Optimizing Docetaxel Production: A Novel Oxazoline-Mediated Semi-Synthetic Strategy
Introduction to Advanced Docetaxel Manufacturing
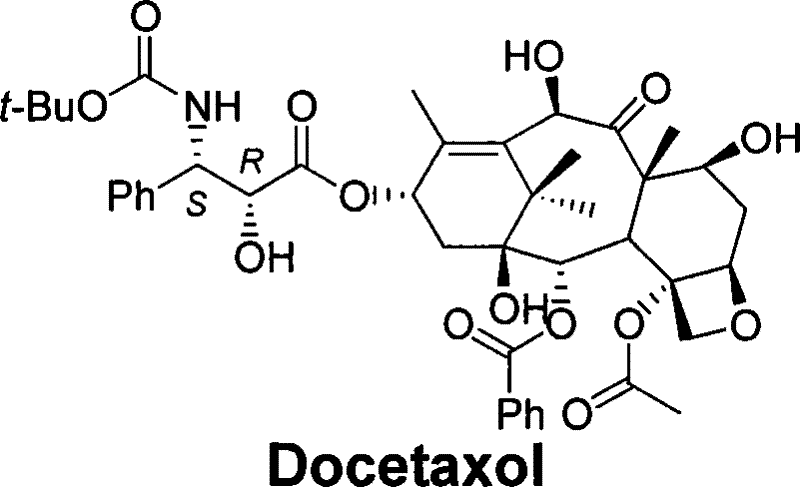
The global demand for high-purity oncology therapeutics continues to drive innovation in the semi-synthesis of complex natural product derivatives. Patent CN101468974B discloses a breakthrough semi-synthetic method for producing Docetaxel, a potent antitumor agent with activity significantly higher than paclitaxel. Unlike total synthesis, which faces immense challenges due to the molecule's numerous chiral centers and functional groups, this semi-synthetic approach leverages 10-deacetylbaccatin III (10-DAB) as a renewable starting material. The core innovation lies in the utilization of a novel oxazoline chiral side chain, which fundamentally alters the coupling and deprotection landscape. By employing specific halogenated acyl groups, the process achieves selective ring opening under mild acidic conditions, effectively bypassing the notorious acyl migration issues that plague conventional alkaline hydrolysis methods. This technical advancement represents a significant leap forward for any reliable pharmaceutical intermediate supplier seeking to optimize yield and purity profiles.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the semi-synthesis of Docetaxel has been categorized into two primary methodologies, both of which present distinct operational hazards and purity challenges for industrial manufacturing. The first conventional class involves the condensation of protected 10-DAB with a five-membered oxazole alkanoic acid side chain, followed by hydrolysis and deprotection. While conceptually straightforward, this route often struggles with the stability of the side chain precursors. The second, more common class utilizes a beta-lactam ring opening strategy under highly basic conditions, typically employing reagents like butyllithium at extremely low temperatures. This approach necessitates rigorous cryogenic control and introduces significant safety risks associated with pyrophoric reagents. Furthermore, the alkaline environment required for beta-lactam opening frequently induces acyl migration, where the acyl group shifts from the oxygen to the amino group on the side chain. This side reaction generates persistent impurities that are notoriously difficult to separate, thereby compromising the overall yield and increasing the cost of goods sold through extensive purification requirements.
The Novel Approach
The methodology outlined in CN101468974B offers a transformative alternative by replacing the beta-lactam or simple oxazole motifs with a specialized oxazoline ring system bearing trifluoroacetyl or tribromoacetyl groups. This structural modification is pivotal because these halogenated acyl groups exhibit unique lability; they are stable enough to survive the initial coupling reaction but are highly susceptible to cleavage under acidic or near-neutral conditions. This allows the oxazoline ring to be opened using diluted acids in alcoholic solutions, completely avoiding the harsh alkaline environments that trigger acyl migration. Consequently, the nitrogen atom remains available for subsequent Boc protection without the interference of migrated acyl byproducts. This shift from basic to acidic processing conditions not only enhances the chemical purity of the intermediate stream but also simplifies the reactor engineering requirements, eliminating the need for extreme cryogenic setups and enabling more robust cost reduction in API manufacturing.
