Advanced Hydrolysis Technology for High-Purity Gabapentin Manufacturing and Scale-Up
Advanced Hydrolysis Technology for High-Purity Gabapentin Manufacturing and Scale-Up
The pharmaceutical industry continuously seeks robust manufacturing routes that balance high purity with operational safety, particularly for high-volume antiepileptic agents like gabapentin. A significant technological breakthrough in this domain is detailed in patent CN1896050A, which outlines a novel preparation method utilizing 3,3-pentylidene butyrolactam as the primary starting material. Unlike traditional methodologies that rely heavily on cumbersome ion-exchange resin columns and hazardous solvent mixtures, this innovative approach employs a direct mineral acid hydrolysis followed by precise pH-controlled crystallization. This shift represents a paradigm change in how we approach the commercial scale-up of complex pharmaceutical intermediates, offering a pathway that drastically simplifies the downstream processing while ensuring the final product meets stringent pharmacopeial standards for toxic impurities and chloride content.
For R&D directors and process chemists evaluating potential technology transfers, the significance of this patent lies in its ability to mitigate the formation of toxic cyclic impurities that often plague gabapentin synthesis. The core innovation involves a carefully managed hydrolysis step where reaction conditions are optimized to prevent the re-cyclization of the open-chain amino acid back into the toxic lactam form. By controlling the temperature and acid concentration during the initial breakdown of the lactam ring, and subsequently managing the crystallization environment, the process achieves a purity profile that exceeds 99.8% with toxic impurities undetectable or below 0.03%. This level of control is critical for maintaining regulatory compliance and ensuring patient safety in the final drug product.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industrial production of gabapentin has been hindered by the reliance on ion-exchange resin technologies, which introduce significant operational bottlenecks and safety hazards. Traditional methods, such as those described in earlier patents like US4024175, require the use of large volumes of water as a solvent, necessitating energy-intensive distillation steps to concentrate the product prior to crystallization. During these distillation processes, strict temperature control below 45°C is mandatory; failure to maintain this threshold often leads to the thermal cyclization of gabapentin back into 3,3-pentylidene butyrolactam, a toxic impurity with an LD50 significantly lower than the active pharmaceutical ingredient itself. Furthermore, the conventional use of mixed ether and alcohol solvents for final crystallization poses severe safety risks due to the formation of explosive peroxides and the difficulty in recovering these volatile organic compounds efficiently.
The Novel Approach
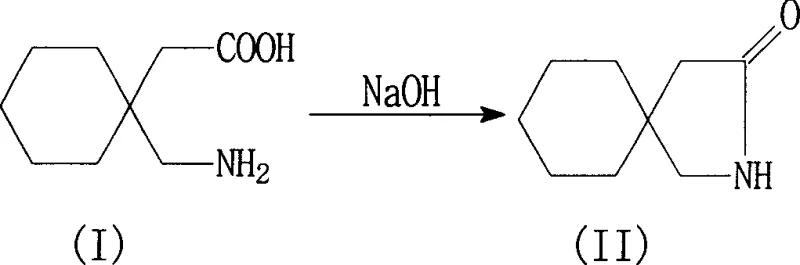
In stark contrast, the methodology disclosed in CN1896050A eliminates the need for ion-exchange resins entirely, replacing them with a direct acid hydrolysis and selective precipitation strategy. This novel approach utilizes mineral acids, preferably hydrochloric acid, to hydrolyze the starting lactam directly into the gabapentin salt, which is then isolated through cooling crystallization. The subsequent purification steps involve a sophisticated pH-swing technique where the salt is dissolved, decolorized using activated carbon and diatomite to adsorb residual toxic impurities, and then re-precipitated as the hydrate form by adjusting the pH to the isoelectric range of 8 to 8.5. This sequence not only simplifies the equipment requirements by removing the need for large resin columns but also significantly reduces the production cycle time and energy consumption associated with water removal.
Mechanistic Insights into Acid-Catalyzed Lactam Hydrolysis
The chemical foundation of this improved process rests on the efficient ring-opening of the 3,3-pentylidene butyrolactam structure under acidic conditions. In the first critical stage, the lactam nitrogen is protonated by the mineral acid, rendering the carbonyl carbon highly susceptible to nucleophilic attack by water molecules. This hydrolysis reaction proceeds effectively at temperatures between 80°C and 110°C, ensuring complete conversion of the starting material into the corresponding gabapentin hydrochloride salt. The reaction mixture is then cooled to a range of -10°C to 20°C, inducing the crystallization of the salt while leaving unreacted starting materials and soluble byproducts in the mother liquor. This temperature-dependent solubility differential is key to the initial purification, allowing for the physical separation of the desired intermediate from the bulk reaction mass without the need for chromatographic techniques.
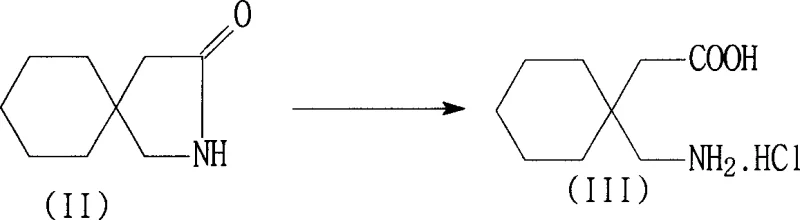
Following the isolation of the salt, the mechanism shifts towards impurity management through adsorption and pH manipulation. The dissolution of the crude salt in purified water allows for the introduction of adsorbents like activated carbon, which physically trap trace amounts of the toxic cyclic lactam and colored organic impurities. Subsequent adjustment of the pH to the 3-7 range ensures the solution is clear before the final precipitation step. By raising the pH to 8-8.5 using a base such as sodium hydroxide, the zwitterionic nature of gabapentin is exploited to precipitate the hydrate form selectively. This precise control over the protonation state of the amino and carboxyl groups prevents the co-precipitation of inorganic salts and ensures that the chloride ion content in the final product remains well within the acceptable limit of 100 ppm, a common failure point in less optimized synthetic routes.
