Efficient Palladium-Catalyzed Synthesis of Carbonyl-Bridged Biheterocyclic Compounds for Drug Discovery
Efficient Palladium-Catalyzed Synthesis of Carbonyl-Bridged Biheterocyclic Compounds for Drug Discovery
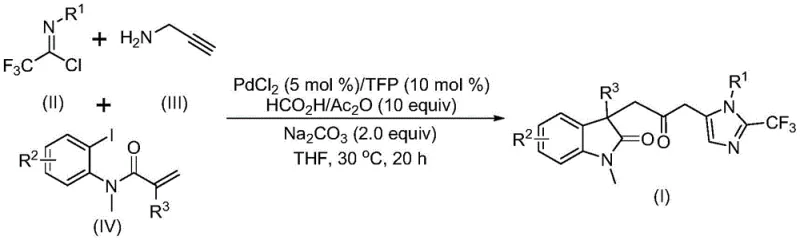
The pharmaceutical industry continuously seeks robust methodologies for constructing complex heterocyclic scaffolds, which serve as the core backbone for numerous bioactive molecules. A significant breakthrough in this domain is detailed in patent CN115353511A, which discloses a novel multi-component method for synthesizing carbonyl-bridged biheterocyclic compounds. This technology represents a paradigm shift in how indolinone and imidazole fused systems are assembled, addressing critical pain points regarding safety, efficiency, and scalability. By leveraging a transition metal palladium-catalyzed carbonylation cascade, this process enables the efficient construction of these valuable motifs without the need for hazardous carbon monoxide gas cylinders. For R&D directors and process chemists, this innovation offers a streamlined pathway to access diversified libraries of trifluoromethyl-containing biheterocycles, which are increasingly relevant in modern medicinal chemistry for their metabolic stability and binding affinity.
The strategic value of this patent lies in its ability to merge three distinct building blocks—trifluoroethylimidoyl chloride, propargylamine, and acrylamide derivatives—into a single, cohesive molecular architecture. This one-pot strategy not only reduces the number of isolation steps but also minimizes waste generation, aligning perfectly with green chemistry principles. As a reliable pharmaceutical intermediate supplier, understanding such advanced synthetic routes is crucial for maintaining a competitive edge in the global market. The method described allows for the rapid generation of structural diversity through simple substrate modification, making it an ideal candidate for lead optimization campaigns in drug discovery programs targeting various therapeutic areas.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of biheterocyclic compounds has relied on laborious multi-step sequences that often suffer from low overall yields and poor atom economy. Traditional approaches typically involve the direct coupling of two pre-formed heterocyclic substrates, which requires harsh reaction conditions and expensive catalysts to overcome the inherent stability of the aromatic systems. Alternatively, oxidative cyclization reactions using activated methyl-substituted heterocycles have been employed, but these methods frequently necessitate stoichiometric amounts of oxidants, leading to significant byproduct formation and challenging purification protocols. Furthermore, conventional carbonylation strategies to introduce the bridging carbonyl group almost universally rely on the use of toxic carbon monoxide gas, posing severe safety risks and requiring specialized high-pressure equipment that is not readily available in standard synthetic laboratories. These limitations create substantial bottlenecks in the supply chain, increasing both the cost and the lead time for producing high-purity intermediates.
The Novel Approach
In stark contrast, the methodology outlined in CN115353511A introduces a transformative one-pot multi-component reaction that elegantly bypasses these traditional hurdles. By utilizing a palladium-catalyzed cascade involving a Heck reaction followed by carbonylation and cyclization, this new approach constructs the complex biheterocyclic framework in a single operational step. The most notable advancement is the replacement of gaseous carbon monoxide with a safe and easy-to-handle mixture of formic acid and acetic anhydride, which generates the necessary CO in situ under mild conditions. This modification drastically reduces the infrastructure requirements for manufacturing, allowing the reaction to proceed at a温和 temperature of 30°C in common solvents like tetrahydrofuran (THF). The result is a process that is not only safer and more environmentally benign but also significantly more cost-effective, as it utilizes cheap and commercially available starting materials such as propargylamine and acrylamide derivatives.
