Advanced Synthesis of Pyrrolidine Derivatives for High-Purity Carbapenem Antibiotic Production
Advanced Synthesis of Pyrrolidine Derivatives for High-Purity Carbapenem Antibiotic Production
The pharmaceutical landscape for carbapenem antibiotics is constantly evolving, driven by the need for more efficient and purer synthetic routes to combat resistant bacterial strains. Patent CN100460389C introduces a groundbreaking methodology for synthesizing a novel pyrrolidine derivative intermediate, specifically designed for the production of S-4661, also known as Doripenem. This innovative approach fundamentally alters the traditional synthetic strategy by eliminating the tert-butoxycarbonyl (Boc) protecting group, which has historically been a source of significant processing challenges. By utilizing a p-nitrobenzyloxycarbonyl (PNZ) protection strategy, the invention circumvents the harsh acidic conditions required for Boc removal, thereby preserving the integrity of the sensitive beta-lactam core. 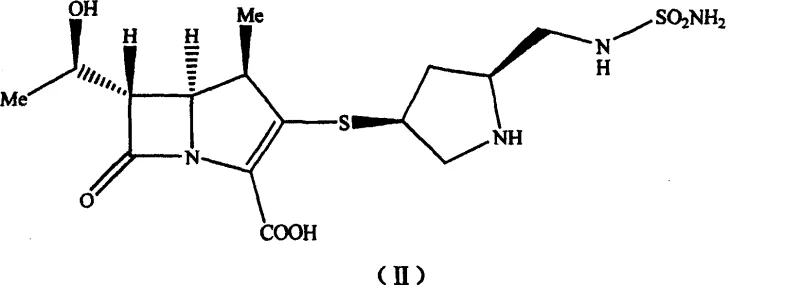 This structural optimization not only enhances the overall yield but also drastically simplifies the downstream purification processes, making it an attractive candidate for reliable pharmaceutical intermediates supplier networks seeking to optimize their manufacturing portfolios.
This structural optimization not only enhances the overall yield but also drastically simplifies the downstream purification processes, making it an attractive candidate for reliable pharmaceutical intermediates supplier networks seeking to optimize their manufacturing portfolios.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industrial synthesis of S-4661 has relied on two primary routes, both of which suffer from substantial drawbacks that hinder cost-effective mass production. The first conventional route utilizes a benzhydryl ester intermediate coupled with a Boc-protected pyrrolidine derivative, requiring Lewis acid aluminum chloride for deprotection. This method is plagued by the difficulty of removing residual aluminum ions, which necessitates the use of expensive divinylbenzene-type macroporous resin columns for purification, creating a bottleneck in suitability for industrialized production. The second route employs sulfuric acid to remove the Boc group, a process that generates reactive carbonium ions leading to numerous side reactions and difficult-to-remove impurities. Furthermore, the intermediate obtained from sulfuric acid deprotection is often an oily substance at room temperature, posing severe challenges for accurate metering and feeding in large-scale reactors. These legacy methods result in lower overall yields, higher waste generation, and increased operational complexity, driving up the cost reduction in pharmaceutical intermediates manufacturing.
The Novel Approach
In stark contrast, the novel approach detailed in the patent utilizes a PNZ-protected pyrrolidine derivative that completely bypasses the need for Boc protection and subsequent acidolysis. This strategic shift allows for the formation of a stable, solid intermediate that can be easily handled, weighed, and fed into reactors without the complications associated with oily substances. The synthesis avoids the use of Lewis acids entirely, thereby eliminating the risk of metal contamination and the need for complex resin-based purification steps. By employing a nucleophilic substitution reaction between the mercaptopyrrolidine derivative and the penem core, the process achieves high selectivity and conversion rates under mild conditions. This streamlined workflow not only reduces the number of unit operations but also significantly lowers the environmental footprint by minimizing solvent usage and waste generation. Consequently, this method represents a paradigm shift towards more sustainable and economically viable commercial scale-up of complex pharmaceutical intermediates.
