Scalable Visible Light Mediated Decarboxylative Ketonization for High-Purity Beta-Ketosulfones
The pharmaceutical and fine chemical industries are constantly seeking sustainable, cost-effective methodologies for constructing complex sulfur-containing scaffolds, particularly beta-ketosulfones, which serve as pivotal intermediates in drug discovery and agrochemical synthesis. A groundbreaking approach detailed in patent CN110981676A introduces a visible light-mediated decarboxylative ketonization reaction that fundamentally shifts the paradigm from traditional transition metal catalysis to organophotocatalysis. This innovative protocol leverages commercially available atropic acid derivatives and stable sulfonyl hydrazides as primary building blocks, utilizing fluorescein as an inexpensive organic photosensitizer and molecular oxygen as a clean terminal oxidant. By operating under mild conditions with simple compact fluorescent lamp (CFL) irradiation, this technology addresses critical pain points regarding metal contamination and hazardous waste generation, positioning it as a highly attractive route for the commercial scale-up of complex pharmaceutical intermediates. The strategic integration of potassium iodide as a catalytic additive further enhances reaction efficiency, enabling high conversion rates without the need for cryogenic temperatures or exotic reagents.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of beta-ketosulfone compounds has relied heavily on transition metal-catalyzed radical oxidative sulfonylation of alkenes or alkynes, often employing catalysts such as silver nitrate or iridium complexes which pose significant economic and environmental burdens. These conventional routes frequently necessitate the use of stoichiometric chemical oxidants like tert-butyl hydroperoxide (TBHP) or persulfates, which introduce safety hazards related to explosion risks and complicate downstream purification due to the formation of difficult-to-remove by-products. Furthermore, many established protocols require rigorous exclusion of moisture and oxygen, demanding specialized equipment for inert gas protection that increases capital expenditure and operational complexity in a manufacturing setting. The presence of heavy metal residues in the final product is another critical concern, particularly for high-purity pharmaceutical intermediates, as it mandates additional, costly scavenging steps to meet stringent regulatory limits for elemental impurities. Additionally, the reliance on high-energy ultraviolet irradiation in some prior art methods can lead to non-selective radical generation and substrate degradation, limiting the overall yield and functional group tolerance of the synthesis.
The Novel Approach
In stark contrast, the methodology disclosed in CN110981676A utilizes a visible light-mediated system that operates at room temperature in a mixed solvent of acetonitrile and water, dramatically simplifying the reaction setup and reducing energy consumption. By replacing toxic metal catalysts with fluorescein, an abundant and biocompatible organic dye, the process inherently eliminates the risk of heavy metal contamination, thereby streamlining the quality control workflow and ensuring compliance with global pharmacopeia standards. The use of molecular oxygen, supplied simply via an oxygen balloon, serves as both the oxidant and the oxygen source for the carbonyl group, offering a greener alternative to hazardous chemical oxidants while generating water as the only benign by-product. This approach demonstrates exceptional functional group compatibility, tolerating various substituents on both the atropic acid and sulfonyl hydrazide moieties, including halogens and trifluoromethyl groups, which are essential for modulating the biological activity of final drug candidates. The robustness of this system is further evidenced by its insensitivity to water, allowing for the use of aqueous solvent mixtures that enhance the solubility of inorganic bases and facilitate easier workup procedures.

Mechanistic Insights into Visible Light Mediated Decarboxylative Ketonization
The mechanistic pathway of this transformation involves a sophisticated interplay of single electron transfer (SET) processes and radical propagation cycles initiated by the excitation of the fluorescein photocatalyst under visible light irradiation. Upon absorption of photons, the ground state fluorescein is promoted to an excited state capable of oxidizing the sulfonyl hydrazide substrate via a single electron transfer event, generating a highly reactive radical cation intermediate. This radical cation subsequently undergoes deprotonation and oxidation by molecular oxygen, releasing nitrogen gas to form the crucial benzenesulfonyl radical species, which acts as the key sulfur-centered radical precursor. Concurrently, the reduced form of the photocatalyst is regenerated by molecular oxygen, producing a superoxide radical anion that participates in the oxidation cycle, ensuring the catalytic turnover of the organic dye without the need for sacrificial reagents. The presence of potassium iodide plays a pivotal role in this mechanism, likely facilitating the regeneration of active radical species or stabilizing transient intermediates through halogen bonding interactions, which explains the significant drop in yield observed when KI is omitted from the reaction mixture.
