Revolutionizing Ondansetron Intermediate Production via Microwave-Assisted One-Step Cyclization
The pharmaceutical industry continuously seeks robust methodologies to optimize the production of critical anti-emetic agents, specifically focusing on the synthesis of ondansetron intermediates. Patent CN115677559A introduces a groundbreaking one-step synthesis method for tetrahydrocarbazolone, a pivotal precursor in the manufacturing of ondansetron. This innovation leverages microwave irradiation to facilitate the cyclization of 2-bromo-1,3-cyclohexanedione and aniline under alkaline conditions, effectively bypassing the multi-step complexities and high costs associated with traditional routes. For R&D directors and procurement specialists, this technology represents a paradigm shift towards greener, faster, and more cost-effective chemical manufacturing, addressing long-standing inefficiencies in the supply chain of serotonin receptor antagonists.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of tetrahydrocarbazolone has been plagued by significant technical and economic hurdles that hinder efficient large-scale production. Conventional literature describes routes relying on expensive transition metal catalysts, such as dimeric rhodium acetate or palladium complexes, which not only inflate raw material costs but also introduce stringent regulatory burdens regarding heavy metal residues in the final API. Furthermore, these traditional methods often require prolonged reaction times, ranging from overnight heating to twelve-hour cycles at elevated temperatures, which drastically limits reactor throughput and increases energy consumption. The necessity for complex purification steps, including silica gel column chromatography, further exacerbates operational expenses and generates substantial chemical waste, rendering these processes less attractive for modern, sustainability-focused manufacturing environments.
The Novel Approach
In stark contrast, the novel approach detailed in the patent utilizes a direct, one-step condensation and cyclization strategy driven by microwave energy. By employing inexpensive and readily available starting materials like 2-bromo-1,3-cyclohexanedione and aniline, the process eliminates the dependency on precious metal catalysts entirely. The reaction is completed within a remarkably short window of 5 to 10 minutes under mild alkaline catalysis, achieving conversion rates and yields that far exceed those of legacy methods. This streamlined workflow not only simplifies the operational procedure by removing the need for column chromatography but also enhances the overall atom economy, making it an ideal candidate for reliable pharmaceutical intermediates supplier networks aiming to reduce lead times and operational overheads.
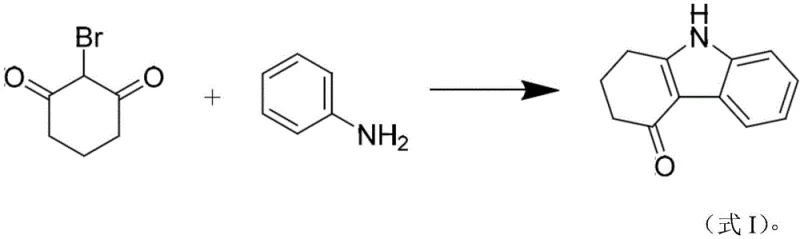
Mechanistic Insights into Microwave-Assisted Alkaline Cyclization
The core of this technological advancement lies in the synergistic effect of microwave irradiation and the dual role of aniline within the reaction matrix. Mechanistically, the alkaline catalyst, such as potassium hydroxide or sodium carbonate, facilitates the deprotonation and activation of the nucleophilic sites on the aniline molecule, promoting a rapid attack on the electrophilic carbonyl centers of the cyclohexanedione derivative. The microwave energy provides uniform and instantaneous heating, which lowers the activation energy barrier for the cyclization step, driving the equilibrium towards the formation of the tetrahydrocarbazolone ring system with exceptional speed and selectivity. This precise thermal control prevents the formation of polymeric by-products often seen in conventional conductive heating, ensuring a cleaner reaction profile.
Furthermore, the process ingeniously utilizes an excess of aniline to serve as an acid-binding agent, effectively neutralizing the hydrogen bromide (HBr) generated as a byproduct during the substitution phase. This in-situ neutralization prevents acid-catalyzed degradation of the product and shifts the reaction equilibrium forward, significantly improving the yield to over 90 percent. The absence of transition metals means there is no risk of metal-catalyzed side reactions or the formation of difficult-to-remove organometallic impurities, resulting in a crude product with HPLC purity exceeding 98 percent. This high level of intrinsic purity simplifies downstream processing, as the product can be isolated through simple filtration and recrystallization rather than complex chromatographic separation.
