Advanced Synthetic Route for High-Purity Resveratrol and Piceatannol Intermediates
The pharmaceutical and nutraceutical industries have long recognized the therapeutic potential of polyhydroxystilbenes, particularly resveratrol and piceatannol, due to their potent antioxidant and anti-inflammatory properties. However, the industrial production of these high-value compounds has historically been hindered by complex synthetic challenges, specifically regarding stereochemical control and purification costs. Patent CN101495436B introduces a transformative methodology for the synthesis of (E)-stilbene derivatives that addresses these critical bottlenecks. By shifting the synthetic strategy away from traditional Wittig or Heck couplings, this novel process utilizes 1,2-diarylethanone intermediates to achieve superior stereoselectivity. This technical breakthrough ensures that manufacturers can access reliable pharmaceutical intermediates supplier channels with materials that require significantly less downstream processing. The ability to directly obtain the desired (E)-isomer without extensive isomerization steps represents a paradigm shift in fine chemical intermediates manufacturing, offering a robust pathway for commercial scale-up.
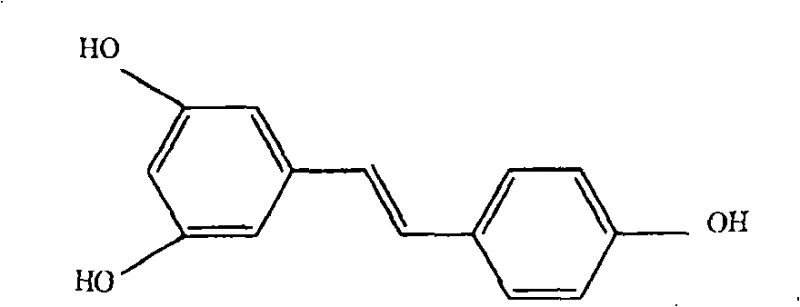
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditional synthetic routes for resveratrol and its analogues, such as the Wittig or Wittig-Horner reactions, suffer from inherent stereochemical deficiencies that complicate large-scale production. These methods typically generate a mixture of (E) and (Z) stilbene isomers, necessitating difficult and costly separation processes to isolate the biologically active (E)-form. Furthermore, converting the unwanted (Z)-isomer often requires additional catalytic steps involving iodine or expensive palladium complexes, which introduce impurities and increase the environmental footprint. Similarly, Heck-type reactions, while powerful, rely on precious metal catalysts that are not only costly but also unstable under certain reaction conditions, leading to inconsistent yields and variable product quality. The Perkin reaction approach, another historical alternative, demands harsh decarboxylation conditions at high temperatures, which can degrade sensitive functional groups and limit the scope of applicable substrates. These cumulative inefficiencies result in prolonged lead times and elevated production costs, making cost reduction in fine chemical intermediates manufacturing a persistent challenge for procurement teams seeking scalable solutions.