Following the generation of the sulfonyl radical, it adds selectively to the electron-rich double bond of the atropic acid derivative, forming a carbon-centered radical intermediate that is subsequently trapped by molecular oxygen. This oxygenation step leads to the formation of a peroxyl radical species, which eventually undergoes fragmentation and decarboxylation to yield the target beta-ketosulfone product with the extrusion of carbon dioxide. Isotope labeling experiments using H2O-18 have confirmed that the carbonyl oxygen atom in the final product originates from molecular oxygen rather than the solvent, underscoring the dual role of oxygen as both an oxidant and an oxygen donor. The mild nature of this radical pathway minimizes side reactions such as polymerization or over-oxidation, resulting in a cleaner impurity profile compared to harsh thermal or metal-catalyzed alternatives. Understanding these mechanistic nuances allows process chemists to fine-tune reaction parameters such as light intensity and oxygen flow to maximize throughput and minimize the formation of trace impurities.
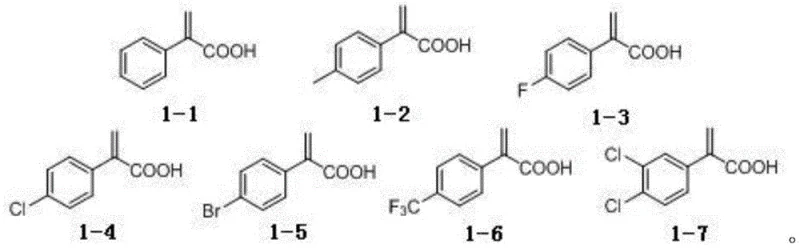
How to Synthesize Beta-Ketosulfone Efficiently
The practical implementation of this visible light-mediated protocol offers a straightforward and scalable pathway for the production of beta-ketosulfone derivatives, suitable for both laboratory discovery and pilot plant operations. The procedure begins with the dissolution of the atropic acid substrate and the corresponding sulfonyl hydrazide in a optimized mixture of acetonitrile and water, followed by the addition of catalytic amounts of fluorescein, an inorganic base such as sodium bicarbonate, and potassium iodide. The reaction vessel is then sealed and connected to an oxygen source to maintain a positive pressure of oxygen throughout the irradiation period, typically using a standard 23W compact fluorescent lamp positioned at a fixed distance to ensure uniform light exposure. Monitoring the reaction progress via thin-layer chromatography (TLC) allows for precise determination of the endpoint, after which the organic solvent is removed under reduced pressure and the crude product is isolated through standard aqueous workup and extraction techniques. For detailed operational parameters and specific stoichiometric ratios optimized for different substrates, please refer to the standardized synthesis guide below.
- Dissolve atropic acid, sulfonyl hydrazide, fluorescein photocatalyst, inorganic base, and potassium iodide in a mixed solvent of acetonitrile and water.
- Seal the reaction vessel, connect an oxygen balloon to maintain an oxygen atmosphere, and irradiate the mixture with a 23W compact fluorescent lamp at room temperature.
- Upon completion monitored by TLC, remove the organic solvent, extract the aqueous residue with ethyl acetate, wash with brine, dry over sodium sulfate, and purify via column chromatography.
Commercial Advantages for Procurement and Supply Chain Teams
From a strategic procurement perspective, the adoption of this metal-free photocatalytic technology presents substantial opportunities for cost reduction in pharmaceutical intermediate manufacturing by eliminating the dependency on volatile precious metal markets. The substitution of expensive iridium or silver catalysts with commodity-grade organic dyes like fluorescein drastically lowers the raw material cost per kilogram of product, while the removal of heavy metal scavenging agents reduces the overall bill of materials and waste disposal fees. Furthermore, the operational simplicity of running reactions at ambient temperature and pressure without the need for specialized inert atmosphere gloveboxes or high-pressure autoclaves translates to lower capital investment requirements for reactor infrastructure. The use of molecular oxygen as a stoichiometric oxidant not only enhances the atom economy of the process but also mitigates the safety risks associated with storing and handling large quantities of hazardous peroxide oxidants, thereby lowering insurance premiums and safety compliance costs. This streamlined process flow facilitates faster batch turnover times and reduces the burden on quality assurance teams who would otherwise need to validate complex metal removal steps.
