Advanced Manufacturing of Losartan Intermediates: A Strategic Technical Analysis for Global Supply Chains
Advanced Manufacturing of Losartan Intermediates: A Strategic Technical Analysis for Global Supply Chains
The pharmaceutical industry continuously seeks robust, scalable, and cost-efficient pathways for the production of high-volume antihypertensive agents like Losartan. Patent CN101328167A introduces a pivotal refinement in the synthetic strategy for Losartan, specifically addressing the critical transformation of protected imidazole precursors into the active pharmaceutical ingredient. This technology leverages a streamlined two-step sequence involving strong acid-catalyzed detritylation followed by selective hydride reduction. For R&D directors and procurement strategists, this approach represents a significant departure from legacy coupling methods, offering a route that minimizes unit operations while maximizing yield and purity. By utilizing widely available reagents such as hydrochloric acid and potassium borohydride, the process mitigates supply chain risks associated with exotic catalysts, positioning it as a highly viable option for reliable pharmaceutical intermediates supplier networks aiming for long-term stability.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of Losartan has been plagued by complex multi-step sequences that introduce significant inefficiencies into the manufacturing value chain. Traditional methodologies, such as those described in US5130439 and EP1777224, often rely on intricate coupling reactions between imidazole derivatives and biphenyl tetrazole components. These legacy routes frequently necessitate the use of hazardous reagents, such as sodium azide for tetrazole ring formation, which imposes severe safety constraints and requires specialized equipment for handling explosive intermediates. Furthermore, the purification of intermediates in these conventional pathways is often arduous, involving multiple chromatographic separations or recrystallizations that erode overall yield and increase waste generation. The reliance on transition metal catalysts in some variations also introduces the risk of heavy metal contamination, necessitating costly downstream purification steps to meet stringent regulatory limits for API production.
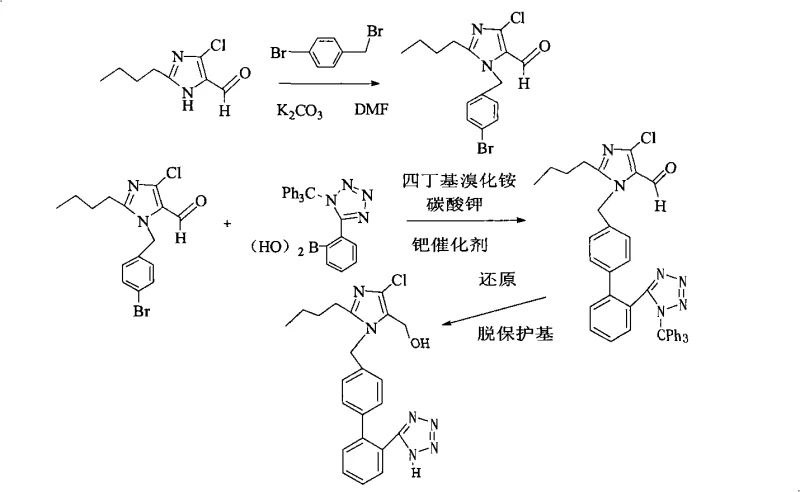
The Novel Approach
In stark contrast to the convoluted nature of prior art, the methodology disclosed in CN101328167A offers a direct and chemically elegant solution by focusing on the late-stage functionalization of a pre-assembled scaffold. Instead of building the molecule through difficult couplings, this novel approach starts with a fully assembled but protected intermediate, 2-butyl-4-chloro-5-formyl-1-[(2'-(1-triphenylmethyl-tetrazole-5-yl)-biphenyl-4-)methyl] imidazole. The innovation lies in the sequential removal of the triphenylmethyl (trityl) protecting group under mild acidic conditions, immediately followed by the reduction of the formyl moiety to the requisite hydroxymethyl group. This telescoped strategy drastically reduces the number of isolation steps, thereby minimizing material loss and solvent consumption. The operational simplicity allows for easier scale-up, making it an attractive candidate for cost reduction in pharmaceutical intermediates manufacturing where margin compression is a constant challenge.