Mechanistic Insights into PNZ-Protection and Coupling Strategy
The core of this technological advancement lies in the meticulous design of the PNZ-protection strategy and the subsequent coupling mechanisms that ensure high fidelity in stereochemistry. The process begins with the preparation of N-p-nitrobenzyloxycarbonyl (PNZ) sulfonamide, achieved through the addition of chlorosulfonyl isocyanate to p-nitrobenzyl alcohol followed by ammonolysis. This PNZ-sulfonamide serves as a robust nitrogen source that is compatible with the sensitive functional groups present in the pyrrolidine ring. The coupling reaction utilizes a Mitsunobu-type mechanism involving triphenylphosphine and diisopropyl azodicarboxylate (DIAD) to link the sulfonamide to the hydroxymethyl group of the pyrrolidine precursor. This activation strategy ensures inversion of configuration where necessary and maintains the critical (2S, 4S) stereochemistry required for biological activity. The resulting acetylthio pyrrolidin derivative is stable and can be isolated as a solid, providing a crucial checkpoint for quality control before proceeding to the final coupling steps.
Impurity control is inherently built into this mechanistic pathway by avoiding the generation of reactive carbocation species that typically arise during acid-catalyzed deprotection. In conventional Boc-chemistry, the formation of tert-butyl cations can lead to alkylation of the nucleophilic sulfur or nitrogen atoms, creating structurally similar impurities that are notoriously difficult to separate. By utilizing the PNZ group, which is removed via hydrogenolysis under neutral conditions, the process preserves the delicate beta-lactam ring from acid-catalyzed hydrolysis or rearrangement. The final deacetylation step uses mild mineral alkalis in an ice bath, ensuring that the thiol group is liberated without affecting the adjacent stereocenters. This precise control over reaction conditions minimizes the formation of diastereomers and regioisomers, resulting in a final product with exceptional optical purity. Such rigorous control over the reaction mechanism is essential for meeting the stringent regulatory requirements for high-purity API intermediates.
How to Synthesize Pyrrolidine Derivative Efficiently
The synthesis of this key intermediate involves a sequence of highly controlled reactions that transform simple starting materials into a complex, chiral building block suitable for antibiotic assembly. The process is designed to be robust and scalable, utilizing reagents that are readily available in the global chemical supply chain. Operators must pay close attention to temperature control during the exothermic addition of chlorosulfonyl isocyanate and the subsequent coupling steps to ensure safety and reproducibility. The detailed standardized synthesis steps, including specific molar ratios, solvent choices, and workup procedures, are outlined in the structured guide below to facilitate technology transfer and process validation.
- Preparation of N-p-nitrobenzyloxycarbonyl (PNZ) sulfonamide via reaction of p-nitrobenzyl alcohol with chlorosulfonyl isocyanate followed by ammonolysis.
- Coupling of the PNZ-sulfonamide with (2S,4S)-4-acetylthio-1-p-nitrobenzyloxycarbonyl-pyrrolidine-2-methanol using triphenylphosphine and DIAD.
- Deacetylation of the coupled product using mineral alkali to yield the key mercaptopyrrolidine derivative intermediate.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this novel synthetic route offers transformative benefits that extend far beyond simple chemical efficiency. The elimination of expensive and hazardous reagents like aluminum chloride and concentrated sulfuric acid directly translates to a safer working environment and reduced costs associated with hazardous waste disposal. The ability to isolate intermediates as free-flowing solids rather than oils simplifies logistics, storage, and handling, reducing the risk of batch-to-batch variability caused by inaccurate dosing. Furthermore, the removal of complex purification steps such as macroporous resin chromatography shortens the overall cycle time, allowing for faster throughput and improved responsiveness to market demand. These factors collectively contribute to a more resilient and cost-effective supply chain for critical antibiotic ingredients.