The Novel Approach
The innovative process detailed in the patent circumvents these issues by employing 1,2-diarylethanone derivatives as pivotal synthetic intermediates. This strategy leverages the structural rigidity of the ketone precursor to facilitate the formation of the (E)-stilbene backbone with high fidelity. As illustrated by the core intermediate structure, the 1,2-diarylethanone framework allows for controlled transformation into the target olefin through either reduction-dehydration or hydrazone elimination pathways. 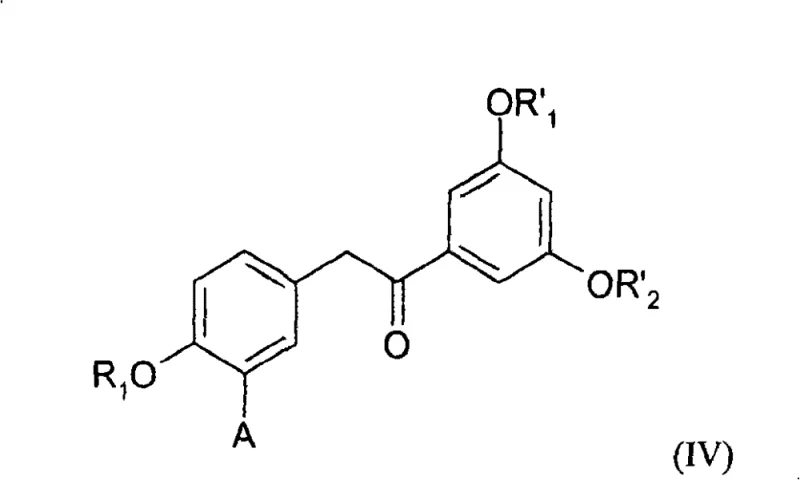 This approach eliminates the need for problematic E/Z isomer separation entirely, as the reaction conditions are tuned to favor the thermodynamic (E)-product. Moreover, the starting materials for this route, such as hydroxyaromatic acids and esters, are commercially abundant and inexpensive compared to the specialized phosphonium salts or vinyl halides required for Wittig and Heck reactions. By simplifying the synthetic sequence and removing the dependency on precious metal catalysts, this novel approach provides a streamlined, cost-effective solution for producing high-purity resveratrol precursors suitable for rigorous pharmaceutical applications.
This approach eliminates the need for problematic E/Z isomer separation entirely, as the reaction conditions are tuned to favor the thermodynamic (E)-product. Moreover, the starting materials for this route, such as hydroxyaromatic acids and esters, are commercially abundant and inexpensive compared to the specialized phosphonium salts or vinyl halides required for Wittig and Heck reactions. By simplifying the synthetic sequence and removing the dependency on precious metal catalysts, this novel approach provides a streamlined, cost-effective solution for producing high-purity resveratrol precursors suitable for rigorous pharmaceutical applications.
Mechanistic Insights into Claisen Condensation and Decarboxylation
The foundation of this synthetic strategy lies in the efficient construction of the carbon skeleton via a Claisen-type condensation followed by a controlled decarboxylation. The process begins with the reaction between ether/ester derivatives, specifically utilizing strong bases such as sodium hydride or sodium ethoxide to drive the condensation. This step generates beta-ketoesters of formula (III), which serve as the precursors to the key ketone intermediates. 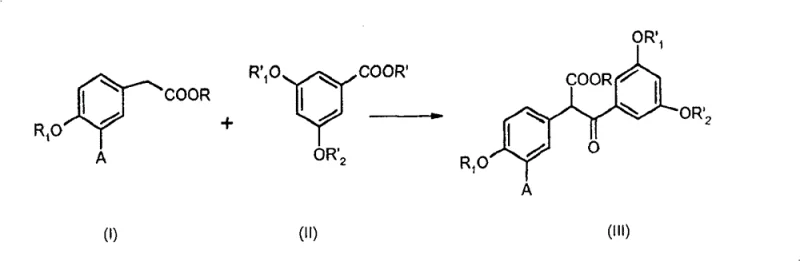 The subsequent decarboxylation of these beta-ketoesters is a critical unit operation that can be performed under remarkably mild and environmentally favorable conditions. Unlike traditional acidic decarboxylation which often requires corrosive reagents and solvents, this patent describes a solvent-free protocol using boric acid or boric anhydride at temperatures between 100°C and 180°C. This specific mechanistic pathway not only enhances atom economy by avoiding solvent waste but also simplifies the isolation of the resulting 1,2-diarylethanone. The use of boric acid acts as a mild Lewis acid catalyst that facilitates the loss of carbon dioxide without compromising the integrity of the sensitive methoxy or benzyloxy protecting groups on the aromatic rings. This level of control over the reaction mechanism is essential for maintaining high purity specifications and minimizing the formation of side products that could complicate downstream purification.