Mechanistic Insights into Oxazoline-Mediated Coupling
The mechanistic elegance of this process centers on the electronic properties of the C-13 side chain precursor. The synthesis begins with trans-phenylisoserine, which is esterified and protected with a trifluoroacetyl or tribromoacetyl group. Cyclization yields the oxazoline derivative, a rigid heterocycle that locks the stereochemistry of the side chain. During the coupling phase with the baccatin core, the oxazoline nitrogen acts as a nucleophile or is activated for esterification, depending on the specific activation strategy employed (e.g., using DCC/DMAP). The critical step occurs post-coupling, where the introduction of a diluted acid (such as dilute hydrochloric acid or trifluoroacetic acid) in an alcohol solvent triggers the ring opening. The electron-withdrawing nature of the trihaloacetyl group weakens the C-O bond within the oxazoline ring, facilitating hydrolysis or alcoholysis at mild temperatures (0°C to room temperature). This selective cleavage regenerates the free hydroxyl group on the side chain while maintaining the integrity of the ester linkage to the baccatin core.
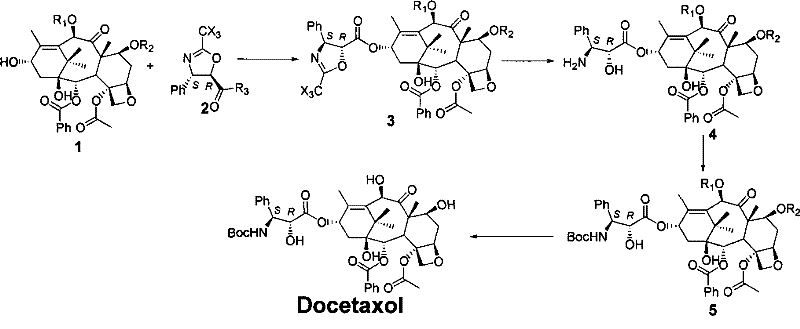
Impurity control is inherently built into this mechanism through the avoidance of basic conditions. In traditional routes, the basic medium can deprotonate the side chain amine, leading to intramolecular attack on the ester carbonyl and subsequent acyl migration (O-to-N acyl shift). By maintaining an acidic or neutral pH during the ring-opening phase, the amine remains protonated or protected, rendering it non-nucleophilic and immune to acyl migration. Furthermore, the use of specific protecting groups on the baccatin core, such as Troc (2,2,2-trichloroethoxycarbonyl) or silyl ethers like TES and TBS, ensures orthogonality. These groups remain stable during the acidic ring opening but can be cleanly removed in the final steps using zinc dust or fluoride sources, respectively. This orthogonal protection strategy ensures that the final deprotection yields Docetaxel with a superior impurity profile, meeting the stringent purity specifications required for oncology injectables.
How to Synthesize Docetaxel Efficiently
The practical execution of this semi-synthetic route involves a logical sequence of four main transformations that convert readily available starting materials into the final active pharmaceutical ingredient. The process initiates with the preparation of the chiral oxazoline side chain from trans-phenylisoserine hydrochloride, involving esterification, acylation, and cyclization. This side chain is then condensed with protected 10-DAB using standard coupling agents. The resulting intermediate undergoes acidic ring opening to reveal the necessary functionality, followed by Boc protection of the amine. Finally, global deprotection of the baccatin core releases the active drug. This streamlined approach minimizes unit operations and maximizes throughput. For detailed operational parameters, stoichiometry, and workup procedures, please refer to the standardized synthesis guide below.
- Condensation Reaction: React protected 10-deacetylbaccatin III (10-DAB) with a synthesized C-13 oxazoline side chain hydrolysate in an organic solvent to form the coupled intermediate.
- Oxazoline Ring Opening: Treat the coupled intermediate with a diluted acid in an alcoholic solution to selectively open the oxazoline ring without affecting other sensitive functional groups.
