Advanced Metal Alkoxide Technology for Scalable Taxol and Taxotere Intermediate Production
Advanced Metal Alkoxide Technology for Scalable Taxol and Taxotere Intermediate Production
The pharmaceutical industry has long recognized the immense therapeutic value of taxanes, particularly Taxol (paclitaxel) and Taxotere (docetaxel), as potent anti-cancer agents. However, the commercial viability of these life-saving drugs has historically been constrained by the complexity and cost of their synthesis. Patent CN1075718A introduces a transformative approach to this challenge by detailing a novel class of metal alkoxides derived from baccatin III and 10-deacetylbaccatin III. This technology addresses the critical bottleneck in taxane synthesis: the efficient and stereoselective attachment of the C-13 beta-amido ester side chain. By leveraging organometallic chemistry, specifically the formation of C-13 metal alkoxides, this method offers a robust pathway for producing high-purity pharmaceutical intermediates. For R&D directors and procurement specialists, understanding this mechanistic breakthrough is essential for securing a reliable pharmaceutical intermediate supplier capable of meeting the rigorous demands of oncology drug manufacturing.
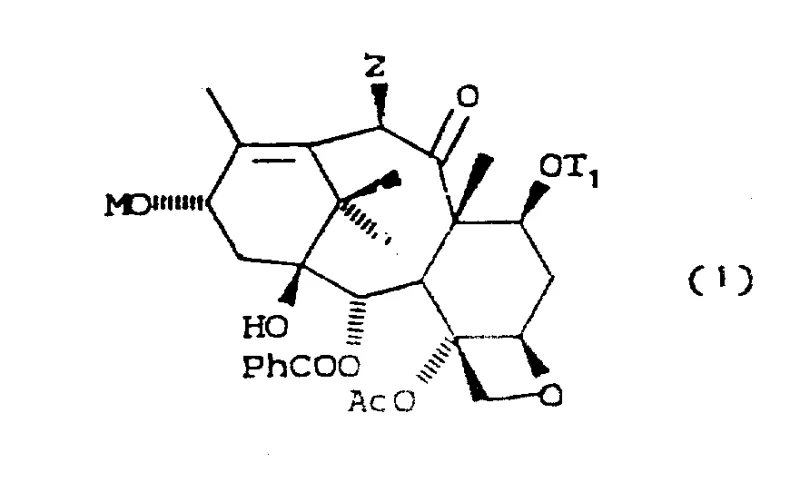
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the semi-synthesis of Taxol from natural precursors like 10-deacetylbaccatin III has faced significant hurdles regarding yield and stereochemical control. Conventional methods, such as those described by Greene et al., typically involve activating the C-13 hydroxyl group followed by coupling with a beta-lactam side chain. A major drawback of these traditional routes is the frequent formation of isomeric mixtures during the coupling step. Because the side chain precursors often exist as racemic mixtures, and the coupling reaction lacks sufficient diastereoselectivity, manufacturers are forced to undertake tedious and costly separation processes to isolate the biologically active (2'R, 3'S) isomer. Furthermore, many existing protocols require multi-step protection and deprotection sequences that degrade the overall yield, making cost reduction in API manufacturing difficult to achieve without compromising quality or supply continuity.
The Novel Approach
The methodology disclosed in CN1075718A represents a paradigm shift by utilizing a metal alkoxide intermediate to drive the coupling reaction. Instead of relying on less reactive alcohol derivatives, the process converts the C-13 hydroxyl group of the baccatin nucleus directly into a highly nucleophilic metal alkoxide using organometallic reagents like n-Butyl Lithium. This activated species reacts with the beta-lactam ring with exceptional stereocontrol. As illustrated in the reaction schemes, this approach effectively bypasses the need for pre-resolving the racemic beta-lactam precursors. The reaction inherently favors the formation of the desired diastereomer, thereby eliminating the expensive chromatographic separation steps that plague older methods. This streamlined workflow not only accelerates production timelines but also drastically reduces the consumption of raw materials and solvents, offering substantial advantages for commercial scale-up of complex pharmaceutical intermediates.