- Cost Reduction in Manufacturing: The elimination of transition metal catalysts removes the necessity for expensive metal scavengers and extensive purification protocols, leading to direct savings in raw material and processing costs. By utilizing inexpensive organic photocatalysts and common inorganic bases, the overall cost of goods sold (COGS) is significantly optimized, making the final intermediate more competitive in the global market. Additionally, the avoidance of hazardous chemical oxidants reduces the costs associated with special waste treatment and regulatory compliance, contributing to a leaner and more efficient production model.
- Enhanced Supply Chain Reliability: The reliance on widely available, commodity chemicals such as atropic acid derivatives and sulfonyl hydrazides ensures a stable and resilient supply chain that is less susceptible to geopolitical disruptions or shortages of specialized reagents. The robustness of the reaction conditions, including tolerance to water and operation under ambient pressure, allows for flexible manufacturing across different facilities without the need for highly customized equipment, thereby diversifying sourcing options and reducing lead times. This flexibility empowers supply chain managers to respond more agilely to fluctuating market demands and secure long-term contracts with greater confidence in delivery continuity.
- Scalability and Environmental Compliance: The green chemistry attributes of this method, characterized by the use of oxygen and the generation of benign by-products, align perfectly with increasingly stringent environmental regulations and corporate sustainability goals. The process is inherently scalable, as the photon flux can be managed through flow chemistry reactors or large-scale batch vessels equipped with efficient lighting arrays, ensuring consistent quality from gram to ton scale. The reduction in hazardous waste generation simplifies the environmental permitting process and enhances the company's reputation as a responsible manufacturer, which is a critical factor for partnerships with top-tier multinational pharmaceutical corporations.
Frequently Asked Questions (FAQ)
The following questions address common technical inquiries regarding the implementation and optimization of this visible light-mediated synthesis, providing clarity for process development teams evaluating this technology for adoption. These insights are derived directly from the experimental data and mechanistic studies presented in the patent literature, ensuring that the information provided is grounded in verified scientific evidence. Understanding these details is crucial for assessing the feasibility of integrating this method into existing production workflows and for troubleshooting potential challenges during scale-up activities.
Q: What are the primary advantages of this visible light method over traditional transition metal catalysis?
A: This method eliminates the need for expensive and toxic transition metal catalysts like silver or iridium complexes. It utilizes inexpensive organic dyes like fluorescein and molecular oxygen as a green oxidant, significantly reducing heavy metal residue concerns and purification costs.
Q: Is the reaction sensitive to moisture or air?
A: Unlike many photo-mediated reactions requiring strict inert atmospheres, this protocol operates efficiently under an oxygen atmosphere and utilizes a mixed solvent system containing water (MeCN/H2O), demonstrating robust tolerance to moisture which simplifies operational requirements.
Q: What is the substrate scope for the sulfonyl hydrazide component?
A: The method exhibits broad substrate tolerance, successfully accommodating electron-rich and electron-deficient aromatic sulfonyl hydrazides, as well as heteroaryl and alkyl variants, although steric hindrance at the ortho-position may slightly reduce yields.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Beta-Ketosulfone Supplier
As a leader in the custom synthesis and manufacturing of complex organic intermediates, NINGBO INNO PHARMCHEM is uniquely positioned to leverage this advanced visible light-mediated technology to deliver high-quality beta-ketosulfone compounds to our global clientele. Our state-of-the-art R&D facility is equipped with specialized photochemical reactors and possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that we can meet your volume requirements with consistency and precision. We maintain stringent purity specifications through our rigorous QC labs, utilizing advanced analytical techniques to verify the absence of metal residues and confirm the structural integrity of every batch, guaranteeing that our products meet the highest standards required for pharmaceutical applications.
We invite you to collaborate with our technical team to explore how this innovative synthetic route can optimize your supply chain and reduce your overall manufacturing costs. Please contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your specific project needs. We are ready to provide specific COA data and comprehensive route feasibility assessments to demonstrate the viability of this metal-free approach for your next-generation drug candidates.