- Protection and Deprotection: Install the tert-butyloxycarbonyl (Boc) group on the side chain nitrogen, followed by the removal of 7-OH and 10-OH protecting groups to yield final Docetaxel.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this oxazoline-mediated semi-synthesis offers compelling strategic advantages beyond mere technical feasibility. The primary value driver is the substantial simplification of the reaction environment. By eliminating the need for cryogenic temperatures and pyrophoric bases like butyllithium, the process significantly reduces energy consumption and safety infrastructure costs. This translates directly into a lower cost of goods sold (COGS) and a more favorable pricing structure for long-term supply agreements. Additionally, the robustness of the acidic ring-opening step enhances batch-to-batch consistency, reducing the risk of costly campaign failures or reprocessing. The ability to use a variety of protecting groups (Troc, TES, SET) provides supply chain flexibility, allowing manufacturers to source the most economically viable 10-DAB derivatives available on the market without being locked into a single proprietary protection scheme.
- Cost Reduction in Manufacturing: The elimination of extreme cryogenic conditions and hazardous reagents drastically lowers utility and waste disposal costs. The avoidance of acyl migration impurities reduces the burden on downstream purification, leading to higher overall yields and less solvent consumption. This qualitative efficiency gain ensures a more competitive market price for the final API.
- Enhanced Supply Chain Reliability: The starting materials, particularly trans-phenylisoserine and protected 10-DAB, are commercially available from multiple global sources. The chemical stability of the oxazoline intermediates allows for easier storage and transport compared to sensitive beta-lactams, reducing lead time for high-purity intermediates and mitigating the risk of raw material degradation during logistics.
- Scalability and Environmental Compliance: The use of common organic solvents (dichloromethane, ethyl acetate, toluene) and mild acids aligns well with existing industrial infrastructure. The process avoids the generation of heavy metal waste associated with certain catalysts and reduces the environmental footprint of the synthesis. This facilitates smoother regulatory approval and supports sustainable manufacturing goals essential for modern commercial scale-up of complex taxanes.
Frequently Asked Questions (FAQ)
The following technical inquiries address common concerns regarding the implementation of this semi-synthetic pathway. These answers are derived directly from the experimental data and mechanistic principles described in the patent literature, providing clarity on reaction conditions and scalability. Understanding these nuances is critical for R&D teams evaluating technology transfer and for quality assurance teams establishing control strategies.
Q: How does the oxazoline route prevent acyl migration compared to traditional methods?
A: Traditional methods often utilize alkaline conditions which can cause the acyl group to migrate from the oxygen to the nitrogen atom, creating difficult-to-remove impurities. This novel method utilizes trifluoroacetyl or tribromoacetyl groups that are labile under acidic or near-neutral conditions, allowing for clean ring opening and preventing migration.
Q: What protecting groups are compatible with this semi-synthetic pathway?
A: The process is highly versatile regarding the baccatin core protection. Compatible groups include TBS (tert-butyldimethylsilyl), TES (triethylsilyl), EE (ethoxyethyl), THP (tetrahydropyranyl), Troc (2,2,2-trichloroethoxycarbonyl), and MOM (methoxymethyl), offering flexibility for existing supply chains.
Q: Is this method suitable for commercial scale-up of complex taxanes?
A: Yes, the method avoids the extremely low temperatures and highly basic reagents (like butyllithium) required by beta-lactam routes. The use of diluted acids and standard organic solvents facilitates safer and more robust commercial scale-up from kilogram to multi-ton production.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Docetaxel Supplier
At NINGBO INNO PHARMCHEM, we recognize that the transition from laboratory innovation to commercial reality requires a partner with deep technical expertise and robust manufacturing capabilities. Our team specializes in the process development of complex oncology intermediates, including the oxazoline-mediated semi-synthesis of Docetaxel. We possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your supply needs are met with unwavering consistency. Our facilities are equipped with state-of-the-art reactors capable of handling the specific temperature and pressure profiles required for this chemistry, and our rigorous QC labs enforce stringent purity specifications to guarantee the quality of every batch delivered.
We invite you to collaborate with us to optimize your supply chain for this critical anticancer agent. Whether you require custom synthesis of the oxazoline side chain or full-scale production of the final API, our technical procurement team is ready to assist. Please contact us to request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We are prepared to provide specific COA data and comprehensive route feasibility assessments to demonstrate how our manufacturing prowess can enhance your project's economic viability.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
