Scaling Green Photochemistry: Industrial Production of 2-Methyl-1,4-naphthoquinone via LED Catalysis
Scaling Green Photochemistry: Industrial Production of 2-Methyl-1,4-naphthoquinone via LED Catalysis
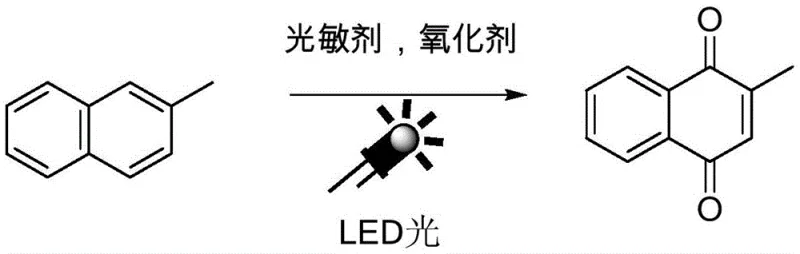
The pharmaceutical and agrochemical industries are currently undergoing a paradigm shift towards sustainable manufacturing processes, driven by stringent environmental regulations and the economic necessity of reducing waste. A pivotal development in this arena is detailed in patent CN112358389A, which discloses a novel preparation method for the photochemical synthesis of 2-methyl-1,4-naphthoquinone (2-MNQ). This compound serves as a critical intermediate for Vitamin K3 (menadione), a vital nutrient supplement in poultry farming and a key building block in various fine chemical applications. The patented technology leverages visible light LED irradiation combined with organic photosensitizers to drive the oxidation of 2-methylnaphthalene, effectively replacing traditional, hazardous heavy metal oxidants. For R&D directors and procurement managers seeking a reliable API intermediate supplier, this methodology represents a significant leap forward in process safety and environmental compliance.
The core innovation lies in the utilization of clean energy sources—specifically LED light—to activate molecular oxygen, thereby generating reactive oxygen species that selectively oxidize the substrate. This approach not only mitigates the generation of toxic chromium waste but also offers superior control over reaction selectivity. By operating under mild conditions, typically between 20°C and 30°C, the process minimizes thermal degradation and energy consumption. The implications for cost reduction in pharmaceutical intermediates manufacturing are profound, as the elimination of heavy metal catalysts removes the need for expensive downstream purification steps such as ion exchange or specialized filtration, streamlining the entire production workflow from raw material to final crystalline product.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industrial synthesis of 2-methyl-1,4-naphthoquinone has been dominated by direct oxidation methods utilizing strong inorganic oxidants. The most prevalent technique involves the use of chromium trioxide (CrO3) in the presence of sulfuric acid (H2SO4). While this method is chemically effective, it suffers from severe ecological and operational drawbacks. The process generates substantial quantities of toxic chromium salts, posing significant disposal challenges and environmental hazards that conflict with modern green chemistry principles. Furthermore, the harsh acidic conditions often lead to over-oxidation and the formation of undesirable byproducts, necessitating complex workup procedures to isolate the target quinone. Alternative metal-catalyzed routes using manganese, palladium, or vanadium have been explored, yet they often struggle with catalyst recovery, high costs of precious metals, and sensitivity to reaction conditions, making them less viable for large-scale commercial scale-up of complex pharmaceutical intermediates.
The Novel Approach
In stark contrast, the photochemical method described in the patent introduces a metal-free catalytic cycle that operates under ambient pressure and near-room temperature. By employing organic dyes such as eosin Y, fluorescein, or acridine salts as photosensitizers, the system harnesses the energy of LED light to excite ground-state triplet oxygen into reactive singlet oxygen. This activated species then reacts with 2-methylnaphthalene to form the desired quinone structure with high efficiency. The use of air or pure oxygen as the sole oxidant means that the only stoichiometric byproduct is water, fundamentally altering the waste profile of the synthesis. This transition from stoichiometric heavy metal oxidants to catalytic photo-oxidation not only enhances the safety profile of the manufacturing plant but also significantly simplifies the regulatory burden associated with heavy metal residues in final drug substances.
