Industrial Scale Paclitaxel Semi-Synthesis: Advanced Catalytic Technology for Global API Supply
The pharmaceutical industry continuously seeks robust methodologies for the production of critical oncology therapeutics, and the semi-synthesis of Paclitaxel remains a cornerstone of this endeavor. Patent CN101863862B introduces a transformative approach to this complex chemical challenge, detailing a method for the semi-synthesis of Paclitaxel on an industrialized basis that fundamentally alters the traditional synthetic sequence. This innovation leverages a high-efficiency specific catalyst, Cerium Chloride hexahydrate (CeCl3.6H2O), to achieve selective acetylation of the 10-hydroxyl group in 10-deacetyl baccatin III (10-DAB). By breaking away from the conventional sequence of protecting the 7-position hydroxyl prior to acetylation, this technology offers a streamlined pathway that significantly enhances reaction efficiency and product purity. The implications for global supply chains are profound, as this method facilitates the reliable production of high-purity Active Pharmaceutical Ingredients (APIs) while mitigating the technical bottlenecks associated with traditional multi-step syntheses. For R&D Directors and Supply Chain Heads, understanding the mechanistic advantages of this patent is crucial for evaluating long-term sourcing strategies and ensuring the continuity of essential cancer treatments.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditional semi-synthetic routes for Paclitaxel have long been plagued by inherent inefficiencies that hinder large-scale commercial viability. Conventional methodologies typically necessitate the protection of the C-7 hydroxyl group before the acetylation of the C-10 hydroxyl can occur, primarily due to the higher reactivity of the C-7 position. This sequential requirement introduces additional synthetic steps, each carrying the risk of yield loss and impurity generation. Furthermore, standard protection groups often require harsh conditions for installation and removal, leading to extended reaction times and the formation of difficult-to-remove by-products. Many existing processes rely on inert gas protection and strict temperature controls, which escalate operational costs and complicate reactor management in a manufacturing setting. The cumulative effect of these limitations is a process that is not only costly but also fragile, with variability in yield and purity that can disrupt the supply of this critical oncology medication. Consequently, procurement teams face challenges in securing consistent volumes of high-purity Paclitaxel intermediates at competitive price points.
The Novel Approach
In stark contrast, the novel approach detailed in the patent data revolutionizes the synthetic landscape by utilizing a specific catalytic system to invert the traditional reaction order. By employing CeCl3.6H2O, the process achieves direct and selective acetylation of the C-10 hydroxyl in 10-DAB without prior protection of the C-7 position. This strategic shift eliminates entire classes of side reactions and reduces the total number of unit operations required to reach the final API. The reaction proceeds under normal pressure and at room temperature, removing the need for expensive inert atmosphere equipment and energy-intensive cooling or heating cycles. This simplification translates directly into enhanced process robustness and scalability, allowing for the commercial scale-up of complex pharmaceutical intermediates with greater ease. The ability to conduct these transformations under mild conditions also preserves the integrity of the sensitive taxane core, ensuring that the final product meets the rigorous quality standards demanded by regulatory bodies worldwide.
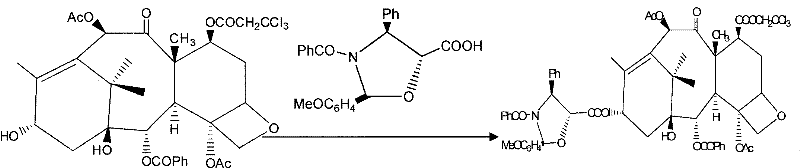
Mechanistic Insights into CeCl3-Catalyzed Selective Acetylation
The core of this technological breakthrough lies in the unique interaction between the Cerium catalyst and the substrate molecules, which dictates the exceptional regioselectivity observed in the acetylation step. In the absence of the catalyst, acetylating agents would indiscriminately react with both the C-7 and C-10 hydroxyl groups due to their comparable nucleophilicity, leading to a mixture of mono- and di-acetylated by-products that are difficult to separate. However, the presence of CeCl3.6H2O coordinates specifically with the oxygen atoms in the vicinity of the C-10 hydroxyl, effectively activating this position while sterically or electronically shielding the C-7 hydroxyl from reaction. This coordination complex lowers the activation energy for the C-10 acetylation specifically, allowing the reaction to proceed rapidly at room temperature with yields exceeding 95%. Such high selectivity is rare in complex natural product synthesis and represents a significant advancement in catalytic methodology. For R&D teams, this mechanism offers a blueprint for designing cleaner synthesis routes that minimize waste and maximize atom economy, aligning with modern green chemistry principles.
