Advanced Pd-Catalyzed Synthesis of Carbonyl-Bridged Biheterocyclic Compounds for Commercial Scale-Up
The pharmaceutical and fine chemical industries are constantly seeking robust methodologies to construct complex heterocyclic scaffolds efficiently. Patent CN115353511A introduces a groundbreaking multi-component method for synthesizing carbonyl-bridged biheterocyclic compounds, specifically targeting the indolinone-imidazole structural motif which is prevalent in bioactive molecules. This innovation addresses the critical need for safer, more scalable synthetic routes by employing a transition metal palladium-catalyzed carbonylation cascade reaction. Unlike traditional approaches that often rely on hazardous gaseous reagents, this protocol utilizes a liquid CO surrogate system, thereby mitigating significant safety risks associated with high-pressure carbon monoxide handling. The reaction operates under remarkably mild conditions, typically at 30°C, which preserves sensitive functional groups and minimizes energy consumption during the manufacturing process. Furthermore, the use of readily available starting materials such as trifluoroethylimidoyl chloride and propargylamine ensures that the supply chain remains resilient and cost-effective for large-scale production demands.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the construction of carbonyl-bridged biheterocyclic systems has been plagued by significant synthetic challenges that hinder efficient commercial production. Conventional methods often necessitate the direct use of toxic carbon monoxide gas, which requires specialized high-pressure equipment and rigorous safety protocols that drastically increase capital expenditure and operational complexity. Additionally, many existing protocols suffer from poor substrate compatibility, where the presence of electron-withdrawing or bulky substituents leads to diminished yields or complete reaction failure. The reliance on harsh reaction conditions, such as elevated temperatures or strong bases, frequently results in the formation of unwanted by-products and complicates the downstream purification process. These factors collectively contribute to extended lead times and inconsistent batch quality, posing severe risks for procurement managers aiming to secure reliable pharmaceutical intermediates supplier partnerships. Consequently, the industry has long awaited a methodology that balances high efficiency with operational safety and economic viability.
The Novel Approach
The methodology disclosed in CN115353511A represents a paradigm shift by replacing hazardous gaseous CO with a safe and manageable formic acid/acetic anhydride mixture. This innovative system generates carbon monoxide in situ, allowing the carbonylation reaction to proceed smoothly at atmospheric pressure and a mild temperature of 30°C. The use of a palladium chloride catalyst paired with a trifurylphosphine ligand creates a highly active catalytic species that facilitates the multi-component coupling with exceptional precision. This approach not only eliminates the need for expensive high-pressure reactors but also significantly simplifies the workflow, making it accessible for both laboratory research and industrial manufacturing. The reaction demonstrates excellent functional group tolerance, accommodating diverse substituents on the aromatic rings without compromising the integrity of the final product. By streamlining the synthesis into a one-pot procedure, this novel approach drastically reduces solvent usage and waste generation, aligning perfectly with modern green chemistry principles and cost reduction in fine chemical manufacturing goals.
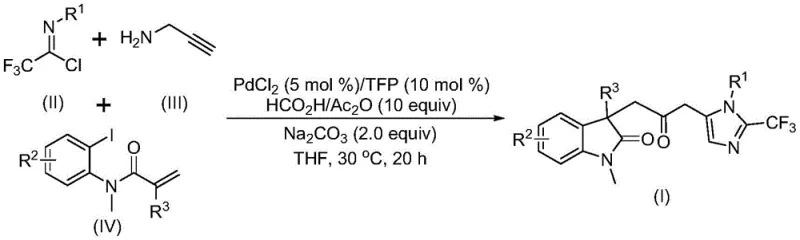
Mechanistic Insights into Pd-Catalyzed Carbonylation Cascade
Understanding the mechanistic pathway is crucial for R&D directors evaluating the feasibility of this technology for high-purity pharmaceutical intermediates. The reaction likely initiates with the oxidative addition of zero-valent palladium into the carbon-iodine bond of the acrylamide substrate, forming a key organopalladium intermediate. This is followed by an intramolecular Heck-type reaction that constructs the initial heterocyclic ring system, setting the stage for the subsequent carbonylation step. The carbon monoxide generated from the formic acid and acetic anhydride mixture then inserts into the palladium-carbon bond, creating an acyl-palladium species that is essential for bridging the two heterocyclic units. Simultaneously, the trifluoroethylimidoyl chloride reacts with propargylamine in a base-promoted manner to form a trifluoroacetamidine intermediate, which subsequently undergoes isomerization. The final cyclization is driven by the nucleophilic attack of this amidine species onto the activated acyl-palladium complex, releasing the product and regenerating the catalyst. This intricate dance of bond formations occurs with high regioselectivity, ensuring that the carbonyl bridge is positioned exactly as required for biological activity.
