Advanced Manufacturing of Tedizolid Phosphate Intermediates via Novel Refining Protocols
Introduction to Patent CN113214239B
The pharmaceutical landscape for oxazolidinone antibiotics continues to evolve, with Tedizolid Phosphate standing out as a potent agent against methicillin-resistant Staphylococcus aureus (MRSA). A critical challenge in the commercial production of this active pharmaceutical ingredient (API) lies not just in the synthesis, but in the rigorous purification of its key intermediate, Tedizolid. Patent CN113214239B, filed in April 2023, discloses a groundbreaking refining process that fundamentally alters the purification landscape. This technology addresses the longstanding issue of impurity carryover which traditionally plagues the subsequent phosphorylation step. By introducing a specialized mixed solvent system, the patent outlines a method that significantly enhances both the purity and yield of the intermediate, thereby securing the quality of the final drug substance. For global supply chain leaders, this represents a pivotal shift towards more robust and efficient manufacturing protocols for high-value antibiotic intermediates.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the purification of Tedizolid intermediates has relied heavily on the use of glacial acetic acid. While effective to a degree, this conventional approach introduces significant downstream complications that impact overall process efficiency. The primary drawback is the retention of acidic residues within the crystal lattice of the intermediate. When this inadequately purified intermediate is subjected to the subsequent phosphorylation reaction using phosphorus oxychloride (POCl3), these residual acids act as catalysts for unwanted side reactions. This leads to the generation of complex, difficult-to-remove impurities that compromise the purity profile of the final Tedizolid Phosphate. Furthermore, the removal of glacial acetic acid often requires harsh drying conditions or extensive washing, which can degrade the sensitive heterocyclic structures within the molecule, ultimately lowering the overall yield and increasing the burden on waste treatment systems due to high acid consumption.
The Novel Approach
The methodology presented in CN113214239B offers a sophisticated alternative by replacing the acidic solvent environment with a precisely tuned ternary solvent system comprising dichloromethane, toluene, and ethanol. This innovation leverages the differential solubility profiles of the target compound versus its impurities. By heating the crude reaction mixture in this specific ratio—typically ranging from 0.2:0.5:1 to 0.5:3:1—the process ensures that impurities remain dissolved while the pure Tedizolid crystallizes out upon cooling. Crucially, the use of dichloromethane eliminates the risk of acidic residue interference, while the toluene and ethanol components facilitate the removal of organic byproducts and allow for gentle drying at temperatures as low as 35°C to 40°C. This mild thermal profile preserves the structural integrity of the oxazolidinone ring, resulting in an intermediate with purity levels exceeding 99.9% and significantly reduced impurity loads for the next synthetic step.
Mechanistic Insights into Suzuki Coupling and Solvent Engineering
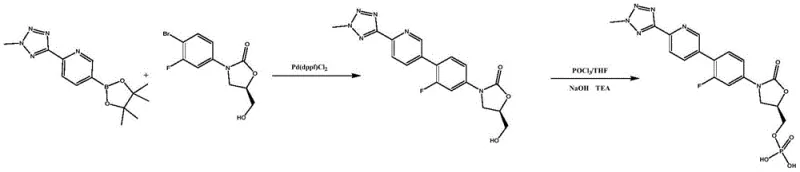
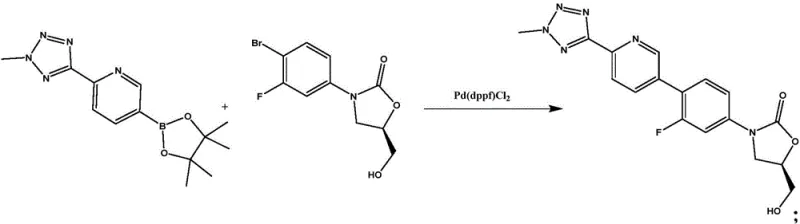
The core of this synthesis relies on a palladium-catalyzed Suzuki-Miyaura cross-coupling reaction, linking a tetrazole-pyridine boronic ester with a fluoro-bromo-oxazolidinone scaffold. The reaction utilizes Pd(dppf)Cl2 as the catalyst in a 1,4-dioxane medium with potassium carbonate as the base. While the coupling itself is standard, the true mechanistic breakthrough lies in the workup and purification phase. The patent elucidates that the specific interaction between the dichloromethane-toluene-ethanol matrix and the molecular surface of Tedizolid creates a thermodynamic environment where impurity inclusion is minimized. Unlike acetic acid, which can protonate basic nitrogen sites on the tetrazole or pyridine rings leading to salt formation and occlusion of impurities, the neutral organic solvent system allows for the growth of highly ordered crystals. This crystal engineering aspect is vital for R&D directors, as it implies that the physical form of the intermediate is optimized not just for purity, but for reactivity in the subsequent phosphorylation.
Furthermore, the purification mechanism effectively targets trace metal residues and organic side-products that are notoriously difficult to separate. By dissolving the crude solid in the heated mixed solvent and then slowly cooling, the process exploits the steep solubility curve of Tedizolid in this specific mixture. The result is a precipitate that is virtually free of the precursors that would otherwise react with phosphorus oxychloride. This preemptive removal of reactive impurities is the key to suppressing side reactions in the phosphorylation stage. It ensures that the nucleophilic attack of the hydroxymethyl group on the phosphorus center proceeds with high selectivity, minimizing the formation of di-phosphorylated species or chlorinated byproducts. This level of control over the impurity profile is essential for meeting the stringent regulatory requirements for antibiotic APIs, ensuring that the final drug substance remains stable over extended shelf lives.