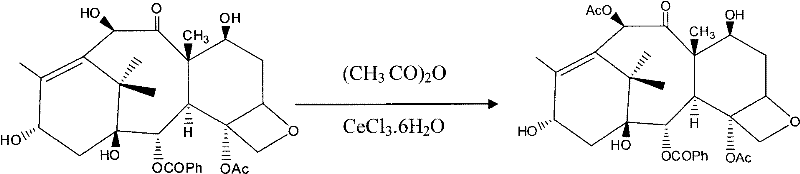
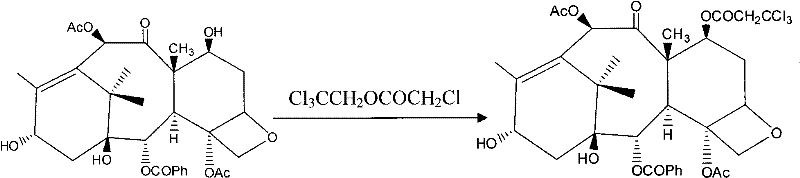
Beyond the initial acetylation, the subsequent protection and coupling steps are equally optimized to maintain high purity and yield throughout the synthetic sequence. The protection of the C-7 hydroxyl using trichloroethyl chloroformate is conducted under mild conditions that prevent degradation of the newly formed C-10 acetate group. This orthogonal protection strategy ensures that each functional group is manipulated independently without affecting the others, a critical factor in maintaining the structural integrity of the taxane skeleton. Furthermore, the final deprotection and ring-opening steps utilize acetone as a solvent, which facilitates rapid reaction kinetics and simplifies the isolation of the final Paclitaxel product. The impurity profile is tightly controlled because the specific reaction conditions minimize the formation of regio-isomers and degradation products. This level of control over the杂质谱 (impurity profile) is essential for meeting the stringent specifications required for injectable oncology drugs, ensuring patient safety and therapeutic efficacy without the need for extensive downstream purification processes.
How to Synthesize Paclitaxel Efficiently
Implementing this advanced semi-synthesis route requires a precise understanding of the reaction parameters and material inputs to ensure consistent quality and yield. The process begins with the dissolution of 10-DAB in anhydrous tetrahydrofuran, followed by the addition of the cerium catalyst and acetic anhydride under controlled stirring. Detailed standard operating procedures dictate the exact molar ratios and temperature profiles necessary to replicate the high yields reported in the patent data. Following the acetylation, the intermediate Baccatin III is isolated and subjected to the protection step using trichloroethyl chloroformate in methylene chloride. The subsequent esterification with the oxazoline side chain and the final deprotection involve careful pH control and temperature management to ensure complete conversion. While the general workflow is simplified compared to traditional methods, adherence to the specific catalytic conditions is paramount for success. The detailed standardized synthesis steps see the guide below for a comprehensive breakdown of the operational protocol.
- Acetylate 10-hydroxyl in 10-DAB using CeCl3.6H2O catalyst to obtain Baccatin III with high selectivity.
- Protect the C-7-hydroxyl of Baccatin III using trichloroethyl chloroformate under mild conditions.
- Conduct esterification between protected Baccatin III and the oxazoline side chain carboxylate.
- Open the oxazole ring and simultaneously remove the 7-protective group to yield final Paclitaxel.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain leaders, the adoption of this catalytic semi-synthesis method offers substantial strategic benefits that extend beyond mere technical novelty. The elimination of inert gas requirements and the ability to operate at ambient pressure and temperature drastically reduce the capital expenditure and operational costs associated with manufacturing infrastructure. This process intensification allows for higher throughput in existing facilities without the need for specialized high-pressure reactors or cryogenic cooling systems. Moreover, the high selectivity of the catalyst minimizes the consumption of raw materials by reducing the formation of waste by-products, leading to significant cost savings in reagent procurement. The simplified purification process further reduces the demand for expensive chromatography resins and solvents, contributing to a leaner and more cost-effective production model. These efficiencies collectively enhance the competitiveness of the supply chain, enabling the delivery of high-purity pharmaceutical intermediates at a more sustainable price point.