The subsequent decarboxylation of these beta-ketoesters is a critical unit operation that can be performed under remarkably mild and environmentally favorable conditions. Unlike traditional acidic decarboxylation which often requires corrosive reagents and solvents, this patent describes a solvent-free protocol using boric acid or boric anhydride at temperatures between 100°C and 180°C. This specific mechanistic pathway not only enhances atom economy by avoiding solvent waste but also simplifies the isolation of the resulting 1,2-diarylethanone. The use of boric acid acts as a mild Lewis acid catalyst that facilitates the loss of carbon dioxide without compromising the integrity of the sensitive methoxy or benzyloxy protecting groups on the aromatic rings. This level of control over the reaction mechanism is essential for maintaining high purity specifications and minimizing the formation of side products that could complicate downstream purification.
Following the formation of the ketone intermediate, the conversion to the stilbene derivative proceeds through highly selective mechanisms that ensure stereochemical purity. In Route A, the ketone is reduced to a 1,2-diarylethanol using metal hydrides like sodium borohydride or catalytic hydrogenation, followed by acid-catalyzed dehydration. The dehydration step, typically conducted in aromatic solvents like toluene with p-toluenesulfonic acid, promotes the elimination of water to form the double bond. The thermodynamic stability of the (E)-isomer drives this equilibrium, ensuring that the final product is predominantly the desired trans-stilbene. Alternatively, Route B employs a Shapiro or Bamford-Stevens type elimination via an arylsulfonylhydrazone intermediate. Reacting the ketone with tosylhydrazide forms a hydrazone, which upon treatment with strong bases like potassium tert-butoxide, undergoes nitrogen extrusion to yield the alkene. This mechanistic diversity offers process chemists flexibility in optimizing conditions for specific substrates, allowing for precise impurity control. By selecting the appropriate pathway, manufacturers can tailor the synthesis to minimize specific byproducts, thereby enhancing the overall quality of the high-purity resveratrol intermediates and ensuring compliance with stringent regulatory standards for pharmaceutical ingredients.
How to Synthesize (E)-Stilbene Derivatives Efficiently
The synthesis of these valuable stilbene derivatives is achieved through a modular sequence that prioritizes operational simplicity and yield optimization. The process initiates with the preparation of the beta-ketoester backbone, followed by decarboxylation to the ketone, and concludes with the formation of the double bond and final deprotection. Each step has been optimized to utilize standard laboratory equipment and readily available reagents, making the transition from bench scale to pilot plant straightforward. The detailed standardized synthesis steps outlined below provide a clear roadmap for replicating the high yields and purity described in the patent documentation, ensuring that technical teams can implement this route with confidence.
- Perform Claisen-type condensation between ether/ester derivatives using strong bases like sodium hydride to form beta-ketoesters.
- Execute decarboxylation of the beta-ketoester using boric acid at elevated temperatures (100-180°C) to yield 1,2-diarylethanones.
- Convert the ketone to the (E)-stilbene derivative via reduction-dehydration or tosylhydrazone elimination, followed by deprotection.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this synthetic route offers substantial strategic benefits that extend beyond simple chemistry. The primary advantage lies in the drastic simplification of the supply chain for raw materials. By utilizing common hydroxyaromatic acids and esters instead of specialized phosphonium salts or palladium catalysts, the process reduces dependency on volatile specialty chemical markets. This shift ensures enhanced supply chain reliability, as the starting materials are produced by multiple global vendors, mitigating the risk of single-source bottlenecks. Furthermore, the elimination of expensive transition metal catalysts removes the need for complex and costly heavy metal scavenging steps, which are often required to meet residual metal specifications in pharmaceutical grades. This reduction in processing steps translates directly into lower operational expenditures and a smaller environmental footprint, aligning with modern sustainability goals.