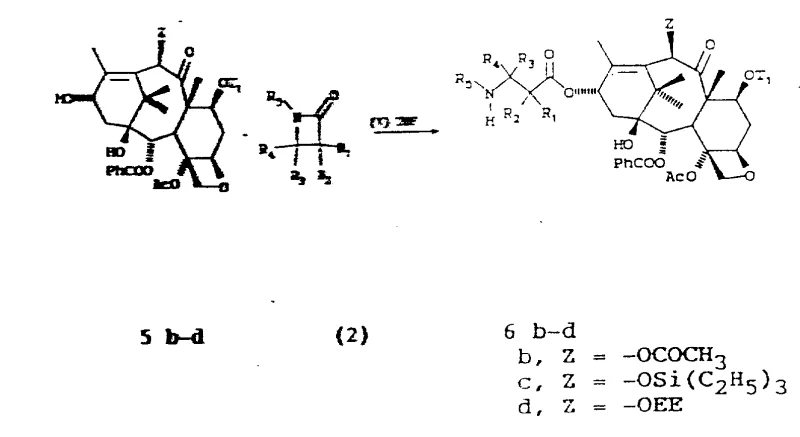
Mechanistic Insights into Organolithium-Mediated Side Chain Coupling
The core innovation of this patent lies in the generation and reactivity of the C-13 metal alkoxide. The process begins with the selective protection of the C-7 and C-10 hydroxyl groups, typically using triethylsilyl chloride, to prevent unwanted side reactions. Subsequently, the C-13 hydroxyl is deprotonated using a strong base, preferably an organolithium compound such as n-Butyl Lithium, in an aprotic solvent like tetrahydrofuran (THF) at low temperatures ranging from -78°C to -45°C. This generates a lithium alkoxide species that acts as a potent nucleophile. When this species encounters the beta-lactam ring, it attacks the carbonyl carbon, leading to the opening of the four-membered lactam ring and the formation of the crucial beta-amido ester bond. The steric environment created by the bulky silyl protecting groups and the specific coordination of the lithium ion directs the attack to occur with high facial selectivity, ensuring that the resulting side chain adopts the correct (2'R, 3'S) configuration required for biological activity.
From an impurity control perspective, this mechanism is superior because it minimizes the formation of the (2'S, 3'R) diastereomer, which is a common impurity in non-selective couplings. The patent data indicates that even when starting with a racemic mixture of beta-lactams, the reaction yields the desired isomer as the major product, often accompanied by only minor amounts of the undesired isomer which can be easily removed. This inherent selectivity reduces the burden on downstream purification units. Furthermore, the use of mild deprotection conditions, such as aqueous HF or acetic acid, ensures that the sensitive oxetane ring and the ester linkages within the taxane core remain intact, preserving the structural integrity of the final API intermediate. This level of chemical precision is vital for maintaining the stringent purity specifications required by global regulatory bodies.
How to Synthesize Metal Alkoxide Taxol Intermediates Efficiently
The synthesis protocol outlined in the patent provides a clear roadmap for implementing this technology in a GMP environment. The process is designed to be operationally simple, utilizing readily available reagents and standard laboratory equipment, which facilitates easy technology transfer from pilot scale to full commercial production. The key to success lies in the strict control of reaction temperatures and the stoichiometry of the organometallic reagent to ensure complete conversion of the baccatin substrate without over-reaction. Detailed standardized synthetic steps for this process are provided in the guide below.
- Protect the C-7 and C-10 hydroxyl groups of 10-deacetylbaccatin III using triethylsilyl chloride to form the disilyl ether derivative.
- Generate the C-13 metal alkoxide by treating the protected baccatin derivative with n-Butyl Lithium in THF at low temperatures (-45°C to -78°C).
- React the resulting metal alkoxide with a racemic beta-lactam side chain precursor to form the coupled intermediate with high diastereoselectivity.
