Advanced Purification of Doripenem: Enhancing Stability and Yield for Commercial Scale-up
Advanced Purification of Doripenem: Enhancing Stability and Yield for Commercial Scale-up
The pharmaceutical landscape for broad-spectrum carbapenem antibiotics demands rigorous control over impurity profiles and process stability, particularly for potent agents like Doripenem (S-4661). Patent CN101880282A introduces a transformative purification methodology that addresses the inherent instability of Doripenem in aqueous environments. Unlike traditional crystallization techniques that rely solely on temperature manipulation in pure water, this innovation incorporates L-proline into the crystallization matrix. This strategic addition fundamentally alters the solvation environment, mitigating the rapid degradation and polymerization often observed during the isolation of this sensitive beta-lactam structure. For R&D directors and process chemists, this represents a critical advancement in securing high-purity active pharmaceutical ingredients (APIs) while maintaining robust process parameters suitable for industrial translation.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the purification of carbapenem antibiotics has been fraught with challenges related to thermal and hydrolytic instability. Prior art, such as the methods disclosed in CN1432016A, typically involves dissolving the crude antibiotic in pure water at elevated temperatures, often around 55°C, followed by cooling to induce crystallization. However, carbapenem compounds exhibit poor stability in aqueous solutions, a defect that is exacerbated at higher concentrations and temperatures. Under these conventional conditions, the molecule is prone to undergoing undesirable side reactions, including color darkening, intermolecular polymerization, and ring-opening degradation. These degradation pathways not only compromise the visual quality of the product but also lead to significant losses in yield and the formation of difficult-to-remove impurities, ultimately resulting in a final product with suboptimal purity that may fail to meet stringent regulatory specifications for parenteral antibiotics.
The Novel Approach
The novel approach detailed in the patent data circumvents these stability issues by introducing an amino acid, specifically proline, into the crystallization solvent system. By dissolving crude Doripenem in an aqueous solution containing L-proline, the process creates a stabilized environment that allows for dissolution at temperatures between 40°C and 55°C without significant degradation. Comparative experimental data highlights the efficacy of this method: while a conventional water-only recrystallization yielded only 74% recovery with a purity of 98.95%, the proline-assisted method achieved a recovery rate of 91% with a purity exceeding 99.65%. This significant improvement in both yield and quality demonstrates the capacity of the proline additive to suppress degradation kinetics during the critical dissolution and cooling phases.
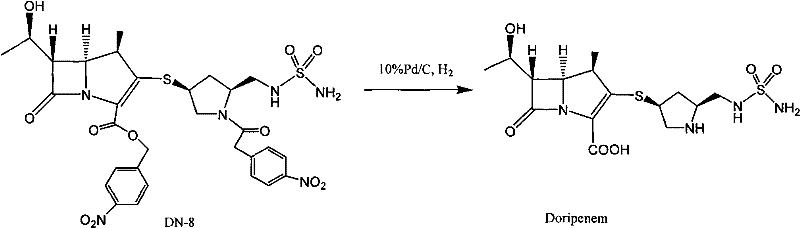
Mechanistic Insights into Proline-Stabilized Crystallization
The underlying mechanism of this purification strategy likely involves complex solute-solvent interactions that stabilize the beta-lactam ring against hydrolytic attack. Carbapenems are notoriously susceptible to nucleophilic attack by water, especially when thermal energy is applied to increase solubility. The presence of proline, a cyclic imino acid, may act as a buffer or a competitive inhibitor for degradation pathways, potentially through hydrogen bonding networks that shield the reactive beta-lactam carbonyl or by modifying the local pH microenvironment to a range more favorable for stability. Furthermore, the zwitterionic nature of proline in aqueous solution could influence the ionic strength and dielectric constant of the medium, thereby reducing the rate of polymerization reactions that lead to high molecular weight impurities. This stabilization allows the process to operate at higher temperatures (45-50°C) which improves solubility and mass transfer, yet upon cooling to 0-5°C, the supersaturation driving force remains sufficient to drive high-yield crystallization without the concomitant formation of degradation by-products.
Impurity control is further enhanced by the selectivity of the crystallization process in the presence of proline. The data indicates that the method effectively excludes related substances and polymeric impurities from the crystal lattice. Analytical results confirm that the final product contains no detectable residues of L-proline, suggesting that the amino acid remains in the mother liquor during the filtration step. This is crucial for regulatory compliance, as it eliminates the need for additional washing steps to remove the stabilizing agent. The ability to achieve purity levels greater than 99.6% directly from the crystallization step significantly reduces the burden on downstream processing and analytical quality control, ensuring that the final API meets the rigorous standards required for injectable formulations.
