Revolutionizing Tedizolid Phosphate Purity: A Scalable Solvent-Efficient Purification Strategy
Revolutionizing Tedizolid Phosphate Purity: A Scalable Solvent-Efficient Purification Strategy
In the highly regulated landscape of antibiotic manufacturing, the purity profile of Active Pharmaceutical Ingredients (APIs) dictates both clinical efficacy and regulatory approval success. Patent CN112961186A introduces a transformative approach to the purification of Tedizolid Phosphate, a next-generation oxazolidinone antibiotic critical for treating Acute Bacterial Skin and Skin Structure Infections (ABSSSI). Traditional synthesis routes often rely on aggressive phosphorylation reagents like phosphorus oxychloride, which inevitably generate a complex matrix of degradation products, including dimers, chloro-impurities, and ring-opening byproducts. This new methodology addresses these challenges by implementing a novel solvent engineering strategy that leverages absolute ethyl alcohol for selective impurity removal, followed by a precise pH-swing crystallization process. For R&D Directors and Procurement Managers alike, this represents a significant leap forward, offering a pathway to achieve purity levels exceeding 99.9% while simultaneously eliminating the reliance on difficult-to-remove high-boiling solvents like dimethyl sulfoxide (DMSO) that have historically plagued the supply chain and increased production costs.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the purification of Tedizolid Phosphate has been fraught with technical inefficiencies that directly impact commercial viability and product quality. Conventional methods typically involve converting the crude product into a disodium salt using sodium methoxide, followed by acidification and recrystallization using mixtures of ethanol and diethyl ether. However, the strong alkaline conditions required for salt formation often induce degradation, leading to the formation of unstable impurities that compromise the final assay. Furthermore, alternative purification protocols relying on dimethyl sulfoxide (DMSO) or N-methyl pyrrolidone (NMP) present severe downstream processing hurdles. These high-boiling point solvents possess strong solvating power that makes them exceptionally difficult to remove completely from the crystal lattice, often requiring prolonged drying times under high vacuum and elevated temperatures. This not only increases energy consumption and cycle time but also risks thermal degradation of the sensitive oxazolidinone core. Additionally, the use of diethyl ether in traditional recrystallization poses significant safety risks due to its low flash point and tendency to form explosive peroxides, making it unsuitable for modern, safety-conscious industrial facilities aiming for robust commercial scale-up.
The Novel Approach
The innovative method disclosed in the patent fundamentally rethinks the purification logic by exploiting the differential solubility characteristics of the target molecule versus its impurities. Instead of attempting to dissolve the product and crystallize it out—a process where impurities often co-precipitate—this approach begins with a solid-liquid extraction using absolute ethyl alcohol. In this step, the crude Tedizolid Phosphate is mixed with absolute ethanol, which selectively dissolves impurities with lower polarity while leaving the target Tedizolid Phosphate in the solid phase. This initial 'wash' drastically reduces the impurity load before the product even enters the solution phase. Following filtration, the solid is converted to the water-soluble disodium salt using a controlled concentration of sodium hydroxide. This aqueous solution is then subjected to microfiltration to remove any remaining insoluble particulates, ensuring a pristine starting point for the final crystallization. By avoiding organic solvents with high boiling points and utilizing a water-based system for the bulk of the purification, the process simplifies the isolation steps, reduces solvent recovery costs, and significantly enhances the overall safety profile of the manufacturing operation.
Mechanistic Insights into Solvent-Driven Impurity Control
The core mechanism driving the success of this purification strategy lies in the precise manipulation of solubility parameters and ionization states. Tedizolid Phosphate contains a phosphate ester moiety that renders it amphoteric, allowing it to exist in different ionic forms depending on the pH of the environment. In the initial absolute ethanol wash, the neutral or less polar impurities—such as unreacted starting materials or non-phosphorylated side products—are solubilized and removed. The target molecule, being more polar due to the phosphate group, remains largely insoluble in the anhydrous alcohol. Upon treatment with aqueous sodium hydroxide, the phosphate group is deprotonated to form the disodium salt, which exhibits high solubility in water. This phase transfer allows for the physical separation of the product from any remaining ethanol-soluble impurities that were not washed away initially. The subsequent acidification step, where concentrated hydrochloric acid adjusts the pH to between 1 and 3, reprotonates the phosphate group, causing the Tedizolid Phosphate to precipitate out of the aqueous solution in a highly crystalline form. This 'dissolve-filter-precipitate' cycle is far more effective at excluding impurities than simple recrystallization, as it physically separates the product from the mother liquor where the impurities remain dissolved.
