Revolutionizing Ulipristal Acetate Production via Advanced One-Pot Catalytic Technology
Revolutionizing Ulipristal Acetate Production via Advanced One-Pot Catalytic Technology
The pharmaceutical landscape for emergency contraceptives is undergoing a significant transformation driven by process intensification and green chemistry principles. Patent CN110256519B introduces a groundbreaking one-pot methodology for the preparation of Ulipristal Acetate, a critical active pharmaceutical ingredient (API) known for its superior efficacy compared to traditional levonorgestrel-based solutions. This technical disclosure moves away from fragmented, multi-step batch processes towards a continuous, integrated reaction sequence that merges acidolysis and acetylation into a single vessel operation. For R&D directors and process chemists, this represents a pivotal shift in how complex steroidal intermediates are managed, offering a pathway to drastically reduce the operational footprint while maintaining stringent purity standards required for global regulatory compliance. The strategic implementation of this technology allows manufacturers to bypass the tedious isolation of the key intermediate CW-c, thereby minimizing exposure to oxidative environments that typically degrade product quality.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditional synthetic routes for Ulipristal Acetate have long been plagued by inefficiencies inherent in stepwise processing. In standard protocols, the Grignard addition product must undergo hydrolysis using inorganic acids, followed by a rigorous series of extraction, washing, and concentration steps to isolate the intermediate ketone before acetylation can even commence. This discontinuous workflow not only extends the overall cycle time but also introduces multiple points of failure where yield loss and impurity generation occur. The repeated exposure of the sensitive steroidal backbone to aqueous workups and thermal stress during solvent removal frequently leads to the formation of oxidation byproducts and degradation impurities, complicating downstream purification. Furthermore, the substantial volume of wastewater and organic solvents generated during these isolation phases creates a heavy burden on environmental compliance teams, driving up the cost of waste treatment and disposal significantly.
The Novel Approach
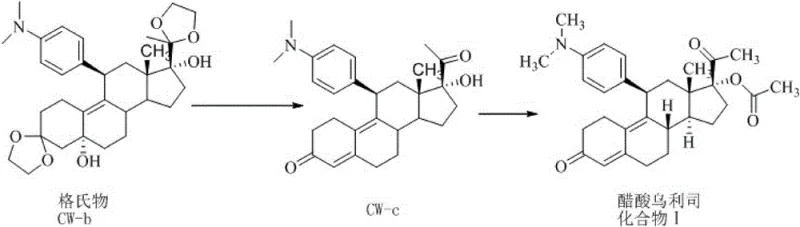
In stark contrast, the novel one-pot strategy detailed in the patent leverages a seamless transition from acidolysis to acetylation without interrupting the reaction stream. By utilizing organic acids for the initial hydrolysis of the Grignard compound CW-b, the process generates the intermediate CW-c in situ, which is immediately subjected to acetylation using acid anhydrides under perchloric acid catalysis. This eliminates the need for intermediate isolation, washing, and drying, effectively collapsing two major synthetic stages into a single operational unit. The result is a streamlined workflow that not only accelerates production throughput but also preserves the chemical integrity of the molecule by maintaining controlled low-temperature conditions throughout. This approach directly addresses the pain points of traditional manufacturing by reducing the 'three wastes' discharge and simplifying the equipment requirements, making it an ideal candidate for modern, sustainable pharmaceutical manufacturing facilities.
Mechanistic Insights into One-Pot Acidolysis and Acetylation
The core of this technological advancement lies in the precise orchestration of reaction conditions that allow for compatible chemistries to coexist in a single pot. The mechanism initiates with the acidolysis of the bis-ketal Grignard adduct (CW-b) using organic acids such as acetic acid or trifluoroacetic acid at temperatures between 0°C and 10°C. This step carefully cleaves the protecting groups to reveal the reactive ketone functionality of intermediate CW-c without inducing premature side reactions. Crucially, the system does not quench or isolate this intermediate; instead, the temperature is modulated to a range of -20°C to 10°C, and an acid anhydride is introduced alongside a perchloric acid catalyst. The perchloric acid acts as a potent promoter for the acetylation of the 17-alpha hydroxyl group, driving the reaction to completion rapidly within 0.5 to 1 hour. This tandem sequence ensures that the reactive intermediate is consumed as soon as it is formed, minimizing its residence time in the reaction mixture and thereby suppressing potential degradation pathways.
From an impurity control perspective, the continuous low-temperature environment is paramount. Traditional methods often require heating during concentration steps to remove solvents between stages, which can trigger thermal decomposition or rearrangement of the steroid nucleus. By avoiding these thermal spikes and keeping the entire process within a cryogenic to ambient window, the formation of degradation impurities is effectively inhibited. The patent data indicates that this meticulous control allows for the achievement of high-purity crude products, often exceeding 99 percent purity after a single recrystallization. This level of selectivity reduces the reliance on extensive chromatographic purification, which is often a bottleneck in API manufacturing. The ability to achieve such high purity directly from crystallization underscores the robustness of the reaction design and its suitability for producing high-quality pharmaceutical intermediates at scale.
