Advanced Synthetic Route for Olaparib Intermediates: Enhancing Purity and Commercial Scalability
The global demand for Poly (ADP-ribose) polymerase (PARP) inhibitors continues to surge as these agents become cornerstone therapies for ovarian, breast, and prostate cancers. At the heart of this therapeutic class lies Olaparib, a molecule whose complex heterocyclic structure presents significant challenges for efficient manufacturing. Patent CN112500379A introduces a transformative approach to Olaparib synthesis, centering on a novel intermediate that streamlines the construction of the phthalazinone core. This technical insight report analyzes the proprietary methodology disclosed in the patent, offering a deep dive into how this route addresses the critical pain points of toxicity, cost, and scalability that have long plagued traditional manufacturing processes. By leveraging a unique amide intermediate, the process bypasses the need for expensive transition metal catalysts and toxic coupling reagents, positioning it as a superior candidate for reliable pharmaceutical intermediates supplier networks seeking to optimize their supply chains.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industrial synthesis of Olaparib has been hindered by reliance on chemically aggressive and economically inefficient pathways. Prior art, such as the method disclosed in J. Med. Chem. 2008, relies heavily on HBTU (O-(Benzotriazol-1-yl)-N,N,N',N'-tetramethyluronium hexafluorophosphate) as a coupling agent in the final steps. While effective in small-scale laboratory settings, HBTU is notoriously toxic and generates substantial amounts of hazardous wastewater, creating a severe environmental burden and inflating post-treatment costs for manufacturers. Furthermore, alternative routes reported in academic literature often utilize palladium-catalyzed cross-coupling reactions involving boronic acids. These methods introduce the risk of heavy metal contamination, necessitating rigorous and costly purification steps to meet residual metal specifications required by regulatory bodies. Additionally, some existing routes suffer from low overall yields, reported as low as 48 percent in certain master thesis evaluations, rendering them economically unviable for multi-ton commercial production.
The Novel Approach
In stark contrast, the methodology outlined in CN112500379A pivots around the strategic utilization of a newly identified intermediate, structurally defined as a fluorinated benzamide derivative. This approach fundamentally alters the retrosynthetic logic by establishing the amide functionality early in the sequence through a mild alkaline hydrolysis of a nitrile precursor. This shift allows for the construction of the critical phthalazinone ring system via a straightforward hydrazine-mediated cyclization, avoiding the harsh conditions typically associated with ring-closing metathesis or oxidative cyclizations. The elegance of this route lies in its operational simplicity; it employs ubiquitous solvents such as water, acetonitrile, and toluene, and utilizes inexpensive inorganic bases like sodium hydroxide or potassium hydroxide. By eliminating the dependency on precious metal catalysts and toxic uronium salts, this novel approach not only enhances the safety profile of the manufacturing process but also drastically simplifies the downstream processing workflow.
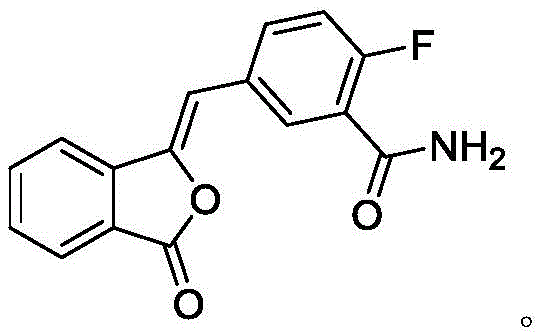
Mechanistic Insights into the Three-Step Cascade Synthesis
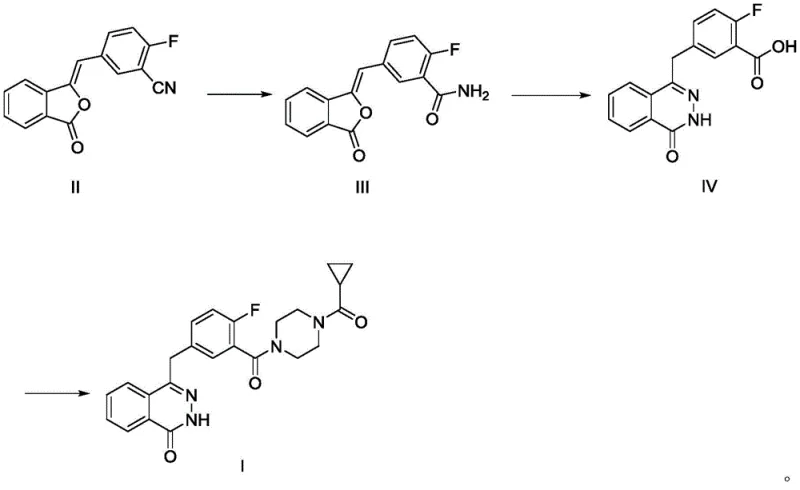