Mechanistic Insights into Pd-Catalyzed Carbonylation Cascade
Understanding the mechanistic underpinnings of this transformation is essential for R&D teams aiming to optimize the process for specific substrates. The reaction is believed to initiate with the oxidative addition of a zero-valent palladium species into the carbon-iodine bond of the acrylamide substrate, generating an aryl-palladium intermediate. This is followed by an intramolecular Heck-type insertion into the alkene moiety, forming a divalent alkyl-palladium species. Crucially, the presence of the formic acid and acetic anhydride mixture facilitates the release of carbon monoxide, which then inserts into the palladium-carbon bond to form an acyl-palladium intermediate. Simultaneously, a parallel pathway involves the base-promoted intermolecular carbon-nitrogen bond formation between the trifluoroethylimidoyl chloride and propargylamine, yielding a trifluoroacetamidine compound that undergoes isomerization. The convergence of these pathways occurs when the acyl-palladium intermediate activates the trifluoroacetamidine, catalyzing an intramolecular cyclization that最终 yields the carbonyl-bridged biheterocyclic product with high regioselectivity.
The choice of ligands and additives plays a pivotal role in stabilizing the active catalytic species and ensuring high turnover numbers. The patent specifies the use of trifurylphosphine (TFP) as the ligand, which likely enhances the electron density on the palladium center, facilitating the oxidative addition step while maintaining stability under the reaction conditions. Sodium carbonate serves as a mild base to neutralize the acidic byproducts generated during the amidine formation and cyclization steps, preventing catalyst deactivation. This delicate balance of reagents allows for excellent functional group tolerance, accommodating electron-withdrawing groups like nitro and trifluoromethyl, as well as electron-donating groups like methoxy and alkyl chains. Such robustness is critical for industrial applications where feedstock purity can vary, ensuring consistent product quality and minimizing the formation of difficult-to-remove impurities.
How to Synthesize Carbonyl-Bridged Biheterocyclic Compounds Efficiently
Implementing this synthesis in a practical setting requires careful attention to reagent stoichiometry and reaction parameters to maximize yield and purity. The protocol is designed to be user-friendly, utilizing standard Schlenk techniques or even simple flask setups under inert atmosphere. The molar ratios are optimized to ensure complete conversion of the limiting reagent, typically the trifluoroethylimidoyl chloride, while using slight excesses of the amine and acrylamide components to drive the equilibrium forward. The reaction time is flexible, ranging from 12 to 20 hours, providing operators with a wide window to monitor progress without risking significant decomposition. Post-reaction workup is straightforward, involving simple filtration to remove inorganic salts followed by silica gel chromatography, a standard purification technique familiar to most process chemistry teams. For detailed standardized synthesis steps, please refer to the guide below.
- Combine palladium chloride, trifurylphosphine, sodium carbonate, and the CO substitute mixture (formic acid/acetic anhydride) in an organic solvent like THF.
- Add the three key substrates: trifluoroethylimidoyl chloride, propargylamine, and the acrylamide derivative to the reaction vessel.
- Stir the mixture at 30°C for 12 to 20 hours, then filter and purify via column chromatography to isolate the final product.
Commercial Advantages for Procurement and Supply Chain Teams
From a procurement and supply chain perspective, this technology offers compelling advantages that directly impact the bottom line and operational resilience. The elimination of toxic carbon monoxide gas removes the need for specialized storage facilities, gas monitoring systems, and high-pressure reactors, which translates to significant capital expenditure savings and reduced regulatory compliance burdens. Furthermore, the reliance on commodity chemicals like propargylamine and acrylamide ensures a stable and diversified supply base, mitigating the risk of shortages that often plague specialized reagents. The mild reaction conditions (30°C) also imply lower energy consumption compared to high-temperature processes, contributing to a smaller carbon footprint and reduced utility costs. These factors combined make the adoption of this synthetic route a strategic move for companies looking to optimize their manufacturing economics while maintaining high standards of safety and sustainability.
