Advanced Catalytic Dehydrogenation Strategy for Industrial Carbazole Manufacturing
The global demand for high-purity carbazole compounds has surged due to their critical applications in the synthesis of advanced dyes, pigments, and pharmaceutical intermediates. Traditionally sourced from coal tar, the supply chain has been plagued by inconsistent purity levels and energy-intensive separation processes that fail to meet modern industrial standards. A significant technological breakthrough is detailed in patent CN102633710A, which introduces a novel method for synthesizing carbazole compounds via catalytic dehydrogenation. This innovation shifts the paradigm from extraction to efficient synthesis, utilizing 1,2,3,4-tetrahydrocarbazole derivatives as raw materials. By employing a unique combinatorial catalysis strategy, the process achieves exceptional reaction conversion rates and selectivity, ensuring product purity exceeds 99 percent. For R&D directors and procurement specialists seeking a reliable carbazole supplier, this technology represents a pivotal advancement in fine chemical manufacturing, offering a scalable solution that balances cost efficiency with rigorous quality control.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industrial production of carbazole has relied heavily on separation from coal tar, a process characterized by complex purification steps, high energy consumption, and limited output capacity that cannot satisfy growing market needs. Alternative synthetic routes, such as the Fisher indole synthesis, often involve harsh reaction conditions and the use of stoichiometric amounts of oxidants or expensive catalysts that drive up manufacturing costs. Previous literature describes methods using Raney nickel at temperatures up to 330°C or palladium on alumina, yet these often suffer from moderate yields ranging between 77 percent and 88 percent, alongside challenges in catalyst recovery. Furthermore, single-catalyst systems frequently struggle to balance activity with cost, leading to either incomplete conversion or the excessive use of precious metals. These inefficiencies create significant bottlenecks in the supply chain for high-purity carbazole, necessitating a more robust and economically viable synthetic pathway for commercial scale-up of complex pharmaceutical intermediates.
The Novel Approach
The patented methodology overcomes these historical constraints by introducing a sophisticated two-step combinatorial catalysis system that optimizes both cost and performance. Instead of relying on a single catalyst throughout the entire reaction, this approach strategically sequences a cheap, low-activity catalyst with a high-activity noble metal catalyst, or vice versa. This synergy allows for the initial bulk dehydrogenation to occur efficiently with inexpensive materials, followed by a finishing step with a highly active catalyst to ensure complete conversion and high selectivity. The result is a streamlined process that operates at moderate temperatures between 160°C and 250°C, significantly reducing energy requirements compared to traditional high-temperature pyrolysis. By minimizing the loading of expensive noble metals while maximizing their utility, this method delivers a cost reduction in fine chemical manufacturing that is both immediate and substantial. The versatility of this approach is visually represented in the core reaction scheme below, highlighting the clean transformation from tetrahydrocarbazole to the final aromatic product.
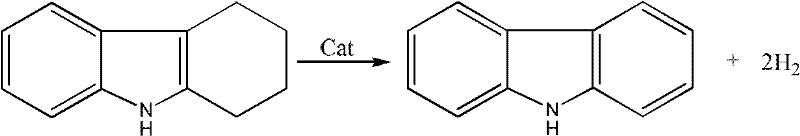
Mechanistic Insights into Combinatorial Catalytic Dehydrogenation
The core of this technological advancement lies in the precise orchestration of catalytic activity during the dehydrogenation of the tetrahydrocarbazole ring system. The reaction mechanism involves the removal of four hydrogen atoms to aromatize the saturated ring, a process that is kinetically challenging without effective catalysis. In the first step, a catalyst such as activated carbon, zeolite, or Raney nickel initiates the dehydrogenation, breaking the initial C-H bonds under reflux conditions in solvents like N,N-dimethylformamide or xylene. Following the removal of the first catalyst via simple filtration, the reaction mixture is treated with a high-activity noble metal catalyst, such as palladium, platinum, or ruthenium supported on carbon or mesoporous materials. This second stage drives the equilibrium towards the fully aromatic carbazole structure, ensuring that intermediate partially dehydrogenated species are fully converted. The ability to switch catalysts mid-process prevents catalyst poisoning and allows each material to operate within its optimal efficiency window, thereby enhancing the overall transformation efficiency.
Impurity control is another critical aspect where this mechanistic design excels, particularly for applications requiring high-purity OLED material or pharmaceutical grade intermediates. The high selectivity of the noble metal catalyst in the second step minimizes side reactions such as ring opening or polymerization, which are common pitfalls in high-temperature dehydrogenation. The substrate scope is broad, accommodating various substituents on the nitrogen atom, including hydrogen, methyl, and ethyl groups, as illustrated in the structural formula below. This flexibility ensures that the process is not limited to unsubstituted carbazole but can be adapted for N-alkylated derivatives widely used in specialty chemical applications. The rigorous control over reaction parameters, including catalyst loading ratios between 1:0.001 and 1:0.1 relative to the substrate, guarantees a consistent impurity profile. Such precision is essential for reducing lead time for high-purity carbazole derivatives, as it eliminates the need for extensive recrystallization or chromatographic purification post-synthesis.