The core of this technological breakthrough is a robust three-step cascade that converts a simple nitrile starting material into the final active pharmaceutical ingredient with exceptional fidelity. The first stage involves the selective hydrolysis of the nitrile group in Compound II to form the primary amide, Compound III. This transformation is conducted under mild thermal conditions, typically between 20-30°C, using aqueous alkali. The mechanistic precision here is vital; the conditions are tuned to hydrolyze the nitrile without affecting the sensitive lactone or other functional groups, ensuring a clean conversion that minimizes the formation of carboxylic acid byproducts. Following isolation, Compound III undergoes a condensation reaction with hydrazine hydrate. This step is the linchpin of the synthesis, where the hydrazine nucleophile attacks the carbonyl centers to close the six-membered nitrogen-containing ring, forming the dihydrophthalazinone scaffold of Compound IV. This cyclization is driven thermally at 90-110°C, a range that provides sufficient energy for ring closure while maintaining stability of the fluorinated aromatic system.
The final stage involves the activation of the carboxylic acid moiety on the phthalazinone intermediate (Compound IV) followed by amidation with cyclopropylpiperazine. Unlike traditional methods that might use carbodiimides, this process utilizes thionyl chloride to generate the reactive acid chloride in situ. This activation is performed in the presence of catalytic amounts of DMF or DMAP, which accelerate the formation of the acyl chloride species. Subsequent addition of the amine nucleophile, cyclopropanoyl piperazine, under controlled cooling (5-20°C) ensures high regioselectivity and prevents thermal degradation of the product. The result is a highly efficient coupling that delivers Olaparib with purity exceeding 99 percent after simple recrystallization. This mechanistic pathway effectively sidesteps the generation of difficult-to-remove urea byproducts common in peptide coupling strategies, thereby enhancing the overall impurity profile of the final drug substance.
How to Synthesize Olaparib Efficiently
The synthesis of Olaparib via this novel intermediate pathway offers a streamlined protocol that is particularly well-suited for pilot and commercial scale operations. The process is characterized by its tolerance to a variety of solvent systems and its reliance on standard unit operations such as filtration, phase separation, and crystallization. For research and development teams looking to replicate or adapt this chemistry, the key lies in the precise control of pH during the work-up phases and the management of exotherms during the thionyl chloride addition. The following guide outlines the standardized operational framework derived from the patent examples, providing a foundational basis for process optimization.
- Suspend the nitrile precursor (Compound II) in a solvent such as water or acetonitrile, add an inorganic base like sodium hydroxide, and heat to 20-30°C to effect hydrolysis, yielding the amide intermediate (Compound III).
- Mix Compound III with hydrazine hydrate and a base in a solvent like water or acetonitrile, heat to 90-110°C, and maintain temperature to complete the cyclization reaction, forming the phthalazinone derivative (Compound IV).
- Activate Compound IV with thionyl chloride in the presence of a catalyst like DMAP, then react with cyclopropanoyl piperazine under controlled cooling (5-20°C) to obtain the final Olaparib product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this synthetic route translates directly into tangible operational efficiencies and risk mitigation. The elimination of palladium catalysts removes a significant variable from the supply chain, insulating production from the volatility of precious metal markets and the complexities of metal scavenging technologies. Furthermore, the substitution of toxic coupling reagents with thionyl chloride and common bases significantly lowers the cost of goods sold (COGS) by reducing raw material expenses and waste disposal fees. The use of water as a primary solvent in the early stages is particularly advantageous, offering a sustainable and cost-effective medium that reduces the facility's organic solvent load and fire hazard potential.