How to Synthesize Tedizolid Efficiently
Implementing this refined synthesis protocol requires precise adherence to the solvent ratios and thermal gradients defined in the patent. The process begins with the Suzuki coupling, followed immediately by the novel extraction and crystallization sequence. Operators must ensure the dichloromethane, toluene, and ethanol are mixed in the correct volume proportions before heating the slurry to between 60°C and 70°C. Maintaining this temperature for 10 to 30 minutes is critical to fully dissolve impurities without degrading the product. Following filtration and ethanol washing, the drying phase must be strictly controlled below 40°C to prevent thermal stress on the molecule. The detailed standardized operating procedures for scaling this reaction from laboratory to pilot plant are outlined in the technical guide below.
- Perform Suzuki coupling between 2-(2-methyl-2H-tetrazole-5-yl)pyridine-5-boronic acid pinacol ester and the bromo-oxazolidinone precursor using Pd(dppf)Cl2 catalyst.
- Purify the crude tedizolid intermediate using a specific mixed solvent system of dichloromethane, toluene, and ethanol to remove impurities effectively.
- React the purified tedizolid with phosphorus oxychloride in tetrahydrofuran under low-temperature conditions to form the final phosphate ester.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this refining process translates directly into tangible operational efficiencies and risk mitigation. The shift away from glacial acetic acid not only simplifies the chemical handling requirements but also drastically reduces the corrosion risks associated with processing equipment, extending the lifespan of reactors and piping. More importantly, the substantial improvement in yield observed in the patent examples—jumping from roughly 49% in conventional methods to over 73% with this new process—means a dramatic reduction in the cost of goods sold (COGS). By maximizing the output from every kilogram of expensive palladium-catalyzed starting material, manufacturers can achieve significant cost reduction in pharmaceutical intermediates manufacturing without compromising on quality standards.
- Cost Reduction in Manufacturing: The elimination of glacial acetic acid removes the need for extensive neutralization and washing steps, thereby reducing water consumption and wastewater treatment costs. Additionally, the higher yield of the intermediate means less raw material is required to produce the same amount of final API. This efficiency gain is compounded by the reduced need for reprocessing or re-crystallization of the final phosphate product, as the cleaner intermediate leads to a cleaner final reaction, saving both time and resources in the purification of the end product.
- Enhanced Supply Chain Reliability: The solvents used in this new protocol—dichloromethane, toluene, and ethanol—are commodity chemicals with robust global supply chains, unlike some specialized reagents that might face shortages. This ensures a reliable tedizolid phosphate intermediate supplier can maintain continuous production schedules without interruption. The simplified workflow also reduces the cycle time per batch, allowing for greater throughput and faster response to market demand fluctuations, which is critical for maintaining inventory levels of essential antibiotics.
- Scalability and Environmental Compliance: The process operates under relatively mild conditions, with heating temperatures capped at 70°C and drying temperatures as low as 35°C. This low-energy profile makes the process highly scalable and easier to implement in existing facilities without major infrastructure upgrades. Furthermore, by avoiding large volumes of acidic waste, the process aligns better with modern environmental, social, and governance (ESG) goals, reducing the environmental footprint of commercial scale-up of complex pharmaceutical intermediates and simplifying regulatory compliance regarding effluent discharge.
Frequently Asked Questions (FAQ)
The following questions address common technical inquiries regarding the implementation and benefits of this novel refining technology. These insights are derived directly from the experimental data and comparative analysis provided in the patent documentation, offering clarity on how this method outperforms traditional synthesis routes in terms of purity, stability, and operational simplicity.
Q: Why is the new solvent system superior to glacial acetic acid for tedizolid purification?
A: The novel mixed solvent system (dichloromethane:toluene:ethanol) avoids the acidic residue issues associated with glacial acetic acid. Residual acetic acid can cause side reactions with phosphorus oxychloride during the subsequent phosphorylation step, leading to new impurities and reduced yields. The new system ensures higher purity (>99.9%) and better stability of the final phosphate product.
Q: What represents the key yield improvement in this patented process?
A: Comparative data in the patent indicates a significant yield enhancement. While conventional refining methods yielded approximately 49% of the intermediate, the novel process achieves yields up to 73% for the refined tedizolid and subsequently improves the final tedizolid phosphate yield to over 87%, drastically reducing raw material consumption.
Q: How does this process impact the long-term stability of the API intermediate?
A: Stability testing demonstrates that tedizolid phosphate produced via this refined intermediate maintains purity above 99.90% even after 28 days of storage. In contrast, products derived from conventionally purified intermediates show noticeable purity degradation over the same period, indicating that the new purification method removes specific trace impurities that catalyze decomposition.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Tedizolid Phosphate Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of high-quality intermediates in the production of life-saving antibiotics. Our technical team has extensively analyzed the pathways described in CN113214239B and possesses the expertise to replicate and optimize this advanced refining process. We bring extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from lab-scale success to industrial reality is seamless. Our facilities are equipped with rigorous QC labs capable of verifying stringent purity specifications, ensuring that every batch of Tedizolid intermediate we supply meets the highest global standards for safety and efficacy.
We invite potential partners to engage with our technical procurement team to discuss how this innovative synthesis route can be integrated into your supply chain. By leveraging our capabilities, you can access a Customized Cost-Saving Analysis tailored to your specific production volumes. We encourage you to contact us today to request specific COA data and route feasibility assessments, and let us demonstrate how our commitment to technological excellence can drive value and reliability in your antibiotic manufacturing operations.