Mechanistic Insights into Acid-Catalyzed Deprotection and Hydride Reduction
The core chemical transformation in this patented process relies on the differential stability of the trityl-tetrazole bond versus the imidazole-biphenyl backbone under acidic conditions. In the first stage, the trityl group acts as a robust protecting group for the tetrazole nitrogen during earlier synthetic steps, preventing unwanted side reactions. However, upon exposure to a strong acid such as hydrochloric acid, sulfuric acid, or phosphoric acid in a polar solvent, the trityl cation is readily generated and cleaved from the tetrazole ring. This deprotection step is highly selective, leaving the sensitive imidazole and biphenyl moieties intact. The choice of solvent plays a critical role here; protic solvents like methanol or polar aprotic solvents like THF stabilize the transition state and facilitate the solubilization of the ionic intermediates, ensuring complete conversion without degrading the core structure.
Following the successful generation of the free tetrazole aldehyde intermediate, the process transitions to a reduction phase utilizing a hydroborate source, typically potassium borohydride or sodium borohydride. This step targets the electrophilic carbonyl carbon of the formyl group at the 5-position of the imidazole ring. The hydride ion attacks the carbonyl, forming an alkoxide intermediate which is subsequently protonated by the solvent to yield the final hydroxymethyl functionality. This reduction is chemoselective, meaning it reduces the aldehyde without affecting the chloro-substituent on the imidazole or the aromatic rings. The ability to perform this reduction in the same or similar solvent systems as the deprotection allows for potential telescoping of the process, further enhancing efficiency and reducing the environmental footprint associated with solvent swaps and intermediate drying.
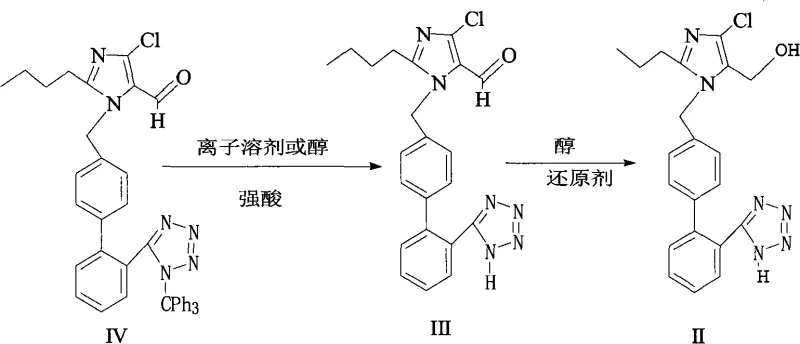
How to Synthesize Losartan Efficiently
The execution of this synthesis requires precise control over reaction parameters to ensure optimal yield and purity. The process begins with the dissolution of the trityl-protected precursor in a suitable solvent system, followed by the controlled addition of aqueous strong acid. Temperature management is crucial during this exothermic deprotection to prevent thermal degradation. Once the deprotection is complete, the reaction mixture is adjusted to a basic pH to isolate the free tetrazole intermediate, or it can be directly subjected to reduction conditions depending on the specific protocol optimization. The subsequent reduction step involves the slow addition of the borohydride reagent to manage gas evolution and heat generation. Detailed standard operating procedures regarding stoichiometry, addition rates, and workup protocols are essential for reproducibility. For a comprehensive guide on the specific molar ratios and temperature profiles validated in the patent examples, please refer to the standardized synthesis instructions below.
- Perform detritylation of the trityl-protected imidazole intermediate using a strong acid (e.g., hydrochloric acid) in an alcoholic or ionizing solvent to yield the free tetrazole aldehyde.
- Subject the resulting aldehyde intermediate to reduction using a hydroborate reagent (such as potassium borohydride) in an alcoholic solvent to generate the final hydroxymethyl product.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, the adoption of this synthetic route offers profound benefits for procurement managers and supply chain directors tasked with optimizing the cost of goods sold (COGS) and ensuring supply continuity. The primary driver of value creation here is the simplification of the chemical bill of materials. By eliminating the need for expensive transition metal catalysts, ligands, or hazardous azide reagents, the direct material costs are significantly lowered. Furthermore, the use of commodity chemicals like hydrochloric acid and methanol ensures that the supply chain is not vulnerable to the volatility often seen with specialized fine chemical reagents. This resilience is critical for maintaining consistent production schedules and meeting the rigorous delivery timelines demanded by global API manufacturers.
