Optimizing Olaparib Production: A Technical Breakthrough in Intermediate Synthesis and Impurity Control
Optimizing Olaparib Production: A Technical Breakthrough in Intermediate Synthesis and Impurity Control
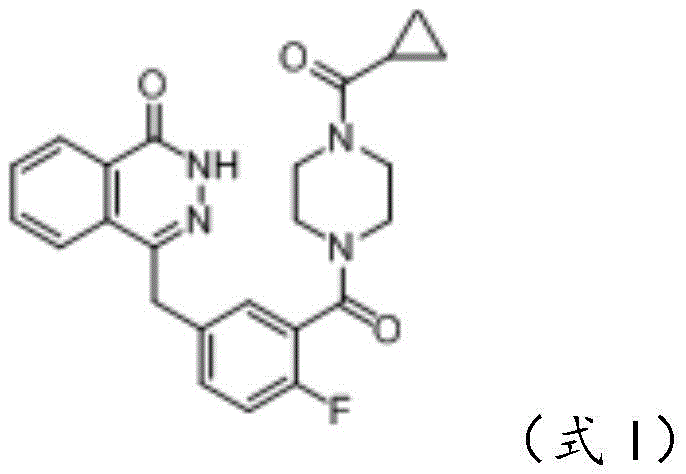
The pharmaceutical landscape for oncology treatments continues to evolve, with Olaparib standing as a cornerstone PARP inhibitor for treating BRCA-deficient ovarian cancer. However, the manufacturing of this critical active pharmaceutical ingredient (API) has historically faced significant challenges regarding yield, purity, and process complexity. Patent CN114075143A introduces a transformative preparation method for Olaparib and its key intermediates, specifically addressing the bottlenecks associated with the synthesis of Compound IV. This technical disclosure outlines a robust one-pot methodology that bypasses the isolation of unstable intermediates, thereby streamlining the production workflow. For R&D directors and supply chain leaders, this represents a pivotal shift towards more efficient, cost-effective, and quality-controlled manufacturing protocols that align with modern Good Manufacturing Practice (GMP) standards.
The strategic value of this patent lies in its ability to enhance the reliability of the olaparib intermediate supplier network by mitigating risks associated with low-yielding steps. By optimizing the synthesis process and purification methods, the invention ensures that the final product meets stringent purity specifications, often exceeding 99.9%. This level of quality control is essential for maintaining the therapeutic efficacy and safety profile of the drug. Furthermore, the method's adaptability to industrial scale-up offers a compelling value proposition for procurement teams seeking to stabilize their supply chains against the volatility of complex chemical manufacturing.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of Olaparib intermediates, as disclosed in prior art such as WO2004080976, relied heavily on the preparation and isolation of a phosphorus ylide intermediate, often referred to as Compound 2 or Compound III in various contexts. This traditional approach presents severe logistical and chemical drawbacks that hinder efficient production. Firstly, the phosphorus ylide intermediate is inherently unstable, prone to degradation during the necessary separation and purification stages. This instability not only complicates the operational workflow but also leads to the generation of degradation products that trigger unnecessary side reactions in subsequent steps.
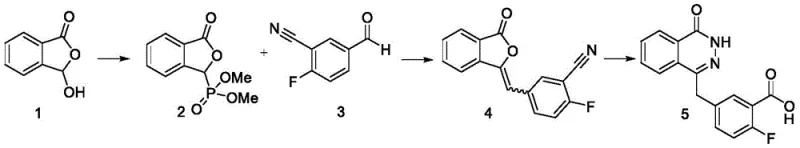
Secondly, the requirement to isolate this intermediate significantly prolongs the production cycle, increasing both labor costs and energy consumption. The separation process itself generates substantial amounts of liquid waste, escalating environmental compliance costs and recycling expenses. Most critically, the conventional method suffers from suboptimal yields, often reported around 59.43% for the key intermediate steps. Additionally, the resulting Olaparib frequently contains Impurity D, a structurally complex byproduct that is exceptionally difficult to remove through standard purification techniques, thereby compromising the overall quality and safety of the medication.
The Novel Approach
In stark contrast to the fragmented and inefficient conventional routes, the novel approach detailed in CN114075143A employs a sophisticated one-pot synthesis strategy for preparing Compound IV. This method ingeniously utilizes the reaction solution containing Compound III directly for the subsequent reaction with Compound IIb, completely eliminating the need for intermediate isolation and purification. By maintaining the reaction mixture in a continuous flow, the process preserves the integrity of the reactive species, preventing the degradation that plagues the traditional method. This seamless transition between reaction stages drastically shortens the process cycle and simplifies the operational requirements.
