Scalable Chemical Resolution Technology for High-Purity Donepezil Intermediates
The pharmaceutical industry continuously seeks robust methodologies for the production of chiral active pharmaceutical ingredients, particularly for neurodegenerative treatments where stereochemistry dictates efficacy. Patent CN103497145A introduces a pivotal advancement in the preparation of optically pure donepezil, a critical acetylcholinesterase inhibitor used in Alzheimer's therapy. This technology shifts the paradigm from expensive chromatographic separations to a more industrially viable chemical resolution process using chiral tartaric acid derivatives. By leveraging diastereomeric salt formation, this method ensures high optical purity while maintaining operational simplicity. For global procurement teams and R&D directors, understanding this patent is essential for securing a reliable API intermediate supplier capable of delivering consistent quality. The transition to this resolution technology represents a significant opportunity for cost reduction in pharmaceutical manufacturing without compromising the stringent purity specifications required for clinical applications.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the isolation of single-enantiomer donepezil has relied heavily on preparative high-performance liquid chromatography (HPLC) or capillary electrophoresis, which present substantial bottlenecks for commercial scale-up of complex pharmaceutical intermediates. These chromatographic techniques, while effective for analytical purposes, suffer from low throughput, high solvent consumption, and the prohibitive cost of chiral stationary phases when applied to ton-scale production. Furthermore, the operational complexity of maintaining chiral columns and the difficulty in recycling mobile phases often lead to extended lead times and inflated manufacturing expenses. For a supply chain head, relying on such methods introduces significant risk regarding supply continuity and cost stability, as any disruption in column availability or solvent supply can halt production entirely. Consequently, the industry has long sought an alternative that balances high enantiomeric excess with economic feasibility and scalability.
The Novel Approach
The novel approach detailed in the patent utilizes a chemical resolution strategy that fundamentally simplifies the production workflow by employing readily available chiral tartaric acid derivatives as resolving agents. Instead of relying on physical separation on a column, this method exploits the differences in solubility between diastereomeric salts formed between the racemic donepezil and the chiral acid. This shift allows for standard crystallization equipment to be used, drastically reducing capital expenditure and operational complexity. The process is designed to be robust, operating effectively within a temperature range of 60-100°C during the salt formation phase, which is easily manageable in standard glass-lined or stainless steel reactors. By eliminating the need for specialized chromatographic infrastructure, manufacturers can achieve reducing lead time for high-purity pharmaceutical intermediates while simultaneously enhancing the overall sustainability of the production process through solvent recycling.
Mechanistic Insights into Chiral Tartaric Acid Resolution
The core of this technology lies in the precise molecular recognition between the racemic donepezil base and the chiral tartaric acid derivative, leading to the selective precipitation of one diastereomeric salt over the other. The resolving agent, typically an O,O'-dibenzoyl tartaric acid derivative, interacts with the basic nitrogen of the piperidine ring in donepezil to form an ionic salt. Due to the three-dimensional arrangement of the atoms, the salt formed with one enantiomer of donepezil possesses a crystal lattice energy that favors precipitation under specific solvent conditions, while the other remains in the mother liquor. This thermodynamic preference is the driving force behind the separation, allowing for the isolation of the desired isomer with high fidelity. Understanding this mechanism is crucial for R&D teams aiming to optimize the process further, as subtle changes in solvent polarity or cooling rates can influence the nucleation and growth of the crystals, thereby impacting the final optical purity.
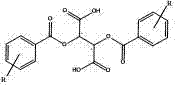
Following the initial resolution, the process incorporates a rigorous recrystallization protocol to elevate the optical purity to pharmaceutical grades, specifically targeting levels above 98% ee. The crude diastereomeric salt is dissolved in a suitable crystallization solvent, such as ethyl acetate or alcohols, and allowed to crystallize under controlled conditions. This step is critical for impurity control, as it effectively excludes the unwanted enantiomer and other organic impurities from the crystal lattice. The patent data indicates that multiple recrystallization cycles can be employed if necessary, although the efficiency of the initial resolution often minimizes the need for excessive processing. Once the salt reaches the desired purity, it undergoes alkalization at a pH of 7.5-10 to liberate the free base, which is then extracted into an organic phase. This multi-stage purification ensures that the final high-purity donepezil intermediate meets the rigorous quality standards expected by regulatory bodies and downstream formulators.
How to Synthesize Optically Pure Donepezil Efficiently
Implementing this synthesis route requires careful attention to solvent selection and temperature control to maximize yield and optical purity. The process begins with the dissolution of racemic donepezil in a solvent like ethyl acetate, followed by the addition of the chiral resolving agent at elevated temperatures to ensure complete salt formation. Detailed standard operating procedures are essential to maintain consistency across batches, particularly during the cooling and filtration stages where the physical separation occurs. The standardized synthesis steps outlined in the technical documentation provide a clear roadmap for process engineers to follow, ensuring that the critical parameters such as molar ratios and pH levels are strictly adhered to. For partners looking to adopt this technology, having access to these validated protocols is invaluable for accelerating technology transfer and minimizing scale-up risks.