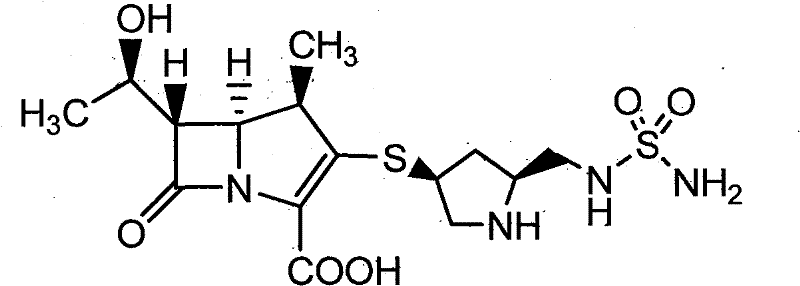
How to Synthesize Doripenem Efficiently
The operational protocol for this purification method is designed for scalability and reproducibility in a GMP manufacturing environment. The process begins with the preparation of a crystallization solution by dissolving crude Doripenem and L-proline in water, preferably heating the mixture to between 45°C and 50°C to ensure complete dissolution. Following optional decolorization with activated carbon, the solution is cooled to a temperature range of 0°C to 5°C under agitation to initiate nucleation and crystal growth. To further maximize recovery, an alcoholic anti-solvent such as ethanol or isopropanol is slowly added to the cooled suspension, inducing additional precipitation of the product. The resulting crystals are then separated by filtration, washed with an alcohol-water mixture to remove surface impurities, and dried under reduced pressure at moderate temperatures (20-60°C) to obtain the final high-purity solid with controlled water content.
- Dissolve crude Doripenem and L-proline in water at 40-55°C to form a stable crystallization solution.
- Cool the solution to 0-5°C under agitation to induce crystallization, optionally adding an alcoholic anti-solvent.
- Separate the crystals via filtration, wash with alcohol-water mixture, and dry under reduced pressure at 20-60°C.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain leaders, the adoption of this proline-assisted purification technology offers substantial strategic benefits beyond mere technical performance. The primary advantage lies in the drastic improvement of process efficiency, which translates directly into cost reduction in pharmaceutical intermediates manufacturing. By increasing the yield from typical levels of roughly 74% to over 90%, the amount of raw material required to produce a kilogram of finished API is significantly reduced. This efficiency gain minimizes waste generation and lowers the cost of goods sold (COGS), providing a competitive edge in pricing negotiations. Furthermore, the enhanced stability of the intermediate solution reduces the risk of batch failures due to degradation, ensuring more predictable production schedules and reliable inventory availability for downstream formulation partners.
- Cost Reduction in Manufacturing: The elimination of extensive reprocessing steps required to salvage degraded batches leads to substantial cost savings. Since the proline additive prevents the formation of polymeric impurities and color bodies, the need for repeated recrystallizations or complex chromatographic purifications is obviated. This streamlining of the purification train reduces solvent consumption, energy usage for heating and cooling cycles, and labor hours, all of which contribute to a leaner and more economical manufacturing process without compromising on the quality of the high-purity Doripenem produced.
- Enhanced Supply Chain Reliability: The robustness of this crystallization method ensures consistent output quality, which is critical for maintaining long-term supply contracts with major pharmaceutical companies. The use of readily available and inexpensive reagents like L-proline and common alcohols (ethanol, isopropanol) mitigates the risk of supply disruptions associated with exotic or highly regulated specialty chemicals. This reliance on commodity-grade inputs enhances the resilience of the supply chain, allowing for rapid scale-up from pilot batches to commercial production volumes of 100 MT annually without the bottleneck of sourcing specialized purification additives.
- Scalability and Environmental Compliance: From an environmental perspective, the process generates less hazardous waste due to higher yields and fewer purification cycles. The solvents used are standard alcohols and water, which are easier to recover and recycle compared to halogenated solvents often used in alternative purification schemes. This aligns with green chemistry principles and simplifies compliance with increasingly stringent environmental regulations regarding solvent emissions and wastewater treatment. The ability to scale this process commercially while maintaining strict purity specifications makes it an ideal candidate for sustainable API manufacturing operations.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this purification technology. Understanding these details is essential for technical teams evaluating the feasibility of integrating this method into existing production lines. The answers are derived directly from the experimental data and process descriptions provided in the patent literature, ensuring accuracy and relevance for decision-makers assessing the potential for technology transfer and commercial partnership.
Q: How does L-proline improve the stability of Doripenem during purification?
A: L-proline acts as a stabilizing agent in the aqueous crystallization medium, significantly reducing degradation pathways such as polymerization and hydrolysis that typically occur at elevated temperatures (40-55°C) in pure water systems.
Q: What purity levels can be achieved with this proline-assisted method?
A: The method consistently achieves Doripenem purity greater than 99.0%, with optimized embodiments reaching purity levels exceeding 99.6% and yields up to 93%, surpassing conventional water-only crystallization techniques.
Q: Is residual proline present in the final purified product?
A: No, analytical testing using HPLC with amino column separation confirms that no L-proline residues are detected in the final purified Doripenem batches, ensuring compliance with strict impurity specifications.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Doripenem Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of purity and stability in the production of complex beta-lactam antibiotics like Doripenem. As a leading CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that our clients receive consistent, high-quality materials regardless of volume. Our state-of-the-art facilities are equipped with rigorous QC labs capable of verifying stringent purity specifications, including the detection of trace impurities and residual solvents, guaranteeing that every batch meets the highest international pharmacopoeial standards.
We invite global pharmaceutical partners to collaborate with us to leverage this advanced purification technology for their supply chains. By contacting our technical procurement team, you can request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We encourage you to reach out today to obtain specific COA data and route feasibility assessments, ensuring that your project moves forward with the most efficient and reliable manufacturing strategy available in the market.