- Cost Reduction in Manufacturing: The economic impact of this process is driven primarily by the simplification of the purification train and the reduction in raw material costs. By avoiding the use of Lewis acid catalysts, manufacturers eliminate the need for specialized resin columns and the associated solvents required for their regeneration, leading to substantial operational savings. Additionally, the higher yields achieved through the suppression of side reactions mean that less starting material is wasted, further driving down the cost per kilogram of the final intermediate. The use of standard reagents like triphenylphosphine and DIAD, which are commercially available at scale, ensures that the process remains economically viable without reliance on exotic or proprietary catalysts. This holistic approach to cost optimization makes the technology highly attractive for large-scale commercial production.
- Enhanced Supply Chain Reliability: Supply chain stability is significantly bolstered by the use of robust, non-hygroscopic solid intermediates that possess excellent shelf-life characteristics. Unlike oily intermediates that may degrade or polymerize over time, the crystalline nature of the PNZ-protected derivatives ensures consistent quality during storage and transportation. The process relies on commodity chemicals that are widely sourced, reducing the risk of supply disruptions caused by the scarcity of specialized reagents. Moreover, the simplified workflow reduces the dependency on highly skilled labor for complex chromatographic separations, making it easier to replicate the process across different manufacturing sites. This reliability is crucial for maintaining continuous production schedules and meeting the rigorous delivery commitments expected by global pharmaceutical partners.
- Scalability and Environmental Compliance: From an environmental and scalability perspective, this route aligns perfectly with modern green chemistry principles and regulatory expectations. The avoidance of heavy metal catalysts eliminates the burden of testing for and removing trace aluminum residues, streamlining the regulatory filing process for the final drug product. The reduction in solvent consumption and waste generation lowers the facility's environmental footprint, facilitating compliance with increasingly strict emissions standards. The process is inherently scalable, as the exothermic reactions can be managed effectively in large reactors using standard cooling protocols, ensuring safe expansion from pilot to commercial scale. This alignment with sustainability goals not only mitigates regulatory risk but also enhances the corporate social responsibility profile of the manufacturing organization.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this patented synthesis route for S-4661 intermediates. These answers are derived directly from the experimental data and comparative analysis provided in the patent documentation, offering clarity on the practical benefits of the technology. Understanding these nuances is vital for technical teams evaluating the feasibility of adopting this new methodology for their existing production lines.
Q: Why is the Boc-free intermediate superior for S-4661 production?
A: The Boc-free intermediate avoids the use of strong acids like sulfuric acid for deprotection, which typically generates carbonium ion side products that are difficult to remove. Additionally, it eliminates the need for Lewis acid aluminum chloride, preventing residual aluminum contamination.
Q: How does this process improve industrial scalability?
A: The process yields solid intermediates (pressed powder) rather than oily substances, significantly simplifying metering and feeding operations in large-scale reactors. It also replaces complex column chromatography purification with simpler crystallization and filtration steps.
Q: What are the key purity advantages of this route?
A: By avoiding acidolysis side reactions and eliminating the need for macroporous resin columns used in conventional routes, this method ensures higher chemical purity and reduces the risk of trace metal impurities in the final API.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable S-4661 Intermediate Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of adopting advanced synthetic routes to maintain competitiveness in the global antibiotic market. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory innovation to industrial reality is seamless and efficient. We are committed to delivering products that meet stringent purity specifications, leveraging our rigorous QC labs to verify every batch against the highest international standards. Our expertise in handling complex heterocyclic chemistry allows us to navigate the intricacies of the PNZ-protection strategy with precision, guaranteeing a consistent supply of high-quality intermediates for your drug development needs.
We invite you to collaborate with us to unlock the full potential of this cost-effective and robust manufacturing technology. Contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your specific production volumes and requirements. We are ready to provide specific COA data and comprehensive route feasibility assessments to demonstrate how our capabilities can enhance your supply chain efficiency. Let us be your partner in delivering life-saving medications to the world with unmatched quality and reliability.
