Advanced Synthesis of Dicyclohexyl-18-Crown-6 via Nano-Ruthenium Catalysis for Industrial Scale-Up
The chemical industry continuously seeks methodologies that enhance both the efficiency and selectivity of complex molecular transformations, particularly for high-value macrocyclic compounds like crown ethers. Patent CN102040584B introduces a groundbreaking synthesis method for Dicyclohexyl-18-Crown-6 (DCH18C6), a critical ligand widely utilized in nuclear fuel reprocessing and phase-transfer catalysis. This innovation specifically addresses the longstanding challenge of isomeric control during the catalytic hydrogenation of Dibenzo-18-Crown-6. By employing a specially prepared nano-ruthenium metal powder catalyst, the process achieves a cis-isomer content exceeding 80% directly from the reaction mixture. This represents a significant technological leap over traditional methods, offering a robust pathway for producing high-purity intermediates required for separating strontium ions from high-level radioactive waste streams.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the production of Dicyclohexyl-18-Crown-6 has been plagued by poor stereoselectivity during the hydrogenation of the aromatic precursor. Conventional techniques, such as those utilizing acidic Pichler ruthenium catalysts described in earlier literature like CN1709888A, typically yield a crude product where the desired cis-syn-cis isomer constitutes only between one-third and two-thirds of the mixture. This low initial selectivity necessitates rigorous and costly downstream purification steps, including repeated column chromatography and multiple recrystallizations from solvents like n-heptane. These extensive purification protocols not only result in substantial material loss, drastically reducing the overall process yield, but also generate significant volumes of chemical waste. Furthermore, the inability to control the isomeric ratio at the source complicates the supply chain for applications demanding strict specification limits, such as nuclear waste treatment where the trans-isomer is functionally inert.
The Novel Approach
The methodology disclosed in patent CN102040584B fundamentally alters the reaction landscape by introducing a nano-structured ruthenium catalyst prepared through a unique activation sequence. This novel approach leverages the specific surface properties of nano-metal powders to direct the stereochemical outcome of the hydrogenation. By operating under controlled conditions of 100~200°C and hydrogen pressures ranging from 2~10 MPa, the reaction selectively favors the formation of the cis-isomer. The most striking advantage is that the crude product already contains over 80% of the target cis-isomer without requiring any intermediate purification. This drastic improvement in crude purity simplifies the entire manufacturing workflow, allowing producers to bypass energy-intensive recrystallization cycles. Consequently, this leads to a more sustainable and economically viable production model for high-performance crown ethers.
Mechanistic Insights into Nano-Ruthenium Catalyzed Hydrogenation
The core of this technological advancement lies in the meticulous preparation of the nano-ruthenium catalyst, which dictates the stereoselectivity of the hydrogenation reaction. The catalyst is synthesized by fusing coarse ruthenium powder with potassium hydroxide and potassium nitrate, followed by precipitation and acidification. Crucially, the final step involves refluxing the dried metal powder in small molecule alcohols (C2~C4). Research indicates that this reflux treatment significantly reduces the particle size distribution and lowers the surface oxygen content of the catalyst. These physical modifications create a highly active surface environment that promotes the syn-addition of hydrogen across the aromatic rings in a specific orientation. This controlled addition is what drives the preferential formation of the cis-syn-cis configuration over the thermodynamically stable but functionally less useful trans-isomer, ensuring high fidelity in the molecular architecture of the final product.
From an impurity control perspective, the enhanced selectivity of the nano-ruthenium system minimizes the generation of the trans-anti-cis isomer, which is the primary impurity of concern. In traditional processes, the presence of high levels of trans-isomers requires aggressive separation techniques that can degrade the product or introduce new contaminants. By suppressing the formation of this unwanted isomer at the catalytic stage, the new method inherently produces a cleaner reaction profile. This reduces the burden on analytical quality control laboratories and ensures that the final Dicyclohexyl-18-Crown-6 meets stringent purity specifications required for sensitive applications like ion exchange in acidic media. The robustness of the catalyst also allows for potential recovery and reuse, further stabilizing the reaction kinetics over multiple batches.
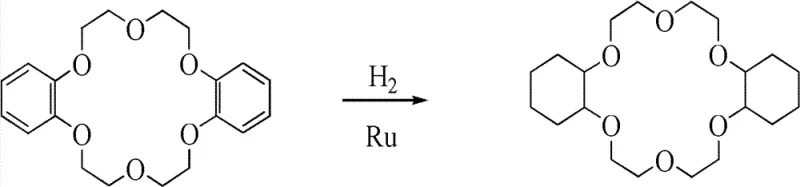
How to Synthesize Dicyclohexyl-18-Crown-6 Efficiently
The synthesis protocol outlined in the patent provides a clear roadmap for transitioning from laboratory scale to commercial production. The process begins with the dissolution of Dibenzo-18-Crown-6 in a suitable organic solvent such as n-butanol, n-octanol, or toluene, followed by the introduction of the activated nano-ruthenium catalyst. The mixture is then subjected to hydrogenation in a high-pressure reactor, where temperature and pressure are tightly regulated to maintain optimal catalytic activity. This standardized approach ensures consistent batch-to-batch reproducibility, a critical factor for industrial clients relying on a steady supply of qualified materials. For detailed operational parameters and safety guidelines, please refer to the specific technical documentation provided below.