How to Synthesize Tetrahydrocarbazolone Efficiently
The synthesis protocol outlined in the patent offers a straightforward pathway for laboratories and pilot plants to replicate these high-yield results. The procedure involves mixing the diketone substrate with aniline in a specific weight ratio, followed by the addition of a dilute alkaline solution. The mixture is then subjected to controlled microwave irradiation, after which the excess aniline is recovered via distillation for reuse, enhancing the green chemistry profile of the operation. For detailed standard operating procedures and specific parameter optimizations, please refer to the technical guide below.
- Mix 2-bromo-1,3-cyclohexanedione and excess aniline (acting as both reactant and solvent) in a reaction vessel equipped with a stirrer.
- Add an alkaline catalyst solution (such as 5% KOH or NaOH) and subject the mixture to microwave irradiation (300-400W) for 5 to 10 minutes.
- Cool the reaction, filter, distill off excess aniline for reuse, and recrystallize the residue from ethanol to obtain the white solid product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this microwave-assisted synthesis route offers transformative benefits that directly impact the bottom line and operational resilience. The elimination of expensive noble metal catalysts such as rhodium and palladium removes a major variable cost driver, leading to substantial cost savings in raw material procurement. Additionally, the drastic reduction in reaction time from hours to minutes allows for significantly higher batch turnover rates within existing infrastructure, effectively increasing production capacity without the need for capital-intensive equipment expansion. The simplified workup procedure, which avoids silica gel columns and toxic solvents, further reduces waste disposal costs and aligns with increasingly strict environmental regulations.
- Cost Reduction in Manufacturing: The removal of precious metal catalysts eliminates the need for costly metal scavenging resins and complex purification steps, directly lowering the cost of goods sold. The ability to recover and reuse excess aniline as a solvent further minimizes material waste and purchasing requirements, creating a closed-loop system that maximizes resource efficiency. Moreover, the reduced energy consumption due to shorter reaction times contributes to lower utility bills, compounding the financial advantages of this method over traditional thermal processes.
- Enhanced Supply Chain Reliability: By relying on commodity chemicals like aniline and simple inorganic bases rather than specialized organometallic catalysts, the supply chain becomes far more robust against market fluctuations and vendor shortages. The simplicity of the reaction setup reduces the risk of batch failures due to catalyst deactivation or sensitivity, ensuring consistent output quality and delivery schedules. This reliability is crucial for maintaining continuous API production lines and meeting the rigorous demands of global pharmaceutical markets without interruption.
- Scalability and Environmental Compliance: The process is inherently scalable, as microwave technology has advanced to support industrial-level continuous flow reactors, allowing for seamless transition from lab to commercial scale. The absence of heavy metals and the reduction in organic solvent usage simplify wastewater treatment and regulatory compliance, reducing the environmental footprint of the manufacturing facility. This alignment with green chemistry principles not only mitigates regulatory risks but also enhances the corporate sustainability profile, which is increasingly valued by stakeholders and partners.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis technology. These insights are derived directly from the experimental data and beneficial effects reported in the patent documentation, providing clarity on yield, purity, and operational feasibility for potential adopters.
Q: What are the primary advantages of this microwave-assisted method over traditional palladium-catalyzed routes?
A: The microwave-assisted method eliminates the need for expensive transition metal catalysts like palladium or rhodium, significantly reducing raw material costs and removing the complex post-reaction heavy metal clearance steps required in conventional synthesis.
Q: How does the process ensure high purity without silica gel column chromatography?
A: By utilizing excess aniline as both a reactant and an acid-binding agent to neutralize HBr, combined with precise microwave control, the reaction achieves high selectivity (>98% HPLC purity), allowing for simple recrystallization instead of labor-intensive column chromatography.
Q: Is this synthesis method suitable for large-scale industrial production?
A: Yes, the process uses readily available raw materials, operates with short reaction times (5-10 minutes), and avoids hazardous solvents or expensive catalysts, making it highly scalable and economically viable for commercial manufacturing.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Tetrahydrocarbazolone Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of efficient intermediate synthesis in the broader context of API manufacturing. As a leading CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that innovations like this microwave-assisted route are translated into tangible supply chain solutions. Our state-of-the-art facilities are equipped with rigorous QC labs capable of meeting stringent purity specifications, guaranteeing that every batch of tetrahydrocarbazolone delivered meets the highest international standards for pharmaceutical applications.
We invite global partners to collaborate with us to leverage this advanced technology for their ondansetron production needs. By contacting our technical procurement team, you can request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We encourage you to reach out today to obtain specific COA data and comprehensive route feasibility assessments, ensuring a seamless integration of this high-efficiency intermediate into your manufacturing portfolio.
