Scalable Production of High-Purity L-Valine and D-Valine via Advanced Chemical Resolution
The landscape of chiral amino acid manufacturing has undergone a significant transformation with the introduction of robust chemical resolution technologies, specifically detailed in patent CN1186313C. This pivotal intellectual property outlines a highly efficient method for preparing L-valine and D-valine, addressing critical bottlenecks in yield and purity that have historically plagued the industry. By utilizing racemic valine (DL-Val) as the starting material and employing dibenzoyl tartaric acid (DBTA) isomers as resolving agents within a dilute inorganic acid medium, this process achieves a level of operational simplicity and economic viability that earlier techniques could not match. For R&D directors and procurement specialists seeking a reliable amino acid intermediate supplier, understanding the mechanistic advantages of this acid-mediated resolution is paramount, as it eliminates the need for toxic precursors and complex enzymatic systems while delivering superior stereochemical control.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the production of optically active valine enantiomers relied heavily on induced crystallization or microbial asymmetric conversion, both of which present substantial industrial drawbacks. Early induced crystallization methods, such as those reported by Susenm Tatsumi in 1965, suffered from critically low yields, often falling below 10%, and required prolonged processing times that were incompatible with high-throughput manufacturing environments. Furthermore, the optical purity achieved through these legacy crystallization techniques was frequently inconsistent, rarely exceeding 93% ee, necessitating costly recrystallization steps that further eroded overall process efficiency. On the biological front, while microbial asymmetric conversion offered a pathway to D-valine, it introduced severe safety and supply chain risks due to the requirement for highly toxic precursors like hydrocyanic acid to synthesize the necessary hydantoin substrates. These conventional approaches created significant barriers to entry for cost reduction in chiral amino acid manufacturing, as they demanded specialized containment facilities and generated hazardous waste streams that complicated regulatory compliance and environmental stewardship.
The Novel Approach
In stark contrast, the novel chemical resolution process described in the patent data leverages the distinct solubility differences of diastereomeric salts formed in an acidic aqueous environment to achieve rapid and high-yielding separation. By dissolving DL-valine and the chiral resolving agent (D-DBTA or L-DBTA) in a dilute inorganic acid solution, such as hydrochloric or sulfuric acid, the system facilitates the selective precipitation of the less soluble diastereomeric salt upon cooling. This approach operates under relatively mild thermal conditions, typically between 60-100°C, and utilizes inexpensive, non-toxic solvents that drastically simplify downstream processing and waste management. The result is a streamlined workflow that not only boosts yields to over 70% but also ensures optical purities exceeding 98%, making it an ideal candidate for the commercial scale-up of complex pharmaceutical intermediates where consistency and safety are non-negotiable requirements for global supply chains.
Mechanistic Insights into Diastereomeric Salt Resolution
The core of this technology lies in the precise manipulation of intermolecular forces and solubility equilibria within an acidic medium. When racemic valine interacts with a chiral resolving agent like D-dibenzoyl tartaric acid (D-DBTA), two distinct diastereomeric salts are formed: D-Val·D-DBTA and L-Val·D-DBTA. Due to the specific three-dimensional arrangement of atoms in these complexes, the D-Val·D-DBTA salt exhibits significantly lower solubility in the dilute inorganic acid solvent compared to its L-Val counterpart. As the reaction mixture is heated to ensure complete dissolution and then gradually cooled, the thermodynamic drive favors the crystallization of the less soluble D-Val·D-DBTA salt, effectively pulling the D-enantiomer out of the solution while leaving the L-enantiomer in the mother liquor. This differential solubility is the fundamental driving force that allows for the physical separation of the enantiomers through simple filtration, bypassing the need for expensive chromatographic columns or enzymatic reactors.