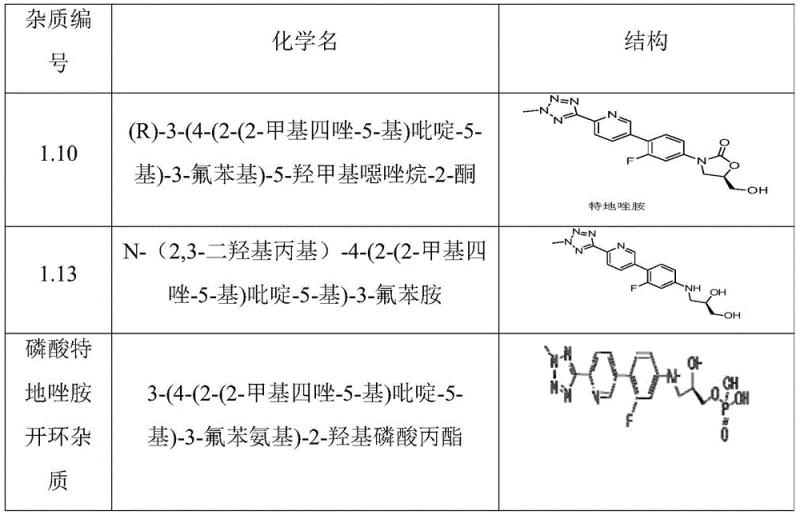
Understanding the specific impurity profile is critical for R&D teams aiming to replicate this high level of purity. The patent identifies a comprehensive list of potential contaminants, including ring-opening impurities, demethylated variants, and various phosphorylation byproducts like dimers and pyrophosphates. As illustrated in the structural analysis, these impurities often differ subtly from the main compound, making them difficult to separate via standard chromatography on a large scale. However, the described purification method effectively targets these specific structural analogs. For instance, the ring-opening impurities often possess different polarity profiles compared to the intact oxazolidinone ring, making them susceptible to removal during the absolute ethanol wash or the aqueous filtration steps. Similarly, dimeric impurities, which are larger and often less soluble or differently charged, are effectively excluded during the controlled crystallization phase. The result is a product where individual specified impurities are controlled to levels below 0.1%, and the total impurity content is minimized to less than 0.5%, meeting the stringent requirements for global pharmaceutical markets.
How to Synthesize Tedizolid Phosphate Efficiently
The implementation of this purification protocol requires careful attention to solvent ratios and pH control to maximize yield and purity. The process is designed to be robust, tolerating slight variations while maintaining high performance, which is essential for technology transfer from the lab to the pilot plant. The key lies in the initial mass-to-volume ratio of the crude product to absolute ethanol, which is optimized to ensure maximum impurity solubility without dissolving the product. Following the ethanol wash, the conversion to the disodium salt must be complete to ensure full dissolution in the aqueous phase, facilitating the microfiltration step that acts as a final barrier against particulate matter. The detailed standardized synthesis steps for replicating this high-efficiency purification process are outlined in the guide below.
- Mix crude Tedizolid Phosphate with absolute ethyl alcohol to dissolve and filter out non-polar impurities, retaining the target compound in the solid phase.
- Dissolve the filtered solid in aqueous sodium hydroxide to form the disodium salt, followed by microfiltration to remove insoluble particulates.
- Acidify the solution to pH 1-3 using concentrated hydrochloric acid to induce crystallization, then wash and dry to obtain high-purity Tedizolid Phosphate.