- Cost Reduction in Manufacturing: The economic impact of this process is driven by the removal of high-cost reagents and the simplification of purification workflows. Traditional methods often incur significant expenses related to palladium catalysts and the chromatographic separation of isomers. By contrast, this novel route relies on inexpensive base metals and crystallization-based purifications, which are inherently more scalable and cost-efficient. The solvent-free decarboxylation step further reduces costs by minimizing solvent purchase, recovery, and disposal expenses. Additionally, the high stereoselectivity of the dehydration and elimination steps means that less material is lost to unwanted isomers, improving the overall mass balance and yield of the final active ingredient. These factors combine to deliver substantial cost savings in pharmaceutical intermediates manufacturing, allowing for more competitive pricing in the final market.
- Enhanced Supply Chain Reliability: Supply chain resilience is significantly improved by the robustness of the chemical transformations involved. The reaction conditions, such as the use of boric acid for decarboxylation and standard bases for condensation, are tolerant of minor variations in feedstock quality, ensuring consistent output even with fluctuating raw material batches. The avoidance of air- and moisture-sensitive catalysts, which are common in Heck couplings, reduces the need for specialized handling equipment and inert atmosphere protocols. This operational robustness facilitates smoother logistics and storage, reducing the risk of production delays caused by reagent degradation or handling errors. Consequently, partners can expect reducing lead time for high-purity stilbene derivatives, enabling faster response to market demand fluctuations and more reliable inventory management for downstream formulation.
- Scalability and Environmental Compliance: From a scale-up perspective, the process is designed to accommodate commercial scale-up of complex polyphenolic compounds with minimal engineering hurdles. The exothermic nature of the condensation and the manageable temperatures of the decarboxylation step allow for safe operation in large-scale reactors without requiring exotic cooling or heating systems. Furthermore, the reduction in solvent usage and the absence of toxic heavy metals simplify waste treatment and regulatory compliance. The ability to perform key steps like decarboxylation without solvents significantly lowers the volume of hazardous waste generated, supporting greener manufacturing initiatives. This alignment with environmental, social, and governance (ESG) criteria makes the process attractive for companies aiming to reduce their carbon footprint while maintaining high production volumes of critical health ingredients.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthetic technology. These insights are derived directly from the experimental data and process descriptions found in the patent literature, providing a factual basis for decision-making. Understanding these details helps stakeholders evaluate the feasibility of integrating this route into their existing manufacturing portfolios.
Q: How does this process avoid E/Z isomer separation issues?
A: Unlike Wittig reactions that produce mixtures, this route utilizes 1,2-diarylethanone intermediates which are converted to (E)-stilbenes via specific dehydration or hydrazone elimination conditions that inherently favor the E-isomer, eliminating the need for difficult chromatographic separation.
Q: What are the cost advantages over Heck coupling methods?
A: This method avoids the use of expensive palladium catalysts required in Heck reactions. Additionally, it utilizes readily available hydroxyaromatic acids and esters as starting materials, significantly reducing raw material costs and catalyst removal expenses.
Q: Is the decarboxylation step environmentally friendly?
A: Yes, the decarboxylation of the beta-ketoester can be performed solvent-free in the presence of boric acid at 100-180°C, which minimizes solvent waste and simplifies the workup procedure compared to traditional acidic decarboxylation methods.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Resveratrol Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of efficient and scalable synthetic routes in the modern pharmaceutical landscape. Our team of expert process chemists has extensively evaluated the technology described in CN101495436B and possesses the capability to adapt and optimize these pathways for industrial production. We bring extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory success to commercial reality is seamless. Our facilities are equipped with rigorous QC labs and advanced analytical instrumentation to guarantee stringent purity specifications for every batch of resveratrol and piceatannol intermediates we produce. We are committed to delivering high-purity resveratrol that meets the exacting standards of the global nutraceutical and pharmaceutical markets.
We invite you to collaborate with us to leverage this advanced synthetic technology for your product pipeline. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements and quality targets. By partnering with us, you gain access to specific COA data and route feasibility assessments that demonstrate the tangible benefits of this novel process. Contact us today to discuss how we can support your supply chain with reliable, cost-effective, and high-quality stilbene derivatives, ensuring your products reach the market faster and more efficiently.
