conventional thienopyrrole synthesis?
A: The patent explicitly addresses the explosion risks associated with prior art methods for preparing the acid precursors. By utilizing a protected intermediate pathway and mild base-mediated cyclization (e.g., sodium methylate in methanol), the process avoids hazardous high-energy intermediates, significantly enhancing operational safety for commercial production.
Q: What strategies are used to control impurities during the formylation step?
A: The process identifies the formation of amidine byproducts (Formula VII) as a key challenge during Vilsmeier-Haack formylation. The patent describes an optimized alternative step using specific solvent ratios and temperature controls (e.g., adding the substrate to the POCl3/DMF complex slowly) to suppress amidine formation and maximize the yield of the desired aldehyde intermediate.
Q: Is this synthetic route suitable for large-scale manufacturing?
A: Yes, the route is designed for scalability. It employs common organic solvents like DMF, methanol, and dichloromethane, and operates at moderate temperatures (ambient to reflux). The use of robust reagents like potassium carbonate and sodium methylate, along with standard workup procedures like filtration and chromatography, facilitates easy translation from laboratory to pilot and commercial plant scales.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Thienopyrrole Intermediate Supplier
At NINGBO INNO PHARMCHEM, we recognize that the successful commercialization of glycogen phosphorylase inhibitors depends on a partner who can deliver complex heterocyclic intermediates with unwavering consistency. Our technical team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from pilot plant to full-scale manufacturing is seamless and efficient. We adhere to stringent purity specifications and operate rigorous QC labs equipped with state-of-the-art analytical instrumentation to guarantee that every batch of thienopyrrole derivative meets the exacting standards required for pharmaceutical applications. Our commitment to quality assurance extends beyond simple compliance; we actively engage in continuous process improvement to enhance yield and reduce impurity levels further.
We invite you to collaborate with us to leverage this advanced synthetic technology for your next project. By engaging with our technical procurement team, you can request a Customized Cost-Saving Analysis tailored to your specific volume requirements and quality targets. We encourage potential partners to reach out for specific COA data and route feasibility assessments, allowing you to make informed decisions based on real-world performance metrics. Let us be your trusted ally in navigating the complexities of fine chemical synthesis, delivering value through innovation, reliability, and scientific excellence.