Commercial Advantages for Procurement and Supply Chain Teams
For Procurement Managers and Supply Chain Heads, the shift from DMSO-based purification to an ethanol-water system offers profound economic and logistical benefits. The most immediate impact is the drastic reduction in solvent recovery costs. High-boiling solvents like DMSO require significant energy input for distillation and often degrade upon recycling, necessitating frequent fresh solvent purchases. In contrast, absolute ethanol and water are commodity chemicals with well-established, low-cost recovery infrastructures. This transition translates directly into a lower Cost of Goods Sold (COGS) for the final API. Furthermore, the elimination of hazardous solvents like diethyl ether reduces the regulatory burden associated with storage and handling, lowering insurance premiums and facility compliance costs. The simplified process flow also means shorter batch cycle times; without the need for prolonged drying to remove tenacious high-boiling solvents, production throughput can be significantly increased, enhancing the reliability of supply for downstream drug product manufacturers.
- Cost Reduction in Manufacturing: The replacement of expensive, high-boiling solvents with readily available absolute ethanol and water creates a substantial opportunity for cost optimization. By eliminating the energy-intensive steps required to strip DMSO or NMP from the crystal lattice, manufacturers can reduce utility consumption and shorten drying cycles. Additionally, the high efficiency of the impurity removal reduces the need for re-processing or second crops, thereby improving the overall material yield and reducing waste disposal costs associated with failed batches or off-spec material.
- Enhanced Supply Chain Reliability: Relying on commodity solvents like ethanol and hydrochloric acid mitigates the risk of supply disruptions that can occur with specialty solvents. The robustness of the purification method ensures consistent quality output, reducing the variability that often leads to supply chain bottlenecks. With a clearer impurity profile and a more stable crystalline form, the risk of batch rejection during Quality Control (QC) testing is minimized, ensuring a steady flow of material to meet market demand for this critical antibiotic.
- Scalability and Environmental Compliance: The process is inherently greener, aligning with modern environmental, social, and governance (ESG) goals. The use of aqueous systems and recyclable ethanol reduces the volatile organic compound (VOC) emissions associated with traditional organic solvent-heavy processes. This environmental friendliness simplifies the permitting process for new manufacturing lines and ensures long-term compliance with increasingly strict environmental regulations. The simplicity of the unit operations—mixing, filtration, and crystallization—makes the process highly scalable from pilot batches to multi-ton commercial production without significant re-engineering.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the purification of Tedizolid Phosphate, derived from the specific advantages and mechanisms detailed in the patent literature. These insights are intended to clarify the operational benefits and quality assurances provided by this advanced manufacturing route.
Q: Why is absolute ethanol preferred over DMSO for Tedizolid Phosphate purification?
A: Absolute ethanol selectively dissolves non-polar impurities while leaving the target compound in the solid phase, whereas DMSO requires complex removal processes due to its high boiling point and can trap impurities within the crystal lattice.
Q: How does this method control specific impurities like dimers and ring-opening products?
A: The combination of polarity-based washing with absolute ethanol and subsequent pH-controlled crystallization effectively separates structurally related impurities, ensuring levels remain below 0.1% for individual specified impurities.
Q: Is this purification method suitable for large-scale industrial production?
A: Yes, the method avoids hazardous high-boiling solvents and complex multi-step recrystallizations, utilizing common solvents like ethanol and water which are easily recoverable and safe for multi-ton scale manufacturing.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Tedizolid Phosphate Supplier
At NINGBO INNO PHARMCHEM, we recognize that the purity of Tedizolid Phosphate is not just a specification but a critical determinant of patient safety and therapeutic outcome. Our technical team has thoroughly analyzed the advancements in purification technologies, including the solvent-efficient methods described in recent patents, and integrated these principles into our own manufacturing workflows. We possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the high purity achieved in the lab is faithfully reproduced at an industrial scale. Our facilities are equipped with rigorous QC labs and state-of-the-art crystallization units capable of maintaining stringent purity specifications, guaranteeing that every batch of Tedizolid Phosphate meets the highest global pharmacopoeia standards.
We invite pharmaceutical partners to collaborate with us to secure a stable and cost-effective supply of this vital antibiotic intermediate. Our team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements, demonstrating how our optimized purification processes can reduce your overall procurement costs. Please contact our technical procurement team today to request specific COA data, route feasibility assessments, and samples for your evaluation, and let us support your journey towards delivering high-quality medicines to patients worldwide.