Mechanistic Insights into Visible-Light Mediated Photo-Oxidation
To fully appreciate the technical robustness of this synthesis, one must understand the underlying photophysical mechanisms. The process initiates when the organic photosensitizer absorbs photons from the LED source, promoting it from a ground state to an excited singlet state, which rapidly undergoes intersystem crossing to a longer-lived triplet state. This triplet excited sensitizer then transfers energy to ground-state molecular oxygen (³O₂), generating highly reactive singlet oxygen (¹O₂). This singlet oxygen acts as a dienophile in a [4+2] cycloaddition with the electron-rich naphthalene ring of the substrate, forming a transient endoperoxide intermediate. This unstable intermediate subsequently undergoes rearrangement and bond cleavage, facilitated by the reaction medium, to yield the 1,4-naphthoquinone skeleton. The elegance of this mechanism lies in its reliance on renewable light energy and abundant atmospheric oxygen, bypassing the need for harsh chemical reagents.
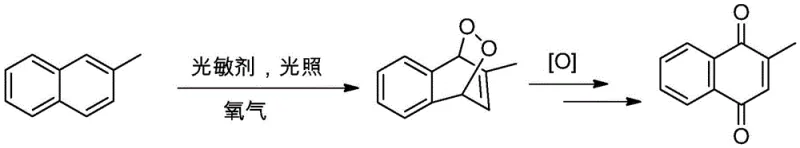
From an impurity control perspective, this mechanism offers distinct advantages over radical-based auto-oxidation or metal-catalyzed pathways. The selectivity of singlet oxygen towards the specific diene system of the naphthalene ring minimizes non-specific oxidation at the methyl group or other positions on the aromatic ring, which are common side reactions in thermal oxidations. Experimental data from the patent indicates that this method can achieve yields as high as 87% with purities exceeding 95%, significantly outperforming the 40-60% yields typical of older chromium-based methods. The absence of metal ions also precludes the formation of metal-complexed impurities, ensuring a cleaner crude product that requires fewer recrystallization steps to meet the stringent specifications required for high-purity API intermediates.
How to Synthesize 2-Methyl-1,4-naphthoquinone Efficiently
Implementing this photochemical protocol in a laboratory or pilot plant setting requires careful attention to light source selection and reactor geometry to ensure uniform photon flux. The standard procedure involves dissolving the substrate and photosensitizer in a polar protic solvent like ethanol, followed by irradiation with blue LEDs (450-455 nm) while maintaining a steady flow of air. The reaction progress is monitored until the starting material is consumed, typically within 20 to 28 hours. Following the reaction, the workup is remarkably straightforward, involving concentration and aqueous quenching to precipitate the product. For detailed operational parameters and specific molar ratios optimized for different scales, please refer to the standardized synthesis guide below.
- Prepare the reaction mixture by dissolving 2-methylnaphthalene and an organic photosensitizer (such as eosin Y or acridine salt) in a suitable solvent like ethanol.
- Irradiate the solution with specific wavelength LED light (preferably 450-455 nm) while bubbling air or oxygen through the mixture at 20-30°C.
- Upon completion, concentrate the reaction mixture, induce crystallization with water, and recrystallize the solid product to achieve high purity.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this photochemical technology translates into tangible strategic benefits beyond mere technical novelty. The primary advantage is the drastic simplification of the supply chain regarding raw materials. By eliminating the dependence on volatile and regulated heavy metal oxidants like chromium trioxide, manufacturers can avoid the logistical complexities and price fluctuations associated with hazardous chemical procurement. Furthermore, the use of air as an oxidant represents a virtually infinite and cost-free resource, decoupling production costs from the market prices of specialized oxidizing agents. This stability in raw material sourcing is crucial for maintaining consistent pricing and supply continuity for long-term contracts in the vitamin and feed additive sectors.
