Advanced Pd-Catalyzed Multicomponent Synthesis Of Carbonyl-Bridged Biheterocyclic Compounds For Commercial Scale-Up
Advanced Pd-Catalyzed Multicomponent Synthesis Of Carbonyl-Bridged Biheterocyclic Compounds For Commercial Scale-Up
The pharmaceutical and agrochemical industries are constantly seeking robust synthetic methodologies that can construct complex molecular architectures with high efficiency and safety. Patent CN115353511A introduces a groundbreaking preparation method for carbonyl-bridged biheterocyclic compounds, specifically targeting the synthesis of indolinone-imidazole derivatives which are prevalent in bioactive molecules. This technology represents a significant leap forward by utilizing a transition metal palladium-catalyzed carbonylation cascade reaction that operates under remarkably mild conditions. By employing cheap and readily available starting materials such as trifluoroethylimidoyl chloride, propargylamine, and acrylamide derivatives, the process eliminates the need for hazardous carbon monoxide gas, thereby addressing critical safety concerns in modern chemical manufacturing. The invention not only simplifies the operational procedure but also demonstrates exceptional substrate compatibility, allowing for the diverse substitution patterns required in drug discovery and development pipelines.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditionally, the synthesis of carbonyl-bridged biheterocyclic compounds has been fraught with significant challenges that hinder efficient production and scalability. Conventional literature reports primarily rely on three strategies: direct coupling of two heterocycle substrates, oxidative cyclization involving activated methyl-substituted heterocycles, or transition metal-catalyzed tandem reactions that often require harsh conditions. A major bottleneck in existing carbonylation protocols is the reliance on exogenous carbon monoxide gas, which is highly toxic, flammable, and requires specialized high-pressure equipment and rigorous safety protocols to handle. Furthermore, many traditional methods suffer from poor atom economy, limited functional group tolerance, and the necessity for expensive or difficult-to-prepare substrates, which collectively drive up the cost of goods and extend the lead time for high-purity pharmaceutical intermediates. These limitations make it difficult to apply carbonylation reactions efficiently to the synthesis of complex biheterocycles on an industrial scale without incurring substantial infrastructure and compliance costs.
The Novel Approach
In stark contrast to these legacy methods, the novel approach disclosed in the patent utilizes a sophisticated multicomponent reaction design that elegantly bypasses the need for external carbon monoxide sources. By integrating a formic acid and acetic anhydride mixture directly into the reaction system, carbon monoxide is generated in situ, serving as a safe and effective carbonyl source for the palladium-catalyzed transformation. This method allows for the one-pot construction of multiple chemical bonds, including carbon-carbon and carbon-nitrogen bonds, through a seamless cascade sequence involving Heck reaction and cyclization steps. The process operates at a mild temperature of 30°C in tetrahydrofuran (THF), which drastically reduces energy consumption compared to high-temperature alternatives. Moreover, the use of commercially available palladium chloride and trifurylphosphine ensures that the catalytic system is both cost-effective and robust, enabling the synthesis of diversified substituted double heterocyclic compounds with trifluoromethyl and carbonyl groups through simple substrate design.
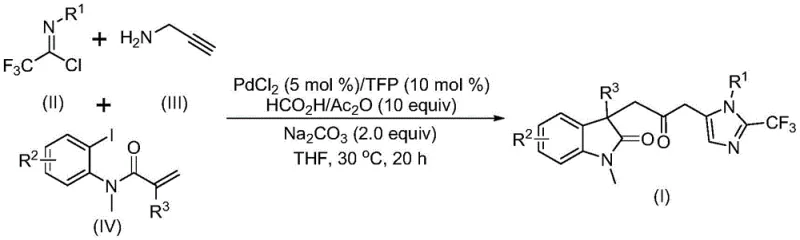
Mechanistically, this transformation is a testament to the power of modern organometallic chemistry in streamlining complex syntheses. The reaction likely initiates with the insertion of zero-valent palladium into the carbon-iodine bond of the acrylamide substrate, followed by an intramolecular Heck reaction to generate a divalent alkyl palladium intermediate. Subsequently, under the influence of the carbon monoxide released from the formic acid/acetic anhydride mixture, a carbonylation event occurs to yield an acyl palladium intermediate. Concurrently, a base-promoted intermolecular carbon-nitrogen bond formation between the trifluoroethylimidoyl chloride and propargylamine generates a trifluoroacetamidine compound, which undergoes isomerization. The final step involves the activation of this trifluoroacetamidine by the acyl palladium intermediate, catalyzing an intramolecular cyclization that furnishes the final carbonyl-bridged biheterocyclic product. This intricate dance of catalytic cycles ensures high regioselectivity and minimizes the formation of unwanted byproducts, which is crucial for maintaining a clean impurity profile in pharmaceutical manufacturing.
From an impurity control perspective, the mild reaction conditions and the specific choice of ligands play a pivotal role in ensuring product purity. The use of trifurylphosphine as a ligand helps stabilize the palladium species, preventing the formation of palladium black and other inactive aggregates that could lead to incomplete reactions or side products. Additionally, the in situ generation of carbon monoxide avoids the fluctuations in gas pressure that can occur with external gas feeds, leading to more consistent reaction kinetics and reproducible results across different batches. The compatibility with various functional groups, such as halogens, alkyls, alkoxy, and nitro groups on the aromatic rings, means that the process can tolerate a wide range of substituents without requiring extensive protecting group strategies. This inherent selectivity reduces the complexity of downstream purification, allowing for simpler workup procedures involving filtration and silica gel column chromatography to achieve the desired high-purity specifications required for clinical applications.