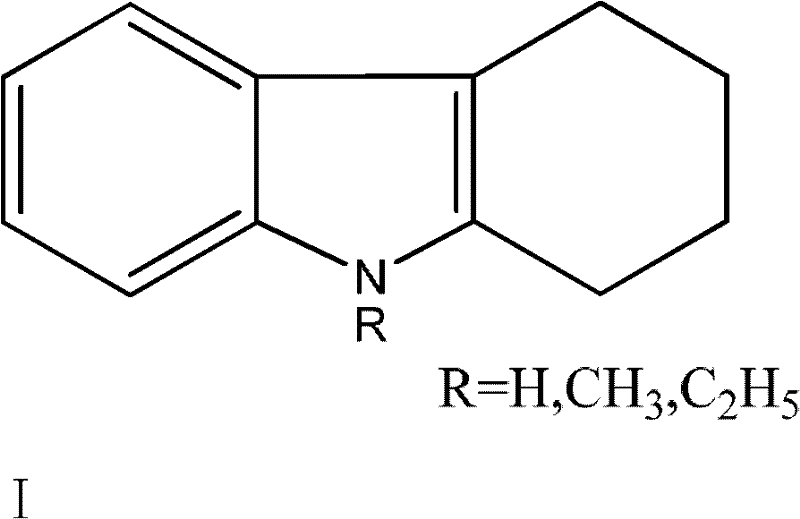
How to Synthesize Carbazole Efficiently
Implementing this synthesis route requires careful attention to the sequencing of catalyst addition and temperature control to maximize yield and purity. The process begins by dissolving the 1,2,3,4-tetrahydrocarbazole compound in a suitable high-boiling organic solvent, followed by the addition of the first catalyst and heating to reflux. After the initial reaction period, the solid catalyst is filtered off while hot to prevent product precipitation, and the filtrate is cooled before introducing the second catalyst for the completion of the dehydrogenation. This sequential addition is the key differentiator that allows for the economic use of noble metals while maintaining high reaction kinetics. The detailed standardized synthesis steps, including specific solvent choices and exact temperature ramps for different substrate variants, are outlined in the technical guide below for process engineers.
- Dissolve 1,2,3,4-tetrahydrocarbazole compound in an organic solvent such as N,N-dimethylformamide or xylene.
- Add the first step catalyst (either cheap low-activity or high-activity) and heat to 160-250°C under reflux for 1-10 hours.
- Filter the first catalyst, cool the filtrate, add the second step catalyst (the opposite type), and reflux for 3-15 hours to obtain the product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this combinatorial catalytic method offers transformative benefits that extend beyond mere technical feasibility. The primary advantage is the drastic simplification of the cost structure associated with catalyst consumption. By utilizing cheap catalysts for the bulk of the reaction and reserving expensive noble metals only for the critical finishing step, the overall cost of goods sold is significantly reduced without compromising on quality. This strategic allocation of resources means that manufacturers can achieve substantial cost savings on raw materials, making the final carbazole product more competitive in the global market. Furthermore, the ability to recover and reuse both the solvents and the solid catalysts adds another layer of economic efficiency, turning what was once a waste stream into a recoverable asset. This circular approach to resource management aligns perfectly with modern sustainability goals and reduces the environmental footprint of the manufacturing process.
- Cost Reduction in Manufacturing: The innovative use of a dual-catalyst system effectively lowers the dependency on high-cost noble metals like palladium and platinum. By optimizing the catalyst loading and sequence, the process achieves high conversion rates with minimal precious metal usage, directly translating to lower production costs. Additionally, the recovery and recycling of organic solvents such as N-methyl pyrrolidone and xylene further diminish operational expenses, ensuring a leaner and more profitable manufacturing model.
- Enhanced Supply Chain Reliability: Unlike coal tar extraction, which is subject to the volatility of the steel industry and fluctuating feedstock availability, this synthetic route relies on readily available chemical precursors. The robustness of the reaction conditions and the stability of the catalysts ensure consistent production schedules, mitigating the risk of supply disruptions. This reliability is crucial for maintaining continuous operations in downstream industries such as dye manufacturing and pharmaceutical synthesis, where interruptions can be costly.
- Scalability and Environmental Compliance: The process is designed with industrial scalability in mind, utilizing standard reflux and filtration equipment that can be easily scaled from pilot plants to multi-ton production facilities. The high selectivity of the reaction minimizes the generation of hazardous by-products, simplifying waste treatment and ensuring compliance with stringent environmental regulations. The ability to recycle catalysts and solvents also reduces the volume of chemical waste, supporting a greener and more sustainable production lifecycle.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this carbazole synthesis technology. These insights are derived directly from the patent specifications and experimental data, providing a clear understanding of the process capabilities and limitations. Understanding these details is vital for technical teams evaluating the feasibility of integrating this method into their existing production lines.
Q: What is the advantage of the combinatorial catalyst system?
A: The method uses a combination of cheap catalysts (like activated carbon or Raney nickel) and high-activity noble metal catalysts (Pd, Pt, Ru). This reduces the overall consumption of expensive noble metals while maintaining high conversion rates and selectivity.
Q: What purity levels can be achieved with this synthesis method?
A: The patented process consistently achieves carbazole content higher than 99 percent, significantly reducing the need for complex downstream purification steps compared to traditional coal tar extraction.
Q: Can the solvents and catalysts be recycled?
A: Yes, the process allows for the recovery and reuse of both the organic solvents and the solid catalysts after filtration, contributing to substantial cost reduction and environmental compliance.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Carbazole Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-quality intermediates play in the success of our clients' final products. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that we can meet your volume requirements with consistency and precision. We are committed to delivering carbazole compounds that meet stringent purity specifications, backed by our rigorous QC labs that employ advanced analytical techniques to verify every batch. Our expertise in catalytic dehydrogenation and fine chemical synthesis allows us to optimize processes for maximum efficiency, guaranteeing a supply of high-purity carbazole that supports your R&D and manufacturing goals.
We invite you to collaborate with us to explore how this advanced synthesis method can benefit your specific application. Please contact our technical procurement team to request a Customized Cost-Saving Analysis tailored to your production needs. We are ready to provide specific COA data and comprehensive route feasibility assessments to demonstrate how our capabilities can enhance your supply chain resilience and drive down your overall manufacturing costs.