How to Synthesize Gabapentin Efficiently
The execution of this synthesis requires careful attention to the stoichiometric ratios of acid and base, as well as strict thermal management during the crystallization phases. The process is designed to be telescoped where possible, with mother liquors from the initial hydrolysis step being recycled to recover unreacted starting material, thereby maximizing atom economy. Operators must ensure that the hydrolysis reaction time is sufficient, typically ranging from 1 to 15 hours depending on the acid concentration, to drive the equilibrium towards the open-chain salt. The detailed standardized synthesis steps, including specific reagent quantities and agitation speeds for optimal crystal habit formation, are outlined in the technical guide below to assist process engineers in replicating these results.
- Hydrolyze 3,3-pentylidene butyrolactam with mineral acid (e.g., HCl) at 80-110°C, then cool to -10 to 20°C to crystallize Gabapentin salt.
- Dissolve the salt in water, adjust pH to 3-7, decolorize with activated carbon, then adjust pH to 8-8.5 to precipitate Gabapentin hydrate.
- Suspend the hydrate in alcohol (e.g., ethanol) at 20-40°C to dehydrate and crystallize final Gabapentin, followed by drying at 40-45°C.
Commercial Advantages for Procurement and Supply Chain Teams
From a procurement and supply chain perspective, the adoption of this hydrolysis-based methodology offers substantial strategic benefits that extend beyond mere technical feasibility. The elimination of ion-exchange resins removes a significant variable cost component, as these consumables require frequent replacement and regeneration, adding to both direct material costs and waste disposal expenses. Furthermore, the simplified workflow reduces the overall facility footprint required for production, as large tanks for resin columns and the associated piping infrastructure are no longer necessary. This reduction in capital expenditure allows manufacturers to allocate resources towards increasing batch sizes or improving quality control laboratories, directly enhancing the reliability of the supply chain for high-purity pharmaceutical intermediates.
- Cost Reduction in Manufacturing: The economic viability of this process is significantly enhanced by the integrated mother liquor recycling loop, which allows for the recovery and reuse of unreacted 3,3-pentylidene butyrolactam. By treating the combined mother liquors with base and heating, the toxic lactam is regenerated and can be fed back into the start of the process, effectively closing the material loop. This circular approach minimizes raw material waste and lowers the effective cost per kilogram of the final API, providing a competitive edge in cost reduction in pharmaceutical intermediates manufacturing without compromising on quality standards.
- Enhanced Supply Chain Reliability: The switch from hazardous ether-alcohol mixtures to safer, single-component alcohols like ethanol for the final dehydration step drastically improves operational safety and solvent availability. Ethanol is a widely available commodity chemical with a stable supply chain, unlike specialized ether solvents which may face logistical constraints or regulatory scrutiny. This solvent optimization ensures that production schedules are not disrupted by solvent shortages or safety shutdowns, thereby reducing lead time for high-purity pharmaceutical intermediates and guaranteeing consistent delivery to downstream formulation partners.
- Scalability and Environmental Compliance: The process is inherently scalable due to its reliance on standard unit operations such as crystallization, filtration, and distillation, which are easily modeled from pilot plant to commercial scale. Additionally, the reduction in wastewater volume—achieved by avoiding the large water loads associated with resin washing—simplifies effluent treatment and lowers environmental compliance costs. The ability to operate with lower energy consumption during the drying and concentration phases further aligns the manufacturing process with modern sustainability goals, making it an attractive option for companies seeking to minimize their carbon footprint while maintaining high production volumes.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this advanced gabapentin synthesis route. These insights are derived directly from the experimental data and process descriptions found in the patent literature, providing a factual basis for decision-making. Understanding these nuances is essential for technical teams evaluating the feasibility of technology transfer and for procurement officers assessing the long-term viability of the supply source.
Q: How does this process control toxic impurities compared to ion-exchange methods?
A: By avoiding high-temperature distillation of aqueous solutions which causes cyclization back to toxic 3,3-pentylidene butyrolactam, and utilizing activated carbon adsorption during the pH swing, toxic impurities are kept below 0.03%.
Q: What are the solvent safety advantages of this new route?
A: The process replaces hazardous ether/alcohol mixed solvents used in traditional crystallization with safer, single-component alcohols like ethanol, significantly reducing explosion risks and simplifying solvent recovery.
Q: Is the mother liquor recycling compatible with large-scale production?
A: Yes, the patent explicitly describes a continuous mother liquor recycling loop where unreacted starting material is recovered and reused, enhancing overall yield above 91% and reducing raw material costs for industrial scale-up.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Gabapentin Supplier
At NINGBO INNO PHARMCHEM, we recognize that the transition from laboratory innovation to commercial reality requires a partner with deep technical expertise and robust manufacturing capabilities. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the intricate pH controls and crystallization parameters defined in this patent are meticulously maintained at every scale. We operate stringent purity specifications and utilize rigorous QC labs equipped with state-of-the-art analytical instrumentation to verify that every batch of gabapentin meets the <0.03% toxic impurity threshold and other critical quality attributes required by global regulatory bodies.
We invite potential partners to engage with our technical procurement team to discuss how this optimized synthesis route can be tailored to your specific supply chain needs. By requesting a Customized Cost-Saving Analysis, you can gain a clearer understanding of the economic benefits inherent in this resin-free process. We encourage you to contact us today to obtain specific COA data and route feasibility assessments, allowing you to make informed decisions that enhance both the quality and cost-efficiency of your gabapentin supply chain.