- Prepare nano-ruthenium catalyst by melting ruthenium powder with KOH and KNO3, followed by precipitation and acidification.
- Reflux the dried catalyst powder in C2-C4 alcohols to reduce particle size and surface oxygen content.
- Hydrogenate dibenzo-18-crown-6 in organic solvents at 100-200°C and 2-10MPa pressure using the treated catalyst.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this nano-ruthenium catalytic process offers tangible strategic benefits beyond mere technical superiority. The primary value driver is the significant reduction in processing complexity, which directly translates to lower operational expenditures. By eliminating the need for multiple recrystallization steps and extensive chromatographic purification, manufacturers can drastically reduce solvent consumption and waste disposal costs. This streamlined workflow also shortens the overall production cycle time, enhancing the responsiveness of the supply chain to market demands. Furthermore, the ability to achieve high purity in the crude stage mitigates the risk of yield loss associated with aggressive purification, ensuring a more predictable and stable output volume for long-term contracts.
- Cost Reduction in Manufacturing: The elimination of expensive and time-consuming purification stages, such as repeated recrystallization and column chromatography, leads to substantial cost savings in both labor and materials. The high selectivity of the catalyst means that less raw material is wasted in the form of unusable isomers, effectively increasing the mass efficiency of the process. Additionally, the potential for catalyst recovery and reuse further decreases the cost per kilogram of the final product, providing a competitive pricing structure for bulk purchasers without compromising on quality standards.
- Enhanced Supply Chain Reliability: A simplified manufacturing process with fewer unit operations inherently reduces the number of potential failure points in the production line. This robustness ensures greater consistency in delivery schedules and reduces the likelihood of supply disruptions caused by purification bottlenecks. The use of readily available starting materials and standard high-pressure hydrogenation equipment means that production can be easily scaled or shifted between facilities if necessary, providing buyers with a secure and resilient source of critical crown ether intermediates for their operations.
- Scalability and Environmental Compliance: The process is designed with scalability in mind, utilizing reaction conditions that are compatible with standard industrial reactors. The reduction in solvent usage and chemical waste generation aligns with increasingly strict environmental regulations and corporate sustainability goals. By minimizing the environmental footprint of the synthesis, suppliers can offer a greener product profile, which is becoming a key differentiator in global tenders. This eco-friendly approach not only ensures regulatory compliance but also enhances the brand reputation of downstream users who prioritize sustainable sourcing in their supply chains.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production and application of Dicyclohexyl-18-Crown-6 using this advanced catalytic method. These insights are derived directly from the experimental data and technical specifications outlined in the patent literature, providing a reliable basis for decision-making. Understanding these details helps stakeholders evaluate the feasibility of integrating this material into their specific chemical processes or product formulations.
Q: Why is the cis-isomer content critical for DCH18C6 applications?
A: The cis-syn-cis isomer exhibits significantly higher selectivity for extracting Strontium-90 from acidic nuclear waste solutions compared to the trans isomer, making high isomeric purity essential for efficient radioactive separation processes.
Q: How does the nano-ruthenium catalyst improve the synthesis process?
A: The specialized preparation involving alcohol reflux creates smaller particle sizes and optimized surface states, which dramatically increases the stereoselectivity of the hydrogenation reaction, yielding over 80% cis-isomer directly without extensive purification.
Q: Can this synthesis method be scaled for commercial production?
A: Yes, the process utilizes standard high-pressure hydrogenation equipment and recoverable heterogeneous catalysts, making it highly suitable for large-scale manufacturing with reduced solvent consumption and simplified downstream processing.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Dicyclohexyl-18-Crown-6 Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-purity crown ethers play in advanced separation technologies and catalytic applications. As a leading CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your supply needs are met with precision and reliability. Our state-of-the-art facilities are equipped with rigorous QC labs capable of verifying stringent purity specifications, including precise isomeric ratios essential for nuclear and pharmaceutical applications. We are committed to delivering Dicyclohexyl-18-Crown-6 that adheres to the highest international quality standards, supporting your R&D and manufacturing goals with unwavering consistency.
We invite you to collaborate with our technical team to explore how this optimized synthesis route can benefit your specific project requirements. Contact us today to request a Customized Cost-Saving Analysis tailored to your volume needs. Our experts are ready to provide specific COA data and comprehensive route feasibility assessments to help you make informed procurement decisions. Reach out to our technical procurement team now to secure a reliable supply of this high-performance intermediate.