- Cost Reduction in Manufacturing: The economic impact of this route is profound, primarily driven by the removal of high-cost reagents. By avoiding the use of HBTU and palladium catalysts, manufacturers can achieve substantial cost savings on raw material procurement. Additionally, the simplified purification process, which relies on crystallization rather than column chromatography or complex extractions, reduces labor hours and solvent consumption. The high yield observed in the final coupling step further contributes to cost efficiency by maximizing the throughput of the most valuable intermediate, ensuring that every kilogram of input material generates a higher yield of saleable product.
- Enhanced Supply Chain Reliability: The robustness of this synthesis enhances supply chain continuity by utilizing commodity chemicals that are globally available. Solvents like toluene, acetonitrile, and water, along with reagents like sodium hydroxide and thionyl chloride, are produced at massive scales worldwide, minimizing the risk of supply disruptions. This contrasts sharply with routes dependent on specialized ligands or custom-synthesized boronic acids, which often have long lead times and limited supplier bases. The ability to source all critical inputs from multiple vendors ensures that production schedules remain uninterrupted, even in the face of regional logistical challenges.
- Scalability and Environmental Compliance: From an environmental, health, and safety (EHS) perspective, this route is designed for scalability. The reaction conditions are mild, operating at temperatures that do not require specialized high-pressure or cryogenic equipment, making it compatible with standard glass-lined or stainless steel reactors. The reduction in hazardous waste generation, particularly the absence of heavy metal sludge and toxic uronium byproducts, simplifies regulatory compliance and wastewater treatment. This "green chemistry" alignment not only reduces the environmental footprint but also accelerates regulatory approval timelines by presenting a cleaner impurity profile to health authorities.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this Olaparib synthesis technology. These insights are derived directly from the experimental data and comparative analysis provided in the patent documentation, offering clarity on the practical implications of adopting this novel intermediate strategy.
Q: What are the primary advantages of this Olaparib synthesis route over conventional methods?
A: This route eliminates the use of toxic coupling agents like HBTU and expensive palladium catalysts found in prior art. It utilizes cheap, readily available raw materials and mild reaction conditions, resulting in significantly reduced wastewater treatment costs and easier post-processing.
Q: What purity levels can be achieved with this novel intermediate method?
A: The described process consistently yields high-purity products. Experimental data indicates that the final Olaparib product can achieve a purity of greater than 99% after recrystallization, meeting stringent pharmaceutical specifications without complex chromatographic purification.
Q: Is this synthesis method suitable for large-scale industrial production?
A: Yes, the method is highly scalable. It avoids hazardous reagents and operates at moderate temperatures (20-110°C) using common solvents like water, toluene, and acetonitrile. The simple work-up procedures, such as filtration and acid-base extraction, facilitate efficient commercial scale-up.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Olaparib Intermediate Supplier
As the pharmaceutical industry increasingly demands greener and more cost-effective manufacturing solutions, the technology described in CN112500379A represents a significant leap forward in PARP inhibitor production. NINGBO INNO PHARMCHEM stands at the forefront of this innovation, leveraging our extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production to bring this advanced chemistry to market. Our state-of-the-art facilities are equipped to handle the specific solvent systems and thermal profiles required for this synthesis, ensuring that every batch meets stringent purity specifications through our rigorous QC labs. We understand that consistency is key in API manufacturing, and our process engineering team is dedicated to optimizing every step of this route to maximize yield and minimize variability.
We invite global partners to collaborate with us to unlock the full commercial potential of this efficient synthetic route. Whether you are looking to secure a stable supply of high-purity intermediates or require a Customized Cost-Saving Analysis for your existing Olaparib supply chain, our technical procurement team is ready to assist. Contact us today to request specific COA data and route feasibility assessments, and let us demonstrate how our expertise in fine chemical synthesis can drive value and reliability for your organization.