- Cost Reduction in Manufacturing: The economic impact of removing heavy metal catalysts extends far beyond the purchase price of the reagents. In traditional processes, a significant portion of the manufacturing budget is allocated to downstream processing, specifically the removal of trace metals to meet pharmacopeial limits. This often involves expensive scavengers, multiple filtration stages, and extensive wastewater treatment to handle toxic sludge. By adopting a metal-free photochemical route, these downstream costs are effectively eliminated. The simplified workup, which relies on basic crystallization rather than complex chromatographic or extraction techniques, reduces both labor hours and solvent consumption, leading to substantial overall cost savings per kilogram of produced 2-MNQ.
- Enhanced Supply Chain Reliability: The robustness of the reaction conditions contributes directly to supply chain resilience. Traditional oxidation methods often require strict anhydrous conditions or inert atmospheres to prevent side reactions or catalyst deactivation. In contrast, this photochemical method is tolerant to moisture and operates effectively in the presence of air, reducing the risk of batch failures due to minor environmental deviations. This operational forgiveness ensures higher batch success rates and more predictable production schedules, allowing suppliers to meet tight delivery windows with greater confidence and reducing the lead time for high-purity pharmaceutical intermediates.
- Scalability and Environmental Compliance: As regulatory bodies worldwide tighten restrictions on industrial emissions, the ability to demonstrate a "green" manufacturing process is a significant competitive advantage. This technology produces water as the only byproduct, drastically reducing the chemical oxygen demand (COD) of the effluent and minimizing the need for costly waste treatment infrastructure. The scalability of LED technology has also improved dramatically, with high-power arrays now available for industrial photoreactors. This allows for the seamless transition from gram-scale optimization to ton-scale production without the fundamental process changes often required when scaling thermal exothermic reactions, ensuring a smoother path to commercialization.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this photochemical synthesis route. These insights are derived directly from the experimental data and comparative analysis provided in the patent documentation, aiming to clarify the feasibility and benefits for potential partners and licensees.
Q: How does this photochemical method improve impurity profiles compared to chromium oxidation?
A: Unlike traditional chromium-based oxidation which generates toxic heavy metal waste and often produces isomeric impurities like 6-MNQ, this LED-driven method utilizes selective singlet oxygen activation. This results in a cleaner reaction profile with significantly reduced isomeric byproducts, achieving purities exceeding 95% without complex heavy metal scavenging steps.
Q: What are the scalability advantages of using air as the oxidant?
A: Using air or oxygen as the terminal oxidant eliminates the need for expensive and hazardous stoichiometric oxidants like chromic acid or permanganates. This drastically simplifies the supply chain logistics, reduces raw material costs, and ensures that the only byproduct is water, aligning perfectly with modern environmental compliance standards for large-scale manufacturing.
Q: Is the process sensitive to moisture or rigorous anhydrous conditions?
A: One of the distinct advantages of this protocol is its robustness against moisture. The reaction proceeds efficiently in common protic solvents like ethanol and does not require strictly anhydrous conditions, which lowers operational complexity and energy consumption associated with solvent drying in an industrial setting.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 2-Methyl-1,4-naphthoquinone Supplier
At NINGBO INNO PHARMCHEM, we recognize that the transition to greener synthesis methods is not just an environmental imperative but a business necessity. Our team of expert process chemists has extensively evaluated the photochemical route described in CN112358389A and possesses the technical capability to adapt and optimize this technology for your specific production needs. We bring extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the efficiencies observed in the laboratory are faithfully reproduced in our manufacturing facilities. Our commitment to quality is underpinned by stringent purity specifications and rigorous QC labs equipped to detect and quantify even trace levels of isomeric impurities, guaranteeing a product that meets the highest global standards.
We invite you to collaborate with us to leverage this advanced technology for your supply chain. Whether you require a custom synthesis campaign or a long-term supply agreement, our technical sales team is ready to provide a Customized Cost-Saving Analysis tailored to your volume requirements. Please contact our technical procurement team today to request specific COA data and discuss route feasibility assessments for integrating this sustainable 2-MNQ production method into your portfolio.
