Revolutionizing Ondansetron Intermediate Production via One-Step Microwave Synthesis
The pharmaceutical industry is constantly seeking more efficient pathways to synthesize critical antiemetic agents, and the recent disclosure in patent CN115677559A presents a transformative approach to producing the key ondansetron intermediate, tetrahydrocarbazolone. This patent details a novel one-step synthesis method that leverages microwave irradiation and alkaline catalysis to convert 2-bromo-1,3-cyclohexanedione and aniline directly into the target carbazole scaffold. Unlike traditional multi-step sequences that often suffer from low overall yields and cumbersome purification requirements, this innovation achieves high conversion rates and exceptional purity in a fraction of the time. For R&D directors and procurement specialists, this represents a significant opportunity to optimize the supply chain for 5-HT3 receptor antagonists by adopting a process that is not only chemically elegant but also economically superior. The ability to bypass expensive transition metal catalysts and lengthy reaction times addresses two of the most persistent pain points in fine chemical manufacturing: cost control and production throughput.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of tetrahydrocarbazolone has relied on complex multi-step protocols that introduce significant inefficiencies into the manufacturing workflow. Prior art, such as methods utilizing dimeric rhodium acetate or palladium catalysts, requires harsh conditions, extended reaction times ranging from several hours to overnight, and the use of chlorinated solvents like 1,2-dichloroethane. These conventional routes are plagued by the high cost of noble metal catalysts, which not only inflate the bill of materials but also necessitate rigorous and expensive post-reaction purification steps to remove trace metal residues to meet pharmaceutical standards. Furthermore, the reliance on silica gel column chromatography for purification in many literature methods creates a bottleneck for scalability, generating substantial solid waste and limiting the batch size that can be practically processed. The cumulative effect of these factors is a process with a low total yield, often hovering around 20%, which severely impacts the economic viability of producing ondansetron on a commercial scale.
The Novel Approach
In stark contrast, the methodology described in CN115677559A streamlines the entire operation into a single, rapid transformation driven by microwave energy. By selecting 2-bromo-1,3-cyclohexanedione and aniline as the starting materials, the process capitalizes on the inherent reactivity of the alpha-bromo ketone to facilitate immediate cyclization under alkaline conditions. The use of microwave irradiation at 300-400W accelerates the reaction kinetics dramatically, allowing the transformation to reach completion in merely 5 to 10 minutes. This drastic reduction in cycle time is complemented by a simplified workup procedure where the product precipitates or is easily isolated without the need for column chromatography. The visual representation of this streamlined reaction pathway highlights the direct conversion from simple precursors to the complex heterocyclic core, underscoring the efficiency gains achievable through this modern synthetic strategy.
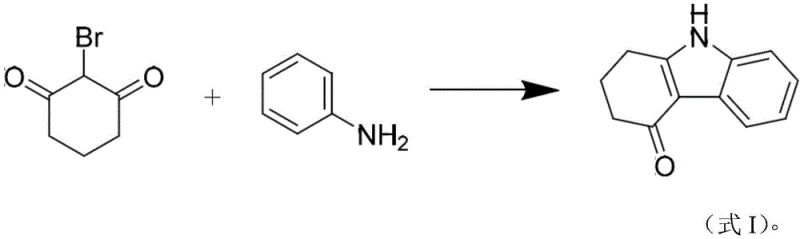
Mechanistic Insights into Alkaline-Catalyzed Microwave Cyclization
The core of this technological advancement lies in the synergistic combination of nucleophilic substitution and intramolecular cyclization facilitated by a basic environment and microwave heating. Mechanistically, the aniline nitrogen acts as a nucleophile, attacking the electrophilic carbon centers of the 2-bromo-1,3-cyclohexanedione. The presence of an alkaline catalyst, such as potassium hydroxide or sodium carbonate, serves a dual function: it activates the nucleophile and simultaneously neutralizes the hydrogen bromide byproduct generated during the substitution step. Crucially, the patent specifies that aniline is used in excess, effectively serving as both a reactant and an acid-binding agent, which drives the equilibrium towards the formation of the target tetrahydrocarbazolone. This self-buffering system prevents the accumulation of acidic species that could otherwise degrade the product or inhibit the reaction progress, ensuring a clean conversion profile.
Furthermore, the application of microwave irradiation provides uniform and rapid heating that enhances the selectivity of the cyclization process. Unlike conventional conductive heating, which can create thermal gradients and hot spots leading to side reactions, microwave energy couples directly with the polar molecules in the reaction mixture, promoting the specific bond formations required for the carbazole ring closure. This results in a product with high structural integrity and minimal impurity formation, as evidenced by the reported HPLC purity exceeding 98%. The mechanism avoids the formation of complex organometallic intermediates associated with palladium catalysis, thereby eliminating the risk of metal contamination and simplifying the impurity profile. This mechanistic clarity provides R&D teams with confidence in the robustness of the process, as the reaction parameters are straightforward to control and optimize for consistent quality.