- Cost Reduction in Manufacturing: The economic benefits of this process are driven primarily by the simplification of the synthetic route and the use of inexpensive raw materials. By consolidating multiple bond-forming events into a single pot, the method eliminates the need for intermediate isolation and purification steps, which are traditionally the most costly and time-consuming parts of chemical manufacturing. Additionally, the avoidance of high-pressure equipment and toxic gas handling reduces both the initial capital investment and the ongoing operational maintenance costs. The high yields reported in the patent examples, often exceeding 80%, further enhance cost efficiency by maximizing the output per unit of input material, thereby reducing waste disposal costs and improving overall material throughput.
- Enhanced Supply Chain Reliability: Supply chain continuity is bolstered by the use of widely available starting materials that are produced on a large industrial scale globally. Unlike proprietary catalysts or exotic reagents that may have long lead times or single-source dependencies, the key components of this reaction can be sourced from multiple vendors, ensuring competitive pricing and reliable delivery schedules. The robustness of the reaction to various functional groups also means that slight variations in raw material quality are less likely to cause batch failures, adding a layer of resilience to the production schedule. This reliability is crucial for meeting the tight deadlines often associated with drug development timelines and commercial manufacturing contracts.
- Scalability and Environmental Compliance: The patent explicitly mentions the successful expansion of the reaction to the gram scale, demonstrating its potential for seamless translation to kilogram and ton scales. The use of a liquid CO source instead of gas simplifies the engineering controls required for scale-up, making it easier to adapt to existing reactor infrastructure without major modifications. Moreover, the generation of fewer byproducts and the use of less hazardous reagents align with increasingly stringent environmental regulations, facilitating easier permitting and waste management. This environmental compatibility not only reduces liability but also enhances the corporate social responsibility profile of the manufacturing operation, which is becoming a key differentiator in the global marketplace.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding this synthesis method, derived from the specific details and beneficial effects highlighted in the patent documentation. These insights are intended to clarify the operational feasibility and strategic value of adopting this technology for your specific production needs. Understanding these nuances helps stakeholders make informed decisions about process integration and resource allocation.
Q: What are the primary advantages of this multi-component synthesis over traditional methods?
A: This method avoids the use of toxic carbon monoxide gas by utilizing a formic acid/acetic anhydride system, operates under mild conditions (30°C), and achieves high yields in a one-pot process, significantly simplifying purification and reducing operational hazards.
Q: Is this process suitable for large-scale industrial production?
A: Yes, the patent explicitly demonstrates successful expansion to gram-scale reactions with high substrate compatibility, indicating strong potential for commercial scale-up due to the use of cheap, readily available starting materials and standard laboratory equipment.
Q: What types of substituents are tolerated in this reaction?
A: The reaction exhibits excellent functional group tolerance, accommodating various substituents on the aryl rings including alkyl, alkoxy, halogens (chloro, bromo, fluoro), trifluoromethyl, and nitro groups, allowing for diverse library synthesis.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Carbonyl-Bridged Biheterocyclic Compounds Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of advanced synthetic methodologies like the one described in CN115353511A for accelerating drug discovery and development. As a premier CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your transition from lab bench to manufacturing plant is seamless and efficient. Our state-of-the-art facilities are equipped to handle complex multi-component reactions with the highest standards of safety and quality control. We maintain stringent purity specifications and operate rigorous QC labs to guarantee that every batch of carbonyl-bridged biheterocyclic compounds meets the exacting requirements of the pharmaceutical industry, providing you with a reliable foundation for your clinical and commercial programs.
We invite you to collaborate with us to leverage this cutting-edge technology for your next project. Our team of experts is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements and timeline constraints. By partnering with us, you gain access to our deep technical expertise and robust supply chain network, enabling you to reduce lead time for high-purity pharmaceutical intermediates and bring your therapies to market faster. Please contact our technical procurement team today to request specific COA data and route feasibility assessments, and let us demonstrate how we can add value to your supply chain through innovation and operational excellence.
