Advanced Semi-Synthetic Route for Baccatin III Derivatives: Technical Upgrade and Commercial Scalability
Advanced Semi-Synthetic Route for Baccatin III Derivatives: Technical Upgrade and Commercial Scalability
The global demand for next-generation antineoplastic agents continues to drive innovation in the synthesis of complex taxane intermediates. Patent CN100349882C discloses a robust and highly efficient method for preparing 14-beta-hydroxy-1,14-carbonate-deacetylbaccatin III, a critical precursor for novel taxane derivatives with potent anti-tumor activity. This technology represents a paradigm shift from relying on scarce natural extraction to a scalable semi-synthetic approach starting from 10-deacetylbaccatin III. By leveraging abundant raw materials and optimizing reaction conditions, this process addresses key bottlenecks in the supply chain of high-value pharmaceutical intermediates. For R&D directors and procurement specialists, understanding the nuances of this pathway is essential for securing a stable supply of high-purity taxane building blocks.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the production of advanced taxane derivatives has been severely constrained by the availability of starting materials. Traditional methods often relied on the direct extraction of 14-beta-hydroxy-baccatin III from the leaves of Taxus wallichiana, a process characterized by extremely low yields and significant ecological impact. The scarcity of this natural precursor created a volatile market environment, leading to unpredictable pricing and supply interruptions for downstream API manufacturers. Furthermore, purification of natural extracts often involves complex chromatography to remove structurally similar impurities, driving up operational costs and extending lead times. These inherent limitations of biological sourcing made it difficult to achieve the consistent quality and volume required for commercial-scale drug development.
The Novel Approach
The methodology outlined in CN100349882C circumvents these challenges by utilizing 10-deacetylbaccatin III (10-DAB III) as the starting material, which can be isolated in large quantities from the renewable needles of Taxus baccata. This strategic shift transforms the production model from extraction-dependent to synthesis-driven. The novel route employs a sophisticated protection strategy at the C-7 and C-10 positions, allowing for selective functionalization at C-13 and C-14 without affecting other sensitive moieties. By introducing trichloroacetyl or silyl protecting groups, the process achieves high regioselectivity, enabling subsequent oxidation and carbonate formation steps to proceed with superior efficiency. This approach not only secures the raw material supply but also enhances the overall process mass intensity.
Mechanistic Insights into Regioselective Oxidation and Carbonate Formation
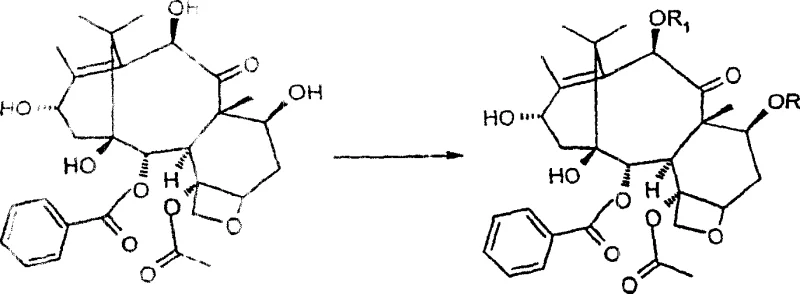
A critical aspect of this synthesis is the controlled oxidation of the C-13 position followed by hydroxylation at C-14. The patent details the use of activated manganese dioxide (MnO2) in solvents such as acetonitrile or acetone to effect this transformation. The reaction kinetics are carefully managed; initial rapid oxidation yields the 13-dehydro derivative, which upon prolonged stirring slowly converts to the desired 14-beta-hydroxy-13-dehydro intermediate. This two-step oxidation sequence is pivotal for installing the oxygen functionality required for the subsequent carbonate bridge. The choice of MnO2 is particularly advantageous due to its mildness and selectivity, preventing over-oxidation or degradation of the delicate taxane core structure.
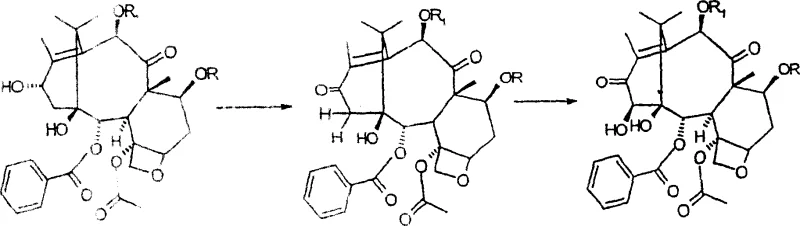
Following oxidation, the formation of the 1,14-carbonate ring is achieved using phosgene or triphosgene in the presence of pyridine. This cyclization step locks the conformation of the A-ring, which is crucial for the biological activity of the final taxane derivatives. The subsequent reduction of the C-13 carbonyl using sodium borohydride in methanol is remarkably stereoselective, yielding almost exclusively the 13-alpha epimer. This stereocontrol is vital, as the configuration at C-13 influences the pharmacological profile of the resulting compounds. The mechanism ensures that the C-9 carbonyl remains untouched, demonstrating the high chemoselectivity of the reducing conditions employed in this protocol.