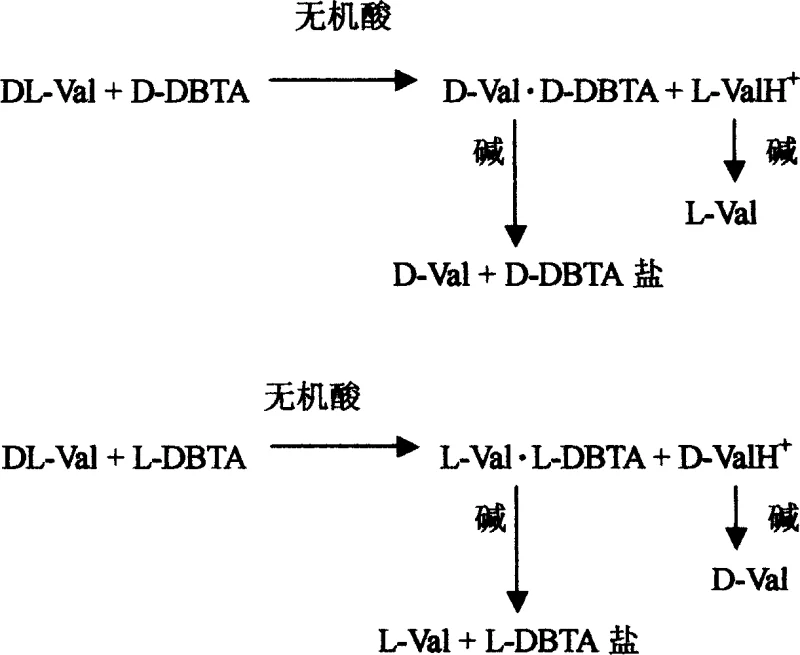
Following the initial separation, the recovery of the free amino acid is achieved through a straightforward acid-base neutralization reaction. The isolated diastereomeric salt is treated with a base, such as ammonia, triethylamine, or sodium hydroxide, in a solvent system comprising water and lower aliphatic alcohols. This neutralization step breaks the ionic bond between the valine and the tartaric acid derivative, regenerating the free amino acid and forming a water-soluble tartrate salt byproduct. Crucially, the resolving agent (DBTA) can be recovered from the aqueous phase and recycled for subsequent batches, which is a key factor in minimizing raw material costs. The mother liquor, which contains the enriched opposite enantiomer, can also be processed by adjusting the pH to approximately 5.5 to precipitate the remaining valine, ensuring that both D-valine and L-valine are harvested from a single batch of racemic starting material with minimal waste.
How to Synthesize L-Valine and D-Valine Efficiently
To implement this resolution strategy effectively, manufacturers must adhere to strict parameters regarding solvent concentration, temperature gradients, and molar ratios to maximize the yield and optical purity of the final product. The process begins by selecting the appropriate resolving agent isomer to target the desired enantiomer, followed by dissolution in a dilute inorganic acid with a concentration range of 0.1 to 1.0 mol/L. Precise control over the cooling rate is essential to promote the formation of large, filterable crystals of the diastereomeric salt, while the subsequent neutralization must be performed under conditions that prevent racemization. For a detailed breakdown of the specific operational parameters, including exact temperatures, stirring times, and workup procedures, please refer to the standardized synthesis guide below.
- Dissolve racemic valine (DL-Val) and the resolving agent (D-DBTA or L-DBTA) in a dilute inorganic acid solution at a molar ratio of 1: 0.5-1.2.
- Heat the mixture to 60-100°C and stir for 0.5-2.0 hours to ensure complete salt formation and dissolution.
- Cool the solution gradually to room temperature to precipitate the less soluble diastereomeric salt, then filter and neutralize with alkali to recover the pure enantiomer.
Commercial Advantages for Procurement and Supply Chain Teams
From a strategic procurement perspective, this chemical resolution methodology offers profound advantages over traditional biosynthetic or crystallization routes, primarily driven by the simplification of the supply chain and the reduction of hazardous material handling. By eliminating the dependency on toxic cyanide-based precursors required for microbial methods, companies can significantly lower their insurance premiums, waste disposal costs, and regulatory compliance burdens associated with hazardous chemical storage. Furthermore, the ability to recycle the dibenzoyl tartaric acid resolving agent means that the effective cost per kilogram of the chiral auxiliary is drastically reduced over time, transforming what is typically a high-cost consumable into a reusable asset. This circular economy approach within the manufacturing process directly contributes to substantial cost savings without compromising the quality or purity of the final amino acid product, making it highly attractive for long-term supply contracts.