- Cost Reduction in Manufacturing: The economic advantage of this process is derived principally from the reduction in unit operations and the avoidance of costly purification technologies. Traditional routes often require chromatography or extensive recrystallization to remove metal catalysts or azide byproducts, which are capital-intensive and time-consuming. In contrast, the acid-base workup described in this patent allows for simple phase separations and crystallizations. The elimination of heavy metal scavengers and the associated validation testing further reduces operational expenditures. Additionally, the high selectivity of the reduction step minimizes the formation of difficult-to-separate impurities, leading to higher overall yields and less waste disposal cost, contributing to substantial cost savings in large-scale production environments.
- Enhanced Supply Chain Reliability: Supply chain robustness is significantly enhanced by the reliance on globally available, bulk-grade reagents. Hydrochloric acid, sulfuric acid, and potassium borohydride are produced in massive quantities worldwide, ensuring that production is never halted due to the shortage of a niche starting material. This contrasts sharply with routes dependent on custom-synthesized building blocks or single-source catalysts. The simplified process flow also reduces the lead time for batch completion, allowing manufacturing facilities to respond more agilely to fluctuations in market demand. This flexibility is a key strategic asset for any reliable pharmaceutical intermediates supplier looking to secure long-term contracts with major generic drug developers.
- Scalability and Environmental Compliance: The environmental profile of this synthesis aligns well with modern green chemistry principles and regulatory expectations. By avoiding azide chemistry, the process eliminates the generation of toxic hydrazoic acid and reduces the safety burden on the manufacturing plant. The solvent systems employed, primarily alcohols and THF, are well-understood and can be efficiently recovered and recycled using standard distillation infrastructure. The high atom economy of the reduction step and the clean deprotection profile result in a lower E-factor (mass of waste per mass of product). This facilitates easier regulatory approval and reduces the environmental compliance costs associated with waste treatment, making the commercial scale-up of complex pharmaceutical intermediates more sustainable and economically viable.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this Losartan synthesis technology. These insights are derived directly from the experimental data and claims presented in the patent literature, providing a factual basis for decision-making. Understanding these nuances is essential for technical teams evaluating the feasibility of technology transfer and for commercial teams assessing the competitive landscape of Losartan production.
Q: What are the primary advantages of the acid-catalyzed deprotection route for Losartan?
A: This route significantly simplifies the synthetic pathway by avoiding complex coupling reactions required in earlier patents. It utilizes common, inexpensive reagents like hydrochloric acid and borohydrides, leading to easier purification and higher overall purity profiles suitable for API manufacturing.
Q: Which solvents are optimal for the detritylation step in this process?
A: The patent specifies that ionizing solvents such as acetone, tetrahydrofuran (THF), dioxane, or DMF, as well as lower alcohols like methanol, are effective. Methanol and THF are particularly preferred for balancing reaction kinetics and ease of downstream processing.
Q: How does this method impact the scalability of Losartan production?
A: By eliminating the need for hazardous azide chemistry or expensive palladium catalysts found in alternative routes, this method enhances operational safety and reduces raw material costs. The straightforward workup procedures, such as pH adjustment and crystallization, facilitate robust scale-up from pilot to commercial tonnage.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Losartan Intermediate Supplier
At NINGBO INNO PHARMCHEM, we recognize that the successful commercialization of antihypertensive therapies depends on the availability of high-quality, cost-effective intermediates. Our technical team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the theoretical benefits of patents like CN101328167A are fully realized in practical manufacturing settings. We operate stringent purity specifications and maintain rigorous QC labs equipped with advanced analytical instrumentation to guarantee that every batch of Losartan intermediate meets the exacting standards required for API synthesis. Our commitment to quality assurance ensures that our clients receive materials that facilitate smooth downstream processing and regulatory filing success.
We invite global partners to collaborate with us to leverage this advanced synthetic technology for their supply chains. By engaging with our technical procurement team, you can request a Customized Cost-Saving Analysis tailored to your specific volume requirements and logistical constraints. We encourage potential partners to contact us directly to obtain specific COA data and route feasibility assessments, allowing you to make informed decisions that drive value and efficiency in your pharmaceutical manufacturing operations. Let us be your strategic partner in delivering high-purity pharmaceutical intermediates to the global market.
