Revolutionizing Taxane Precursor Production: A Facile One-Pot Synthesis for Commercial Scale-Up
Revolutionizing Taxane Precursor Production: A Facile One-Pot Synthesis for Commercial Scale-Up
The global demand for semi-synthetic taxanes, particularly paclitaxel and docetaxel, necessitates robust and scalable supply chains for their critical precursors. Patent CN1942459A introduces a transformative methodology for synthesizing C-7 protected baccatin III compounds, addressing longstanding bottlenecks in pharmaceutical intermediate manufacturing. This technology leverages a novel combination of secondary amines and nitrogen-containing compounds to facilitate selective protection under mild conditions. By eliminating the need for cryogenic temperatures and inert atmospheres, this process represents a paradigm shift towards greener and more cost-effective production. For R&D directors and procurement specialists, understanding the mechanistic advantages of this route is essential for optimizing supply chain resilience. The following analysis details how this innovation supports the commercial scale-up of complex pharmaceutical intermediates while maintaining stringent quality standards.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of 7-triethylsilyl baccatin III has been plagued by operational inefficiencies and harsh reaction conditions. Traditional protocols, such as those described by Denis et al., typically require the use of excess triethylchlorosilane and pyridine at strictly controlled low temperatures. These legacy methods often mandate reaction times extending up to 48 hours for acylation and 20 hours for silylation, creating significant throughput limitations. Furthermore, the necessity for cryogenic cooling, sometimes reaching -40°C, imposes a heavy burden on energy consumption and specialized reactor infrastructure. Multi-step procedures involving intermediate purification not only increase solvent waste but also lead to cumulative yield losses, making the final product economically challenging to produce at a commercial scale. These factors collectively hinder the ability of manufacturers to respond rapidly to market fluctuations in taxane demand.
The Novel Approach
In stark contrast, the method disclosed in CN1942459A utilizes a synergistic catalytic system comprising a secondary amine, preferably imidazole, and a nitrogen-containing compound like pyridine. This innovation enables the conversion of 10-deacetylbaccatin III to the desired C-7 protected derivative in a single pot without isolating intermediates. The reaction proceeds smoothly at ambient temperature, typically between 20°C and 25°C, removing the need for expensive cooling systems. Remarkably, the C-7 protection step achieves completion in less than 20 minutes, while the subsequent acylation finishes within 5 hours. This drastic reduction in processing time, coupled with the elimination of inert gas requirements, streamlines the workflow significantly. The result is a highly efficient pathway that enhances overall yield and simplifies the operational complexity for large-scale manufacturing facilities.
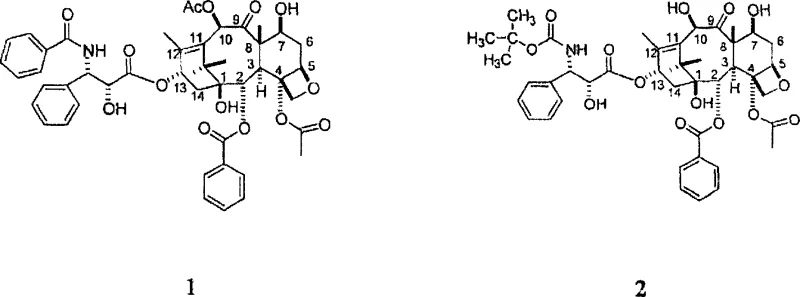
Mechanistic Insights into Imidazole-Promoted Selective Protection
The core of this technological breakthrough lies in the specific activation of the sterically hindered hydroxyl groups on the baccatin III core. In the presence of imidazole, the nucleophilicity of the C-7 hydroxyl group is enhanced relative to the C-10 and C-13 positions, allowing for highly selective silylation. The nitrogen-containing compound, acting as both solvent and base, stabilizes the transition state and scavenges the acid byproduct generated during silylation. This dual-amine system creates a reactive environment where the triethylchlorosilane reacts preferentially at the C-7 position before the acylating agent is introduced. Once the C-7 position is capped, the addition of acetic anhydride targets the remaining C-10 hydroxyl group. This sequential reactivity within a single vessel prevents the formation of over-protected byproducts or regioisomers that often complicate purification in traditional routes.
Impurity control is inherently superior in this one-pot design because the intermediate C-7 protected species is not exposed to external workup conditions that could lead to degradation or deprotection. The use of stoichiometric amounts of reagents, specifically about 2 equivalents of protecting agent and 6 equivalents of secondary amine, ensures complete conversion while minimizing excess reagent carryover. The reaction mixture remains stable even if stirred overnight, indicating a robust thermodynamic profile that tolerates minor variations in processing time. For quality assurance teams, this stability translates to a consistent impurity profile and reduced risk of batch failure. The mechanism effectively bypasses the kinetic traps associated with low-temperature reactions, ensuring that the desired 7-TES-10-acetyl baccatin III is formed with high fidelity.