Impurity control is inherently built into this mechanism due to the mild reaction conditions and the specific selectivity of the palladium catalyst. By operating at 30°C, the thermal energy available for side reactions such as polymerization or decomposition of sensitive intermediates is minimized. The use of sodium carbonate as a mild base further prevents the hydrolysis of the imidoyl chloride or other acid-sensitive functionalities that might be present on the substrate. The compatibility with various substituents, including halogens and trifluoromethyl groups, indicates that the catalytic cycle is robust against electronic variations in the starting materials. This stability translates directly to a cleaner crude reaction profile, reducing the burden on purification teams and increasing the overall yield of the desired high-purity OLED material or API intermediate. For supply chain heads, this predictability means fewer batch failures and a more consistent supply of critical building blocks for downstream drug synthesis.
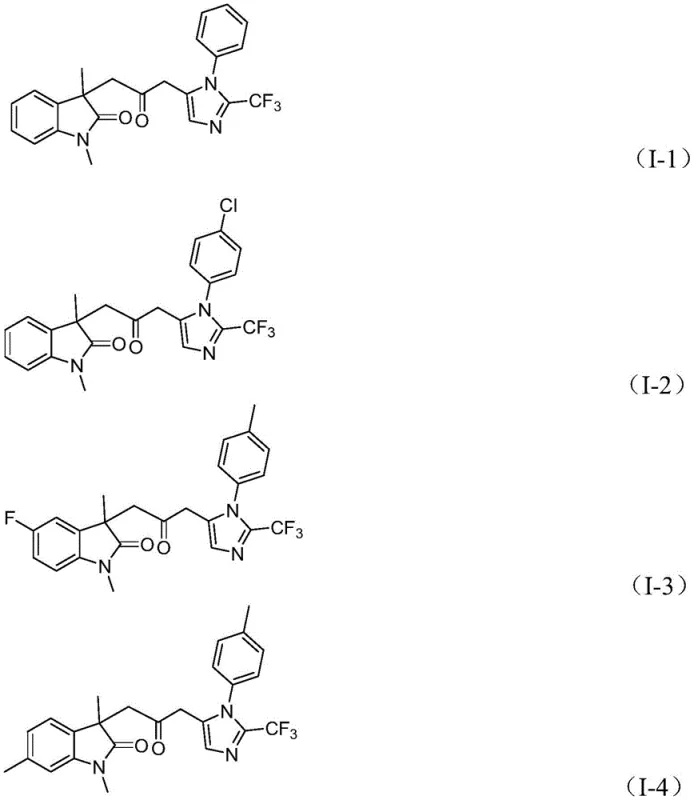
How to Synthesize Carbonyl-Bridged Biheterocyclic Compounds Efficiently
To implement this synthesis effectively, operators must adhere to precise stoichiometric ratios and mixing protocols to ensure optimal catalyst performance. The process begins with the preparation of the catalytic system, where palladium chloride and trifurylphosphine are combined with the CO surrogate mixture in a dry aprotic solvent like tetrahydrofuran. Substrates including trifluoroethylimidoyl chloride, propargylamine, and the specific acrylamide derivative are then introduced sequentially or as a mixture, depending on the specific scale and equipment available. Maintaining the temperature strictly at 30°C is vital, as deviations could alter the rate of CO generation or affect the stability of the palladium intermediates. After the reaction period of 12 to 20 hours, the mixture is filtered to remove inorganic salts, and the product is isolated through standard silica gel chromatography. Detailed standardized synthesis steps are provided in the guide below to ensure reproducibility across different manufacturing sites.
- Mix palladium chloride, trifurylphosphine, sodium carbonate, and a formic acid/acetic anhydride mixture in an organic solvent like THF.
- Add trifluoroethylimidoyl chloride, propargylamine, and the acrylamide substrate to the reaction vessel under stirring.
- Maintain the reaction at 30°C for 12 to 20 hours, then filter and purify via column chromatography to isolate the target compound.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, this patent offers substantial benefits that directly address the pain points of procurement managers and supply chain heads in the specialty chemical sector. The elimination of toxic carbon monoxide gas removes a major regulatory and safety hurdle, allowing for production in facilities that may not be equipped for high-pressure gas handling. This flexibility expands the pool of potential manufacturing partners and reduces the logistical costs associated with transporting hazardous materials. Furthermore, the use of commercially available and inexpensive starting materials ensures that the raw material costs remain stable and predictable, shielding buyers from volatile market fluctuations. The simplicity of the post-treatment process, involving basic filtration and chromatography, minimizes the consumption of solvents and consumables, contributing to significant cost savings in the overall production budget. These factors combine to create a supply chain that is not only more resilient but also capable of delivering high-quality intermediates with reduced lead time for high-purity intermediates.