- Cost Reduction in Manufacturing: The utilization of dilute inorganic acids like hydrochloric or sulfuric acid as the primary solvent represents a major departure from expensive organic solvents or complex fermentation broths. These acids are commodity chemicals with stable pricing and widespread availability, ensuring that the variable costs associated with solvent procurement remain low and predictable. Additionally, the high yield of the process, often exceeding 70% for both enantiomers, means that less raw racemic valine is required to produce a given amount of optical pure product, thereby improving the overall atom economy and reducing the cost of goods sold (COGS) for the final API or intermediate.
- Enhanced Supply Chain Reliability: Unlike enzymatic processes that rely on the stability and activity of biological cultures, which can be susceptible to contamination or batch-to-batch variability, this chemical resolution relies on robust physicochemical principles that are easily reproducible at any scale. The raw materials, including DL-valine and DBTA isomers, are commercially available from multiple global sources, mitigating the risk of single-supplier dependency. This redundancy in the supply base ensures reducing lead time for high-purity amino acid intermediates, as production schedules are not held hostage by biological fermentation cycles or the availability of specialized enzymes, allowing for more responsive inventory management.
- Scalability and Environmental Compliance: The process is inherently scalable because it relies on standard unit operations such as heating, cooling, filtration, and neutralization, which are well-understood and easily implemented in existing multipurpose chemical plants. The absence of heavy metal catalysts or toxic organic byproducts simplifies the wastewater treatment process, as the effluent primarily consists of benign inorganic salts and recoverable organic acids. This environmental profile facilitates easier permitting and aligns with the increasingly stringent sustainability goals of multinational pharmaceutical corporations, positioning this technology as a future-proof solution for high-purity valine enantiomers production.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this resolution technology, derived directly from the patent specifications and experimental data. Understanding these nuances is critical for technical teams evaluating the feasibility of integrating this process into their existing manufacturing lines or for procurement officers assessing the quality assurances of potential suppliers. The answers provided reflect the specific capabilities of the DBTA-mediated resolution method in dilute acid media.
Q: What is the optical purity achievable with this DBTA resolution method?
A: Under optimized conditions using dilute inorganic acid solvents, this process consistently achieves optical purities greater than 98%, with specific embodiments reaching 100% ee.
Q: Can the resolving agent be recovered after the separation process?
A: Yes, the dibenzoyl tartaric acid (DBTA) resolving agent can be effectively recovered and recycled after the neutralization step, significantly reducing material costs.
Q: How does this method compare to microbial asymmetric conversion?
A: Unlike microbial methods that require toxic precursors like hydrocyanic acid, this chemical resolution uses safe, inexpensive inorganic acids and avoids hazardous reagents entirely.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable L-Valine and D-Valine Supplier
At NINGBO INNO PHARMCHEM, we recognize that the transition from laboratory-scale discovery to commercial production requires a partner with deep technical expertise and a commitment to quality excellence. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the intricate balance of temperature, pH, and crystallization kinetics required for this valine resolution process is maintained with absolute precision. We operate stringent purity specifications and utilize rigorous QC labs to verify that every batch of L-valine and D-valine meets the >98% optical purity standards demanded by the global pharmaceutical and nutraceutical industries, providing our clients with the confidence they need to move forward with their drug development pipelines.
We invite you to engage with our technical procurement team to discuss how this advanced resolution technology can be tailored to your specific project requirements. By requesting a Customized Cost-Saving Analysis, you can gain deeper insights into how switching to this chemical resolution method might optimize your current supply chain economics. We encourage potential partners to contact us directly to obtain specific COA data and route feasibility assessments, allowing us to demonstrate our capability as a trusted provider of high-quality chiral building blocks for your most critical applications.