How to Synthesize Ulipristal Acetate Efficiently
The execution of this one-pot synthesis requires careful attention to reagent stoichiometry and thermal management to ensure optimal yields and safety. The process begins by establishing a non-polar solvent system, such as dichloromethane or ethyl acetate, into which the organic acid is introduced prior to the addition of the Grignard substrate. Maintaining the temperature at roughly 5°C during the initial acidolysis phase is critical to prevent exothermic runaway while ensuring complete deprotection. Once the intermediate is generated, the system is cooled further to sub-zero temperatures before the introduction of the acetylating agent and catalyst. This precise thermal ramping is essential for controlling the kinetics of the acetylation step. For a comprehensive breakdown of the specific mass-to-volume ratios and timing parameters required for successful replication, please refer to the standardized synthesis guide below.
- Perform controlled acidolysis of Grignard compound CW-b with organic acid at 0-10°C to generate intermediate CW-c.
- Without isolating the intermediate, add acid anhydride and perchloric acid catalyst at -20 to 10°C for direct acetylation.
- Quench the reaction with sodium acetate solution, followed by washing, drying, and crystallization to obtain high-purity Ulipristal Acetate.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain leaders, the adoption of this one-pot technology translates into tangible operational efficiencies and risk mitigation. The elimination of intermediate isolation steps fundamentally alters the cost structure of Ulipristal Acetate manufacturing by removing entire unit operations such as filtration, drying, and re-dissolution. This reduction in processing complexity leads to a significant decrease in utility consumption, including electricity for drying ovens and water for washing procedures. Moreover, the simplified workflow reduces the demand for labor hours per batch, allowing existing personnel to manage higher throughput without proportional increases in headcount. These factors combine to drive down the overall cost of goods sold (COGS), providing a competitive edge in pricing negotiations with downstream API formulators.
- Cost Reduction in Manufacturing: The primary economic benefit stems from the drastic simplification of the process flow. By removing the isolation of intermediate CW-c, manufacturers save substantially on solvent volumes, as there is no need for the large quantities of wash solvents typically required to purify a solid intermediate. Additionally, the avoidance of concentration steps between stages reduces energy costs associated with solvent recovery and distillation. The high yield reported, reaching approximately 90 percent, further enhances material efficiency, ensuring that expensive starting materials like the Grignard compound CW-b are converted into final product with minimal loss. This maximizes the return on raw material investment and stabilizes production budgets against fluctuating feedstock prices.
- Enhanced Supply Chain Reliability: Shortening the synthetic route inherently reduces the lead time required to produce a batch of Ulipristal Acetate. With fewer steps comes less opportunity for equipment downtime, cleaning validation delays, or quality control hold-ups associated with intermediate testing. The robustness of the one-pot method means that production schedules can be tightened, allowing for more responsive fulfillment of market demand. Furthermore, the use of common, commercially available reagents such as acetic anhydride and perchloric acid ensures that the supply chain is not vulnerable to shortages of exotic catalysts. This reliability is crucial for maintaining continuous supply to global markets, especially for essential medications like emergency contraceptives.
- Scalability and Environmental Compliance: The reduction in 'three wastes' discharge is a major advantage for facilities operating under strict environmental regulations. By generating less wastewater and organic waste, the facility lowers its expenditure on waste treatment and disposal services. The process is designed to be scalable, with the patent explicitly noting its suitability for industrial production. The ability to run this reaction in standard glass-lined or stainless steel reactors without specialized equipment facilitates easy technology transfer from pilot plant to commercial scale. This scalability ensures that suppliers can ramp up production capacity quickly to meet surges in demand without requiring massive capital investment in new infrastructure.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this one-pot synthesis method. These insights are derived directly from the experimental data and comparative analysis provided in the patent documentation. Understanding these nuances is essential for stakeholders evaluating the feasibility of adopting this technology for their own manufacturing lines or sourcing strategies.
Q: How does the one-pot method improve impurity profiles compared to traditional synthesis?
A: The one-pot method maintains low-temperature conditions throughout the acidolysis and acetylation steps, which effectively inhibits the formation of degradation impurities often seen during the isolation and workup of intermediates in conventional routes.
Q: What are the primary cost drivers eliminated in this new process?
A: By eliminating the isolation, washing, and concentration steps required for the intermediate CW-c, the process significantly reduces solvent consumption, energy usage for drying, and labor costs associated with multiple unit operations.
Q: Is this synthesis method suitable for large-scale industrial manufacturing?
A: Yes, the patent explicitly states the method is suitable for industrial production due to its simple operation, mild reaction conditions, and high product yield reaching approximately 90 percent, ensuring robust scalability.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Ulipristal Acetate Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of advanced synthetic methodologies like the one-pot acidolysis-acetylation route. As a leading CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that innovative lab-scale discoveries are successfully translated into robust industrial realities. Our commitment to quality is unwavering, supported by stringent purity specifications and rigorous QC labs that utilize state-of-the-art analytical instrumentation to verify every batch. We understand that in the pharmaceutical sector, consistency is key, and our processes are designed to deliver Ulipristal Acetate with the high purity and low impurity profiles demanded by global regulatory bodies.
We invite procurement teams and R&D leaders to engage with us for a Customized Cost-Saving Analysis tailored to your specific volume requirements. By leveraging our optimized one-pot capabilities, we can help you achieve significant reductions in manufacturing costs while securing a stable supply of high-quality intermediates. Please contact our technical procurement team today to request specific COA data and route feasibility assessments. Let us collaborate to streamline your supply chain and bring life-saving medications to market faster and more efficiently.