How to Synthesize Carbonyl-Bridged Biheterocyclic Compounds Efficiently
To implement this synthesis effectively, operators must adhere to precise stoichiometric ratios and mixing protocols to maximize yield and minimize waste. The standard procedure involves charging a reaction vessel with palladium chloride, trifurylphosphine, sodium carbonate, and the formic acid/acetic anhydride mixture in an organic solvent, followed by the sequential addition of the trifluoroethylimidoyl chloride, propargylamine, and acrylamide substrates. The detailed standardized synthesis steps, including specific molar ratios and purification techniques validated by the patent data, are outlined below to ensure reproducibility and safety during scale-up operations.
- Combine palladium chloride, trifurylphosphine, sodium carbonate, and a formic acid/acetic anhydride mixture in an organic solvent like THF.
- Add trifluoroethylimidoyl chloride, propargylamine, and the acrylamide substrate to the reaction mixture under stirring.
- Maintain the reaction at 30°C for 12 to 20 hours, then filter and purify via column chromatography to isolate the target biheterocyclic compound.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this patented methodology offers tangible strategic advantages that extend beyond mere chemical novelty. The shift away from toxic carbon monoxide gas to a liquid-phase CO surrogate fundamentally alters the risk profile of the manufacturing process, eliminating the need for specialized gas handling infrastructure and reducing insurance and compliance overheads. This transition not only enhances workplace safety but also streamlines the regulatory approval process for new manufacturing sites, as the hazards associated with high-pressure toxic gases are completely removed from the production floor. Furthermore, the reliance on cheap and readily available starting materials ensures a stable supply chain, mitigating the risks associated with sourcing exotic or proprietary reagents that might be subject to market volatility or geopolitical disruptions.
- Cost Reduction in Manufacturing: The economic benefits of this process are driven by the elimination of expensive safety infrastructure and the use of commodity chemicals. By avoiding the need for high-pressure reactors and toxic gas monitoring systems, capital expenditure for new production lines is significantly reduced. Additionally, the mild reaction temperature of 30°C implies a drastic reduction in energy consumption for heating and cooling compared to traditional high-temperature processes, leading to lower utility costs per kilogram of product. The high atom economy of the multicomponent reaction means that fewer raw materials are wasted, and the simplified purification process reduces the consumption of solvents and silica gel, further driving down the overall cost of goods sold for these complex intermediates.
- Enhanced Supply Chain Reliability: The robustness of this synthetic route contributes directly to supply chain resilience. Since the key reagents such as propargylamine, acrylamide derivatives, and trifluoroethylimidoyl chloride are commercially available or easily synthesized from common precursors, the risk of raw material shortages is minimized. The broad substrate scope allows for flexibility in sourcing; if a specific substituted acrylamide is unavailable, alternative analogs can often be substituted without re-optimizing the entire process. This flexibility ensures continuous production capability even when facing minor disruptions in the supply of specific building blocks, thereby guaranteeing consistent delivery schedules to downstream customers.
- Scalability and Environmental Compliance: Scaling this process from gram-level to industrial tonnage is facilitated by the homogeneous nature of the reaction and the absence of gas-liquid mass transfer limitations typically associated with CO gas carbonylations. The liquid-phase generation of carbon monoxide ensures uniform concentration throughout the reactor, which is critical for maintaining consistent quality during scale-up. From an environmental standpoint, the process generates less hazardous waste compared to methods using heavy metal oxidants or toxic gases, aligning with green chemistry principles. The ability to produce high-purity products with minimal byproducts reduces the burden on waste treatment facilities, helping manufacturers meet increasingly stringent environmental regulations without compromising production throughput.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis technology. These answers are derived directly from the experimental data and beneficial effects described in the patent documentation, providing clarity on reaction parameters, safety profiles, and scalability potential for stakeholders evaluating this technology for integration into their manufacturing portfolios.
Q: What is the primary safety advantage of this synthesis method compared to traditional carbonylation?
A: Unlike conventional methods that require handling toxic and high-pressure carbon monoxide gas, this protocol utilizes a formic acid and acetic anhydride mixture to generate carbon monoxide in situ, significantly enhancing operational safety and reducing regulatory compliance costs.
Q: What are the typical reaction conditions and catalysts used in this process?
A: The reaction employs a palladium chloride (PdCl2) catalyst with trifurylphosphine (TFP) as a ligand in tetrahydrofuran (THF) solvent, operating at a mild temperature of 30°C for 12 to 20 hours to ensure high conversion rates.
Q: Is this method suitable for large-scale industrial production?
A: Yes, the patent explicitly demonstrates the scalability of this method to gram-level synthesis with excellent substrate compatibility and functional group tolerance, making it highly viable for commercial scale-up of complex pharmaceutical intermediates.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Carbonyl-Bridged Biheterocyclic Compounds Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of this palladium-catalyzed multicomponent synthesis in accelerating the development of next-generation therapeutics. As a leading CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your transition from laboratory discovery to market supply is seamless and efficient. Our state-of-the-art facilities are equipped to handle complex organometallic reactions with the highest standards of safety and quality, supported by rigorous QC labs that enforce stringent purity specifications to meet the demanding requirements of global pharmaceutical clients.
We invite you to leverage our technical expertise to optimize your supply chain for these valuable intermediates. Our team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements, demonstrating how this innovative route can reduce your overall manufacturing expenses. Please contact our technical procurement team today to request specific COA data for our catalog items or to discuss route feasibility assessments for your custom synthesis projects, ensuring a reliable partnership for your long-term growth.