- Cost Reduction in Manufacturing: The replacement of expensive and hazardous CO gas cylinders with a liquid formic acid/anhydride system drastically lowers the infrastructure and operational costs required for safe production. By avoiding the need for specialized high-pressure reactors and complex gas monitoring systems, manufacturers can allocate resources more efficiently towards quality control and capacity expansion. The high atom economy of the multi-component reaction ensures that a larger proportion of the starting materials are converted into the final product, reducing waste disposal costs. Additionally, the mild reaction conditions reduce energy consumption for heating and cooling, further contributing to the overall economic efficiency of the process. These cumulative savings allow for more competitive pricing strategies without compromising on the quality or purity of the final chemical products.
- Enhanced Supply Chain Reliability: The reliance on readily available commodity chemicals like propargylamine and acrylamide derivatives ensures that the supply chain is not vulnerable to shortages of exotic or proprietary reagents. Since the reaction tolerates a wide range of functional groups, manufacturers can source substrates from multiple vendors without needing to revalidate the entire process for minor structural variations. The robustness of the catalytic system means that batch-to-batch variability is minimized, providing procurement teams with the confidence to plan long-term production schedules. This stability is crucial for maintaining continuous operations in the pharmaceutical industry, where interruptions in the supply of key intermediates can delay clinical trials or commercial launches. Consequently, partnering with suppliers utilizing this technology offers a strategic advantage in securing a steady flow of materials.
- Scalability and Environmental Compliance: The patent explicitly demonstrates the feasibility of scaling this reaction to gram levels, indicating a clear path towards kilogram and ton-scale commercial production. The use of THF as a solvent, which is easily recoverable and recyclable, aligns with increasingly stringent environmental regulations regarding volatile organic compound emissions. The absence of heavy metal waste streams, beyond the minimal palladium catalyst which can be recovered, simplifies the wastewater treatment process and reduces the environmental footprint of the manufacturing site. This compliance with green chemistry standards enhances the corporate social responsibility profile of the supply chain, appealing to end-clients who prioritize sustainability. Ultimately, the ease of scale-up ensures that demand surges can be met promptly without the need for extensive process re-engineering or new facility construction.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this carbonylation technology. These answers are derived directly from the experimental data and beneficial effects described in the patent documentation to provide clarity for decision-makers. Understanding these details helps in assessing the fit of this technology within existing production workflows and quality assurance frameworks. The responses cover aspects ranging from safety improvements to substrate flexibility and scalability potential.
Q: How does this method improve safety compared to traditional carbonylation?
A: Unlike conventional methods requiring toxic carbon monoxide gas cylinders, this patent utilizes a formic acid and acetic anhydride mixture to generate CO in situ, significantly enhancing operational safety.
Q: What is the substrate compatibility of this Pd-catalyzed reaction?
A: The process demonstrates broad functional group tolerance, accommodating various substituents such as halogens, alkyl groups, and trifluoromethyl groups on the aromatic rings without compromising yield.
Q: Is this synthesis route suitable for industrial scale-up?
A: Yes, the patent explicitly confirms that the reaction can be expanded to gram-scale levels with simple post-treatment procedures, indicating strong potential for commercial manufacturing.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Carbonyl-Bridged Biheterocyclic Compounds Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of the synthetic route described in CN115353511A for producing advanced pharmaceutical intermediates. As a leading CDMO expert, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your project transitions smoothly from benchtop discovery to full-scale manufacturing. Our facilities are equipped with state-of-the-art rigorous QC labs capable of meeting stringent purity specifications required by global regulatory bodies. We understand that consistency is key in the fine chemical industry, and our dedicated technical team is committed to optimizing every parameter of this palladium-catalyzed process to maximize yield and minimize impurities. By leveraging our expertise, you can accelerate your development timelines and secure a reliable source of high-quality biheterocyclic building blocks.
We invite you to collaborate with us to explore how this innovative chemistry can enhance your product portfolio and reduce your overall manufacturing costs. Please contact our technical procurement team to request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We are ready to provide specific COA data and comprehensive route feasibility assessments to demonstrate our capability to deliver excellence. Let us be your partner in navigating the complexities of modern chemical synthesis and achieving your commercial goals efficiently.