- React racemic donepezil with a chiral tartaric acid resolving agent in a solvent at 60-100°C to form diastereomeric salts.
- Recrystallize the precipitated salt multiple times to enhance optical purity to over 98% ee.
- Alkalize the purified salt to pH 7.5-10 and extract with organic solvent to isolate the free optically pure base.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, this resolution technology offers profound advantages that directly address the pain points of modern pharmaceutical supply chains, particularly regarding cost efficiency and material availability. By replacing chromatographic methods with crystallization-based separation, the process significantly reduces the consumption of expensive solvents and eliminates the need for costly chiral columns, leading to substantial cost savings in raw material procurement. Additionally, the ability to recover and recycle the chiral resolving agent with high efficiency means that the effective cost per kilogram of the active ingredient is drastically lowered over time. For procurement managers, this translates into a more stable pricing structure and reduced vulnerability to market fluctuations in specialized reagents. The simplicity of the equipment requirements also means that production can be scaled up rapidly without significant capital investment, ensuring that supply can meet demand spikes without compromising quality.
- Cost Reduction in Manufacturing: The elimination of transition metal catalysts and expensive chromatographic media removes significant cost centers from the manufacturing budget, allowing for a more competitive pricing model. The use of common organic solvents and inexpensive tartaric acid derivatives further drives down the variable costs associated with production. Moreover, the high recovery rate of the resolving agent means that the effective consumption of this key chiral pool material is minimized, contributing to long-term economic sustainability. This economic efficiency makes the process highly attractive for generic drug manufacturers looking to optimize their margins while maintaining high quality standards.
- Enhanced Supply Chain Reliability: The reliance on readily available commodity chemicals rather than specialized proprietary reagents enhances the resilience of the supply chain against disruptions. Since the resolving agents and solvents are sourced from broad chemical markets, the risk of single-source dependency is significantly mitigated. This availability ensures that production schedules can be maintained consistently, reducing the lead time for high-purity pharmaceutical intermediates and preventing stockouts. For supply chain heads, this reliability is critical for maintaining uninterrupted drug supply to patients and meeting contractual obligations with downstream partners.
- Scalability and Environmental Compliance: The process is inherently scalable, moving seamlessly from laboratory benchtop to multi-ton commercial production without the need for complex engineering redesigns. The reduced solvent usage and the ability to recycle aqueous waste streams align with modern green chemistry principles, minimizing the environmental footprint of the manufacturing process. This compliance with environmental regulations reduces the burden of waste disposal and associated costs, making the facility more sustainable. The robustness of the crystallization process also ensures consistent product quality across different batch sizes, which is essential for regulatory approval and commercial success.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this resolution technology, based on the specific advantages and operational details found in the patent literature. These insights are designed to clarify the feasibility of the process for potential partners and to highlight the key differentiators compared to traditional synthesis routes. Understanding these nuances is vital for making informed decisions about technology adoption and supply chain integration. The answers provided reflect the technical reality of the process as described in the intellectual property documentation.
Q: What is the primary advantage of this resolution method over chromatography?
A: This chemical resolution method avoids the high costs and low throughput associated with preparative HPLC, offering a significantly more scalable and cost-effective route for industrial production.
Q: Can the chiral resolving agent be recovered?
A: Yes, the process allows for the recovery and recycling of the chiral tartaric acid resolving agent with high efficiency, reducing material waste and overall production costs.
Q: What optical purity can be achieved with this process?
A: Through optimized recrystallization steps, the process consistently achieves optical purity levels exceeding 98% ee, meeting stringent pharmaceutical standards.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Donepezil Supplier
NINGBO INNO PHARMCHEM stands at the forefront of translating complex patent technologies into commercial reality, offering extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production. Our technical team possesses the expertise to adapt the chiral resolution process described in CN103497145A to meet your specific volume and purity requirements, ensuring stringent purity specifications are met consistently. With rigorous QC labs and a commitment to process optimization, we guarantee that every batch of donepezil intermediate delivered meets the highest international standards. Our infrastructure is designed to handle the specific solvent and temperature requirements of this resolution chemistry, ensuring a seamless transition from development to full-scale manufacturing.
We invite you to engage with our technical procurement team to discuss how this technology can optimize your supply chain and reduce your overall cost of goods. By requesting a Customized Cost-Saving Analysis, you can gain a clear understanding of the economic benefits specific to your project volume. We encourage potential partners to contact us for specific COA data and route feasibility assessments to verify our capability to deliver high-quality optically pure donepezil. Let us collaborate to bring this efficient and scalable solution to your production line, ensuring a reliable supply of this critical Alzheimer's therapeutic intermediate.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