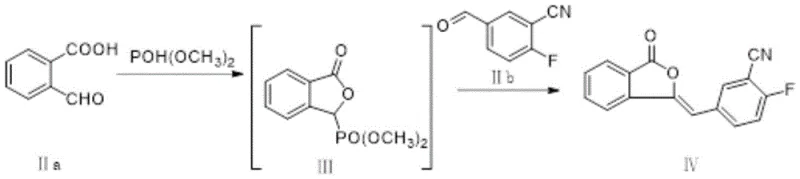
The implementation of this one-pot technique results in a marked improvement in both yield and purity. Experimental data indicates that the yield of Compound IV can be elevated to approximately 76.2%, a significant enhancement over the historical benchmarks. Moreover, the purity of the intermediate consistently reaches levels above 99.8%, with total impurities kept below 0.13%. This high-quality intermediate serves as a superior foundation for the subsequent synthesis of Olaparib, effectively minimizing the formation of hard-to-remove byproducts like Impurity D and ensuring a more robust and predictable manufacturing outcome.
Mechanistic Insights into One-Pot Phosphonate Coupling and Impurity Suppression
The core mechanistic advantage of this new process lies in the precise control of reaction conditions during the formation of the carbon-carbon double bond in Compound IV. In the conventional route, the isolation of the phosphorus ylide exposes it to moisture and oxygen, leading to hydrolysis and oxidation byproducts. These byproducts then participate in side reactions during the coupling with the aldehyde component (Compound IIb), generating a complex mixture of impurities. By keeping the ylide species in situ within the reaction solvent—typically methanol or ethanol—the new method maintains a homogeneous environment that favors the desired Horner-Wadsworth-Emmons type coupling. The addition of acid-binding agents like triethylamine or DIEA in the second stage further drives the equilibrium towards the product, ensuring high conversion rates.
Furthermore, the suppression of Impurity D is a critical mechanistic achievement of this protocol. Impurity D is essentially a dimeric structure formed through the coupling of two phthalazinone units, a side reaction that is promoted by the presence of degraded intermediates and prolonged reaction times. The optimized process reduces the residence time of reactive intermediates and eliminates the purification steps where degradation typically occurs. Consequently, the concentration of precursors that lead to Impurity D is kept to a minimum. Subsequent purification steps, such as recrystallization from dioxane and methanol mixtures, are then far more effective at removing trace amounts of this impurity, allowing the final API to achieve purity levels of 99.92% with Impurity D content restricted to less than 0.08%.
How to Synthesize Olaparib Efficiently
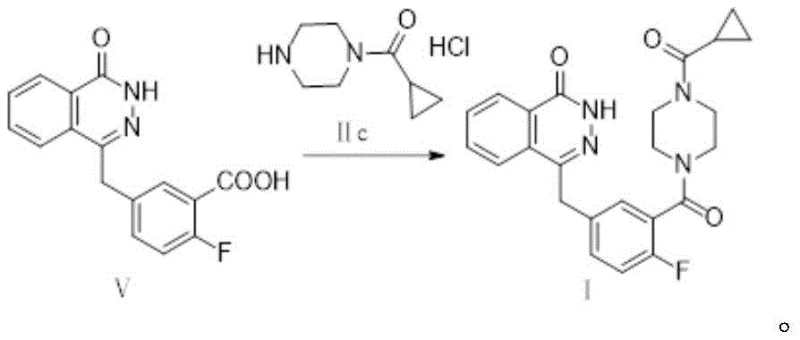
The synthesis of Olaparib via this optimized route involves three distinct but interconnected stages, beginning with the one-pot formation of the key alkene intermediate. The process starts by reacting Compound IIa with dimethyl phosphite in the presence of a base like sodium methoxide to generate the phosphonate species in situ. Without isolation, Compound IIb is introduced to form Compound IV. This intermediate is then subjected to hydrolysis and cyclization with hydrazine hydrate to construct the phthalazinone ring system, yielding Compound V. Finally, Compound V is coupled with the piperazine derivative (Compound IIc) to complete the molecule.
- Perform a one-pot reaction of Compound IIa with dimethyl phosphite and base, followed directly by reaction with Compound IIb to generate Compound IV without isolating the unstable phosphorus ylide intermediate.
- Hydrolyze Compound IV using alkali and subsequently react with hydrazine hydrate under controlled heating to form the phthalazinone core, Compound V.