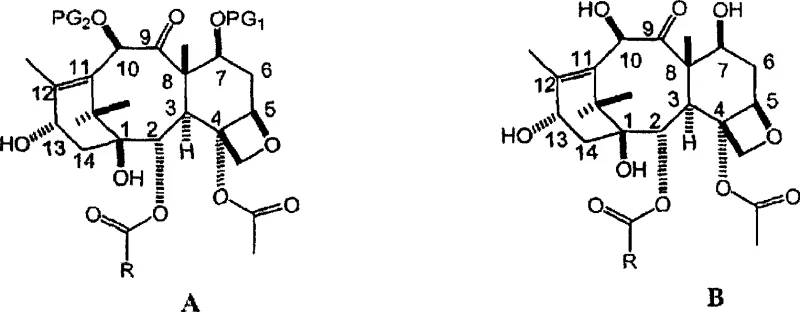
How to Synthesize C-7 Protected Baccatin III Efficiently
Implementing this synthesis requires precise adherence to the reagent ratios and addition sequences outlined in the patent to maximize yield and purity. The process begins by dissolving the starting material, 10-deacetylbaccatin III, in anhydrous pyridine, which serves as the reaction medium. It is critical to maintain an ambient atmosphere, as the method does not require nitrogen or argon blanketing, further simplifying the setup. The addition of imidazole initiates the activation phase, followed immediately by the dropwise addition of triethylchlorosilane to effect the C-7 protection. After a brief stirring period to ensure complete silylation, acetic anhydride is introduced to cap the C-10 position. Detailed standardized synthetic steps see the guide below.
- Dissolve 10-deacetylbaccatin III in anhydrous pyridine under ambient atmosphere without inert gas protection.
- Add imidazole (secondary amine) followed by triethylchlorosilane (protecting agent) to effect C-7 silylation.
- Introduce acetic anhydride to the mixture to acylate the C-10 hydroxyl group, completing the transformation in less than 5 hours.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this synthetic route offers tangible strategic benefits beyond mere chemical efficiency. The elimination of cryogenic cooling requirements directly translates to substantial cost reduction in API manufacturing by lowering utility expenses and reducing the need for specialized low-temperature reactors. Furthermore, the one-pot nature of the reaction minimizes solvent consumption and waste generation, aligning with increasingly strict environmental regulations and sustainability goals. The shortened reaction cycle time allows for higher throughput per batch, effectively increasing production capacity without additional capital investment in new equipment. These factors combine to create a more resilient supply chain capable of meeting volatile market demands for taxane intermediates with greater agility and lower operational risk.
- Cost Reduction in Manufacturing: The removal of energy-intensive cooling steps and the reduction in reaction time significantly lower the operational expenditure per kilogram of product. By avoiding multi-step isolation and purification processes, labor costs and solvent recovery expenses are drastically minimized. The use of common, inexpensive reagents like imidazole and acetic anhydride instead of exotic catalysts further drives down the raw material bill. Consequently, the overall cost of goods sold (COGS) for the protected baccatin intermediate is optimized, providing a competitive pricing advantage in the global marketplace.
- Enhanced Supply Chain Reliability: Operating at ambient temperature and pressure reduces the dependency on complex utility infrastructure, making the process less susceptible to facility disruptions. The robustness of the reaction conditions ensures consistent batch-to-batch quality, reducing the likelihood of production delays caused by out-of-specification results. Additionally, the scalability of the method from laboratory to commercial production ensures that supply volumes can be ramped up quickly to satisfy urgent orders from downstream API manufacturers. This reliability is crucial for maintaining uninterrupted production schedules for life-saving oncology drugs.
- Scalability and Environmental Compliance: The simplified workup procedure, involving standard extraction and crystallization, facilitates easy scale-up to multi-ton production levels. The reduction in solvent usage and waste generation supports compliance with green chemistry principles and local environmental discharge limits. By minimizing the number of unit operations, the potential for safety incidents related to hazardous material handling is also reduced. This makes the process not only economically attractive but also socially responsible, appealing to partners who prioritize sustainable manufacturing practices in their supply chain audits.
Frequently Asked Questions (FAQ)
The following questions address common technical inquiries regarding the implementation and optimization of this synthesis method. These answers are derived directly from the experimental data and claims presented in the patent documentation to ensure accuracy and relevance for technical stakeholders. Understanding these nuances is vital for process engineers evaluating the feasibility of technology transfer. The responses cover critical aspects such as reaction conditions, reagent selection, and product quality parameters.
Q: Does this synthesis method require cryogenic cooling?
A: No, unlike conventional methods requiring temperatures as low as -40°C, this patented process operates efficiently at ambient temperature (20-25°C), significantly reducing energy consumption and equipment complexity.
Q: What is the reaction time for the C-7 protection step?
A: The C-7 protection step is exceptionally fast, completing in less than 20 minutes with quantitative yield, while the full dual-protection sequence finishes within 5 hours.
Q: Is intermediate purification necessary between steps?
A: No, the process is designed as a one-pot synthesis where the C-7 protected intermediate is not isolated, thereby minimizing material loss and solvent usage.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Baccatin III Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-quality intermediates play in the successful development of oncology therapeutics. Our technical team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your supply needs are met with precision and consistency. We are committed to delivering products that meet stringent purity specifications through our rigorous QC labs and advanced analytical capabilities. By leveraging advanced synthetic methodologies like the one described in CN1942459A, we can offer superior value propositions to our global partners. Our facility is equipped to handle complex chemistries safely and efficiently, guaranteeing a steady flow of materials for your drug development pipelines.
We invite you to engage with our technical procurement team to discuss how our manufacturing capabilities can support your specific project requirements. Request a Customized Cost-Saving Analysis to understand the economic benefits of switching to our optimized supply chain solutions. We are ready to provide specific COA data and route feasibility assessments to demonstrate our commitment to quality and transparency. Let us be your partner in navigating the complexities of pharmaceutical intermediate sourcing with confidence and reliability.