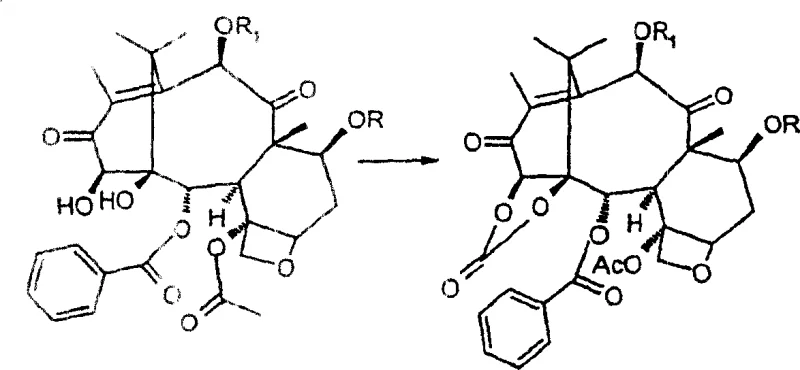
How to Synthesize 14-beta-Hydroxy-1,14-Carbonate-Deacetylbaccatin III Efficiently
Executing this synthesis requires precise control over reaction parameters to maximize yield and purity. The process begins with the selective protection of the C-7 and C-10 hydroxyl groups, typically using trichloroacetic anhydride with a catalytic amount of DMAP to avoid excessive pyridine usage. Following protection, the oxidation sequence with MnO2 must be monitored closely via TLC to ensure complete conversion to the 14-hydroxy species without decomposition. The carbonate formation and reduction steps follow, culminating in a final deprotection stage to reveal the target intermediate. For detailed operational parameters and safety guidelines, refer to the standardized synthesis protocol below.
- Protect the 7-position and 10-position hydroxyl groups of 10-deacetylbaccatin III using trichloroacetic anhydride or chlorides with a catalytic amount of DMAP.
- Perform a two-step oxidation using activated Manganese Dioxide to oxidize the 13-position and hydroxylate the 14-position.
- Form the 1,14-carbonate ester using phosgene or triphosgene, followed by regioselective reduction of the 13-carbonyl and final deprotection.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, this patented process offers substantial benefits for organizations aiming to optimize their taxane supply chains. By shifting the reliance from scarce natural extracts to abundant semi-synthetic precursors, manufacturers can mitigate the risks associated with agricultural variability and geopolitical sourcing issues. The use of 10-DAB III, a commodity chemical in the taxane industry, ensures a stable and predictable cost base. Furthermore, the streamlined reaction sequence reduces the number of purification steps required, directly translating to lower manufacturing costs and faster throughput times for high-volume production.
- Cost Reduction in Manufacturing: The elimination of stoichiometric pyridine in favor of catalytic DMAP and triethylamine significantly reduces solvent costs and waste disposal expenses. Additionally, the high yields reported in the oxidation and reduction steps minimize material loss, enhancing the overall economic viability of the process. By avoiding expensive and hazardous reagents where possible, the process aligns with green chemistry principles while driving down the cost of goods sold for complex pharmaceutical intermediates.
- Enhanced Supply Chain Reliability: Sourcing 10-deacetylbaccatin III from Taxus baccata needles provides a renewable and scalable feedstock compared to the limited availability of Taxus wallichiana extracts. This abundance ensures that production schedules can be maintained without interruption, even during periods of high market demand. The robustness of the synthetic route also allows for flexible manufacturing strategies, enabling suppliers to respond quickly to changing procurement requirements from global API producers.
- Scalability and Environmental Compliance: The reaction conditions described are amenable to large-scale implementation, with straightforward workup procedures involving standard extraction and crystallization techniques. The reduction in hazardous solvent usage and the potential for solvent recovery contribute to a lower environmental footprint. This compliance with stringent environmental regulations facilitates smoother regulatory approvals and supports sustainable manufacturing initiatives within the pharmaceutical sector.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the synthesis of baccatin III derivatives as described in the patent literature. These insights are derived from the specific experimental data and process advantages detailed in CN100349882C, providing clarity on the feasibility and benefits of this methodology for industrial application.
Q: What is the primary advantage of using 10-deacetylbaccatin III over natural 14-beta-hydroxy baccatin III?
A: 10-deacetylbaccatin III can be extracted in large quantities from the needles of European yew (Taxus baccata), whereas 14-beta-hydroxy baccatin III is a scarce natural compound found in low yields in Taxus wallichiana, making the semi-synthetic route far more scalable and cost-effective.
Q: How does the patent address environmental concerns regarding pyridine usage?
A: The process utilizes a catalytic amount of DMAP (4-dimethylaminopyridine) with triethylamine instead of stoichiometric pyridine for acylation, significantly reducing solvent residue issues and simplifying waste treatment.
Q: What ensures the stereoselectivity at the C-13 position during reduction?
A: The reduction of the 13-carbonyl group using sodium borohydride in methanol is highly regioselective and stereoselective, yielding almost exclusively the 13-alpha epimer while leaving the 9-position carbonyl unchanged.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 14-beta-Hydroxy-1,14-Carbonate-Deacetylbaccatin III Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of high-quality intermediates in the development of life-saving oncology therapies. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the complex chemistry involved in taxane synthesis is executed with precision and consistency. We adhere to stringent purity specifications and operate rigorous QC labs to guarantee that every batch of 14-beta-hydroxy-1,14-carbonate-deacetylbaccatin III meets the highest international standards. Our commitment to technical excellence allows us to deliver reliable solutions for your most challenging synthesis projects.
We invite you to collaborate with us to optimize your supply chain and reduce costs through advanced process chemistry. Contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We are prepared to provide specific COA data and comprehensive route feasibility assessments to support your R&D and manufacturing goals. Let us be your partner in bringing innovative cancer treatments to market faster and more efficiently.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