- Cost Reduction in Manufacturing: The streamlined synthetic route eliminates multiple protection and deprotection steps, which directly translates to reduced labor hours and lower utility consumption per kilogram of product. By avoiding the use of expensive transition metal catalysts that require complex removal procedures, the process simplifies the downstream processing workflow. The high yield of each individual step ensures that less starting material is wasted, optimizing the overall material balance and reducing the cost of goods sold. These qualitative improvements in process efficiency allow for a more aggressive pricing strategy while maintaining healthy margins, providing a distinct advantage in the competitive global API market.
- Enhanced Supply Chain Reliability: The robustness of the reaction conditions means that production is less susceptible to disruptions caused by equipment failure or utility fluctuations. Operating without the need for inert gas protection removes a common bottleneck in chemical manufacturing, ensuring that production schedules can be met consistently. The availability of raw materials such as 10-DAB and the catalyst is stable, and the process tolerance reduces the risk of batch failures due to minor parameter deviations. This reliability is crucial for maintaining continuous supply to downstream formulation partners, minimizing the risk of stockouts for critical cancer medications and strengthening the trust between suppliers and pharmaceutical clients.
- Scalability and Environmental Compliance: The use of common solvents and mild reaction conditions facilitates easy scale-up from pilot plant to commercial production volumes without significant re-engineering. The reduction in by-product formation inherently lowers the environmental burden, simplifying waste treatment and disposal protocols. This alignment with green chemistry principles supports corporate sustainability goals and ensures compliance with increasingly stringent environmental regulations. The ability to scale complex synthetic pathways efficiently ensures that supply can grow in tandem with market demand, providing a secure foundation for long-term business growth and partnership.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this semi-synthesis technology. They are derived from the specific advantages and operational details outlined in the patent documentation to provide clarity for potential partners. Understanding these aspects is vital for assessing the feasibility of integrating this route into your existing supply chain or R&D pipeline. The answers reflect the proven capabilities of the method to deliver high-quality results under industrial conditions.
Q: How does the CeCl3 catalyst improve Paclitaxel synthesis yield?
A: The CeCl3.6H2O catalyst provides high specificity for the C-10 hydroxyl group, preventing unwanted acetylation at the C-7 position. This selectivity eliminates the need for complex protection-deprotection sequences initially, resulting in single-step reaction yields exceeding 90% and significantly reducing by-product formation.
Q: Is this semi-synthesis method suitable for large-scale commercial production?
A: Yes, the process is designed for industrialization. It operates at normal pressure and room temperature without the need for inert gas protection. The simplified workflow reduces reaction times and operational complexity, making it highly scalable for metric-ton annual production capacities.
Q: What are the purity advantages of this novel synthetic route?
A: By reversing the traditional order of protection and acetylation, this method minimizes side reactions. The specific catalytic conditions and the use of acetone in the final deprotection step ensure fewer impurities, often eliminating the need for chromatography purification and ensuring stringent purity specifications for pharmaceutical use.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Paclitaxel Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of reliable supply chains for life-saving oncology medications like Paclitaxel. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that we can meet the volumetric demands of global pharmaceutical companies. We are committed to delivering products that meet stringent purity specifications through our rigorous QC labs and advanced analytical capabilities. By leveraging advanced catalytic technologies such as the CeCl3-mediated semi-synthesis route, we optimize our manufacturing processes to provide cost-effective solutions without compromising on quality. Our dedication to technical excellence positions us as a strategic partner capable of navigating the complexities of API manufacturing.
We invite you to discuss how our manufacturing capabilities can support your specific project requirements and help optimize your supply chain. Contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your volume needs. We are prepared to provide specific COA data and route feasibility assessments to demonstrate how our advanced processes can enhance your product portfolio. Let us collaborate to ensure a stable and efficient supply of high-quality Paclitaxel for your patients.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