- Couple Compound V with Compound IIc (cyclopropylcarbonyl piperazine derivative) using a condensing agent like HBTU to finalize the Olaparib structure, followed by rigorous crystallization.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this novel synthesis route offers profound strategic benefits that extend beyond simple chemical efficiency. The elimination of the intermediate isolation step translates directly into a reduction in raw material consumption, particularly solvents and drying agents, which are major cost drivers in fine chemical manufacturing. By simplifying the workflow, the method reduces the dependency on specialized equipment for filtration and drying of unstable intermediates, thereby lowering capital expenditure requirements for production facilities. This streamlined approach ensures a more consistent supply of high-quality intermediates, mitigating the risk of batch failures that can disrupt downstream API production schedules.
- Cost Reduction in Manufacturing: The primary driver for cost optimization in this process is the removal of the separation and purification unit operations for the phosphorus ylide intermediate. In traditional manufacturing, these steps consume significant amounts of energy, solvents, and labor hours. By adopting the one-pot method, manufacturers can achieve substantial cost savings through reduced utility consumption and lower waste disposal fees. The increased yield from roughly 59% to over 76% means that less starting material is required to produce the same amount of final product, directly improving the cost of goods sold (COGS) and enhancing profit margins for the final API.
- Enhanced Supply Chain Reliability: Supply chain resilience is heavily dependent on the robustness of the manufacturing process. The instability of the phosphorus ylide in conventional methods introduces a variable that can lead to unpredictable batch qualities and delays. The new method's reliance on stable, in-situ generated reagents removes this variability, ensuring that production timelines are met with greater certainty. This reliability is crucial for maintaining continuous supply to pharmaceutical partners, especially in the oncology sector where treatment interruptions are not an option. The simplified process also allows for faster turnaround times between batches, increasing overall facility throughput.
- Scalability and Environmental Compliance: As the demand for Olaparib grows globally, the ability to scale production efficiently is paramount. The one-pot synthesis is inherently more scalable because it reduces the number of unit operations, minimizing the potential for errors during scale-up. Furthermore, the reduction in solvent usage and waste generation aligns with increasingly stringent environmental regulations. By generating less hazardous waste, manufacturers can reduce their environmental footprint and avoid the high costs associated with waste treatment and regulatory compliance, making the process sustainable for long-term commercial production.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this patented synthesis method. These insights are derived directly from the experimental data and process descriptions found in CN114075143A, providing a clear understanding of how this technology improves upon existing standards. Understanding these details is vital for technical teams evaluating the feasibility of adopting this route for commercial manufacturing.
Q: How does the new one-pot method improve Olaparib purity compared to conventional routes?
A: The conventional route requires isolating an unstable phosphorus ylide intermediate, which degrades and causes side reactions leading to Impurity D. The new one-pot method avoids this isolation, significantly reducing Impurity D content to below 0.1% and achieving overall purity exceeding 99.9%.
Q: What are the scalability advantages of this synthesis protocol?
A: By eliminating the complex separation and purification of the phosphorus ylide intermediate, the process cycle is drastically shortened. This simplification reduces solvent consumption and waste generation, making the method highly suitable for large-scale industrial production ranging from 100 kgs to multi-ton batches.
Q: Why is controlling Impurity D critical in Olaparib manufacturing?
A: Impurity D is a structurally related dimer that is notoriously difficult to remove via standard purification techniques. High levels of this impurity compromise medication safety and regulatory compliance. The optimized process inherently suppresses its formation, ensuring a safer final API.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Olaparib Intermediate Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of high-purity intermediates in the development and production of life-saving oncology medications. Our technical team has extensively analyzed the pathways described in CN114075143A and possesses the expertise to implement this advanced one-pot synthesis on a commercial scale. We bring extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory innovation to industrial reality is seamless. Our state-of-the-art facilities are equipped with rigorous QC labs capable of detecting impurities at trace levels, guaranteeing that every batch of Olaparib intermediate meets stringent purity specifications required by global regulatory bodies.
We invite pharmaceutical partners to collaborate with us to leverage this cutting-edge technology for their supply chains. By choosing NINGBO INNO PHARMCHEM, you gain access to a Customized Cost-Saving Analysis tailored to your specific production volumes and quality requirements. We encourage you to contact our technical procurement team today to request specific COA data and route feasibility assessments. Together, we can drive down costs while elevating the quality standards of Olaparib manufacturing, ensuring a reliable supply of this essential medicine for patients worldwide.