How to Synthesize Tetrahydrocarbazolone Efficiently
Implementing this synthesis route requires precise control over the stoichiometry and energy input to maximize the benefits of the microwave-assisted protocol. The process begins with the careful mixing of the bromo-dione substrate and an excess of aniline, followed by the addition of a dilute alkaline catalyst solution. The reaction mixture is then subjected to controlled microwave irradiation, where the power output and duration are critical variables to monitor. Detailed standard operating procedures regarding the specific ratios, temperature monitoring, and safety protocols for microwave reactors are essential for successful technology transfer.
- Mix 2-bromo-1,3-cyclohexanedione and excess aniline in a reaction vessel equipped with a stirrer.
- Add an alkaline catalyst solution (e.g., 5% KOH or NaOH) and initiate microwave irradiation at 300-400W for 5-10 minutes.
- Cool the mixture, filter, distill off excess aniline for reuse, and recrystallize the residue from ethanol to obtain the pure solid product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain leaders, the adoption of this one-step microwave synthesis offers compelling economic and logistical benefits that extend far beyond simple yield improvements. The elimination of precious metal catalysts like palladium and rhodium removes a major source of cost volatility and supply risk, as these metals are subject to fluctuating market prices and geopolitical constraints. Additionally, the drastic reduction in reaction time from hours to minutes significantly increases the throughput capacity of existing manufacturing facilities, allowing for faster turnaround times on orders and improved responsiveness to market demand. The simplified purification process, which foregoes silica gel chromatography in favor of crystallization and washing, reduces the consumption of solvents and stationary phases, leading to lower operational expenditures and a smaller environmental footprint.
- Cost Reduction in Manufacturing: The replacement of expensive transition metal catalysts with inexpensive alkaline bases such as sodium hydroxide or potassium carbonate results in a substantial decrease in raw material costs. Moreover, the ability to recover and reuse the excess aniline solvent further enhances the economic efficiency of the process by minimizing waste disposal costs and reducing the net consumption of reagents. The avoidance of column chromatography also translates to significant savings in labor and materials, as this step is notoriously resource-intensive and difficult to scale economically.
- Enhanced Supply Chain Reliability: By relying on commodity chemicals like aniline and cyclohexanedione derivatives, the supply chain becomes more resilient against disruptions that often affect specialized reagents. The short reaction cycle time of 5 to 10 minutes allows for just-in-time manufacturing strategies, reducing the need for large inventory buffers and freeing up working capital. This agility ensures a steady and reliable flow of high-purity intermediates to downstream API manufacturers, mitigating the risk of production delays.
- Scalability and Environmental Compliance: The process is inherently greener due to the absence of heavy metals and the reduction in solvent usage, aligning with increasingly stringent environmental regulations. The simplicity of the workup procedure facilitates easier scale-up from laboratory to pilot and commercial scales without the need for complex engineering modifications. This scalability ensures that the method can meet the growing global demand for ondansetron without compromising on quality or sustainability goals.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this patented synthesis method. These answers are derived directly from the experimental data and beneficial effects reported in the patent documentation, providing a clear understanding of the process capabilities.
Q: What are the primary advantages of the microwave-assisted method over traditional palladium-catalyzed routes?
A: The microwave-assisted method eliminates the need for expensive transition metal catalysts like palladium or rhodium, significantly reducing raw material costs. Furthermore, it reduces reaction time from several hours or overnight to merely 5-10 minutes and removes the necessity for silica gel column chromatography, simplifying downstream processing.
Q: How does the process handle the removal of byproducts and unreacted starting materials?
A: The process utilizes excess aniline which serves a dual purpose as both a reactant and a solvent. Post-reaction, the excess aniline is distilled under reduced pressure and can be recovered and reused. The crude product is then purified via simple washing with dilute hydrochloric acid and water, followed by ethanol recrystallization, avoiding complex extraction procedures.
Q: Is this synthesis method suitable for large-scale industrial manufacturing?
A: Yes, the method is highly suitable for industrial scale-up due to its one-step nature, use of inexpensive and readily available raw materials, and the elimination of hazardous heavy metal catalysts. The short reaction cycle time allows for high throughput, and the simplified workup procedure reduces waste generation and operational complexity.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Tetrahydrocarbazolone Supplier
At NINGBO INNO PHARMCHEM, we recognize the strategic value of adopting advanced synthetic methodologies like the one described in CN115677559A to enhance the competitiveness of our pharmaceutical intermediate portfolio. As a dedicated CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory innovation to industrial reality is seamless and efficient. Our commitment to quality is underpinned by stringent purity specifications and rigorous QC labs that utilize state-of-the-art analytical instrumentation to verify every batch, guaranteeing that our tetrahydrocarbazolone meets the highest standards required for API synthesis.
We invite potential partners to engage with our technical team to explore how this cost-effective and high-yield route can be integrated into your supply chain. By requesting a Customized Cost-Saving Analysis, you can gain deeper insights into the specific economic benefits tailored to your volume requirements. We encourage you to contact our technical procurement team today to索取 specific COA data and route feasibility assessments, ensuring that your project moves forward with the most robust and economical chemistry available.