- Perform final deprotection using mild acidic conditions or fluoride sources to yield the biologically active Taxol or Taxotere derivative.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this metal alkoxide technology translates into tangible strategic benefits beyond mere chemical elegance. The primary advantage is the decoupling of the side chain supply from chiral resolution constraints. Since the process tolerates racemic beta-lactam precursors, suppliers can source these critical starting materials from a broader range of vendors who may not possess specialized chiral separation capabilities. This diversification of the supply base significantly enhances supply chain reliability and mitigates the risk of shortages that often occur when relying on single-source chiral providers. Additionally, the elimination of resolution steps reduces the overall material throughput required, leading to significant cost optimization in the procurement of raw materials.
- Cost Reduction in Manufacturing: The ability to use racemic mixtures directly eliminates the need for expensive chiral chromatography or enzymatic resolution processes, which are traditionally the most cost-intensive stages of side chain production. By removing these unit operations, the overall manufacturing cost is significantly reduced. Furthermore, the higher yields associated with the metal alkoxide coupling mean that less starting baccatin III is wasted, maximizing the value extracted from every kilogram of this precious natural precursor. The simplified workflow also reduces labor hours and utility consumption, contributing to a leaner and more cost-effective production model.
- Enhanced Supply Chain Reliability: Racemic beta-lactams are chemically simpler and faster to synthesize than their enantiopure counterparts. This means that lead times for raw material delivery can be drastically shortened. In a volatile global market, having access to a wider pool of suppliers for achiral intermediates provides a buffer against geopolitical or logistical disruptions. The robustness of the reaction conditions, which tolerate standard industrial solvents and reagents, further ensures that production schedules can be maintained consistently without the delays often caused by finicky, low-yielding chemical transformations.
- Scalability and Environmental Compliance: The process utilizes common organometallic reagents and solvents that are well-understood in large-scale chemical manufacturing, facilitating straightforward scale-up from kilograms to metric tons. The reduction in processing steps inherently lowers the volume of chemical waste generated, aligning with modern green chemistry principles and environmental compliance standards. Fewer purification steps mean less solvent usage and lower energy requirements for distillation and drying, resulting in a smaller environmental footprint and reduced costs associated with waste disposal and treatment.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this metal alkoxide synthesis route. These answers are derived directly from the experimental data and claims presented in the patent documentation, providing a factual basis for evaluating the technology's fit within your existing manufacturing portfolio. Understanding these details is crucial for making informed decisions about process adoption and vendor qualification.
Q: Can racemic beta-lactam precursors be used in this synthesis?
A: Yes, a key advantage of the metal alkoxide method described in CN1075718A is its high diastereoselectivity, which allows for the direct use of racemic beta-lactam mixtures without the need for prior chiral resolution, significantly simplifying the supply chain.
Q: What metals are suitable for forming the alkoxide intermediate?
A: The patent specifies that metals from Group IA, IIA, or transition metals can be used, with Lithium (via n-Butyl Lithium), Magnesium, Sodium, Potassium, or Titanium being preferred for optimal reactivity and yield.
Q: How does this method improve upon traditional Taxol synthesis routes?
A: Traditional methods often suffer from low yields and difficult separation of isomeric byproducts during the C-13 side chain attachment. This metal alkoxide approach minimizes isomer formation and eliminates expensive purification steps, enhancing overall process efficiency.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Baccatin III Derivative Supplier
At NINGBO INNO PHARMCHEM, we recognize that the transition from patent literature to commercial reality requires deep technical expertise and unwavering commitment to quality. As a leading CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the sophisticated metal alkoxide chemistry described in CN1075718A can be executed with precision at any volume. Our facilities are equipped with state-of-the-art cryogenic reactors capable of handling the low-temperature organolithium reactions required for this process, and our rigorous QC labs enforce stringent purity specifications to guarantee that every batch of Baccatin III derivative meets the highest international standards for oncology APIs.
We invite you to collaborate with us to leverage this advanced technology for your taxane projects. Our technical team is ready to provide a Customized Cost-Saving Analysis tailored to your specific production volumes and quality requirements. We encourage you to contact our technical procurement team today to request specific COA data and route feasibility assessments, allowing us to demonstrate how our optimized metal alkoxide process can enhance your supply chain efficiency and reduce your overall cost of goods sold.
